Introduction
The study on Experimental measurement of thermodynamic properties and phase equilibrium for some systems is very difficult since these systems may be costly. For these systems using modeling and simulation is of great interest. This modeling and simulation reduces the time and to find equilibrium conditions for binary and multi-component systems. J.W. Gibbs was established the relationship between thermodynamics and phase equilibria [1].
Iron-cobalt alloy has magnetic properties, with high Curie temperatures, the highest saturation magnetizations and high permeability etc. Elmen discovered these alloys in 1929 [2].
Also (Fe-Co) alloy has been studied by Ellis and Greiner [3]. Normanton et al [4]. In 2002 by Ohnuma et al [5]. This alloy has applications in electric engine, which contain magnetic materials [6]. And it is now a good thermodynamic explanation of it.
Thermo-Calc is powerful and flexible software to performing various kinds of thermodynamic and phase diagram calculations. This software can helpful to solve difficult problems interaction of many elements and phases that shows highly non ideal behavior [7].
The first version of thermo-calc was shown in 1981. In 2002 modern version of thermo- calc was present. The first description on thermo-calc was in 1985 [8].
Calphad method
Calphad stands for calculation of phase diagram. The Calphad method is used to find the thermodynamic properties of different materials system. The CALPHAD method was established as tool for phase equilibria of different multicomponent systems and for treating thermodynamics. For the calculations, the modules reported are used [9]. CALPHAD method has been employed for the material properties [10]. Recently the Calphad approach has been applied to a systems having more phase-based properties, as reported molar volume, elastic moduli and the database for processing these functions have been developed [11]. For materials the Calphad method can be used to describing the composition, temperature of optical activity, thermo-electric and acoustic properties [12]. All the calculations have been done by Calphad method.
Results and Discussion
A thermodynamic calculation in Fe-Co has been done using Thermo-Calc Software and Databases. The results of calculated activities in (Fe-Co) at 1200 K, 1225 K and 1250 K are presented in fig1. Gibbs energy for the investigated binary system is shown in figure2. In this paper the reaction, equilibria, phase diagram and figures modules were used.
In liquid (Fe-Co) alloy there negative deviation can occur in activities. The activities increase proportionally with increasing of the temperature and deviation decreases in activity according to the Raoults law [13].
Activities show that there is strong interaction between the atoms of the alloy. Considering calculated integral thermodynamic properties both the alloys show negative values for Gibbs energy which represents the stability of (Fe-Co) alloy according to [14]. Gibbs energy decreases with increasing the temperature.
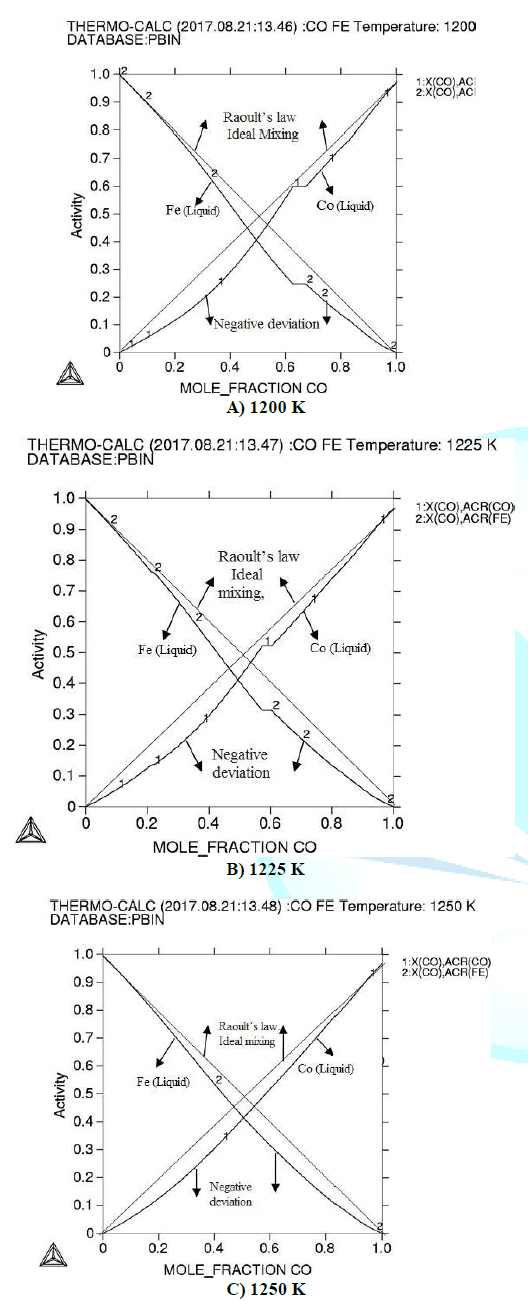
Figure 1: Activity of Fe-Co alloy at different temperatures (a) 1200 K (b) 1225 K (c) 1250 K.
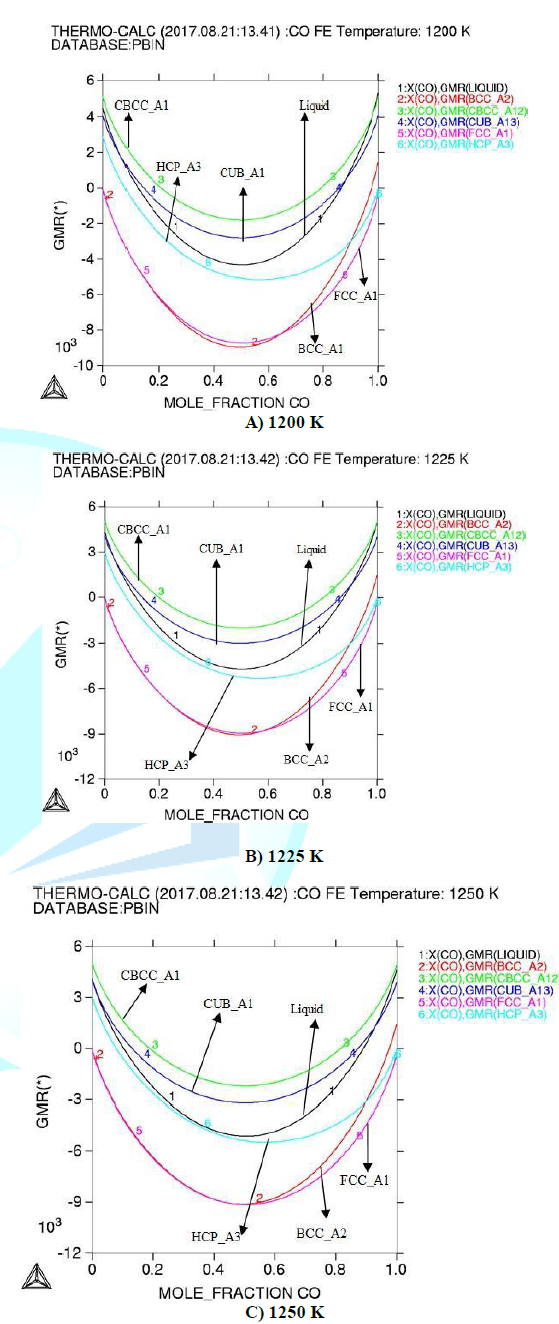
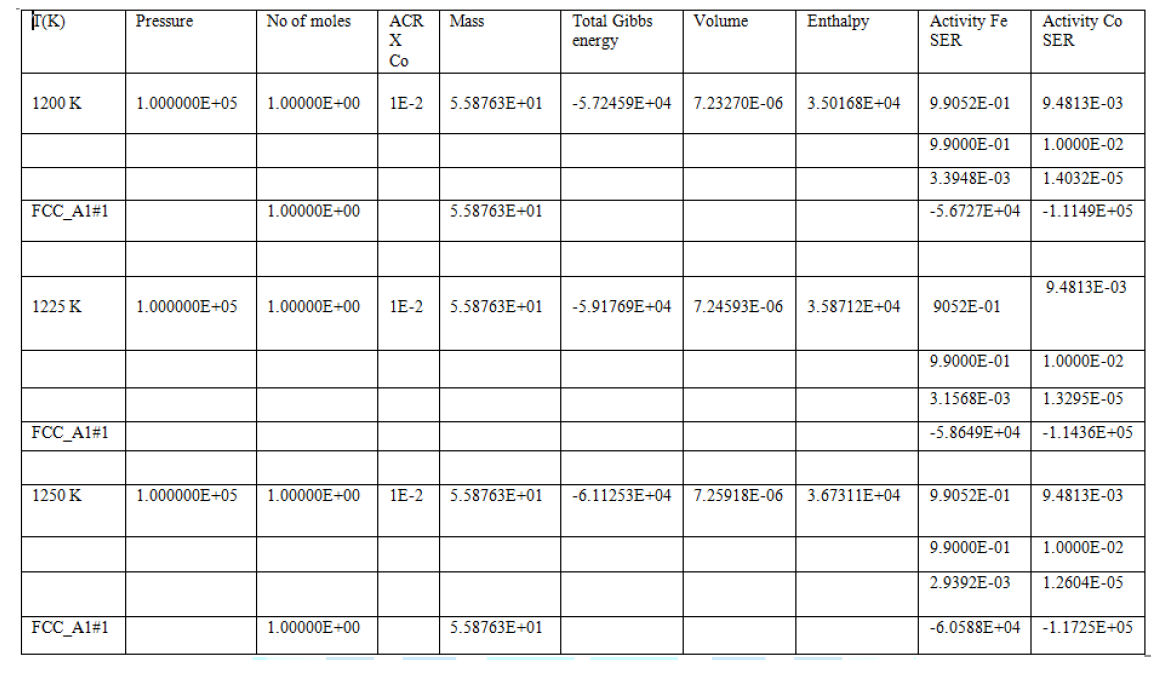
Table1: Various Thermodynamic properties of (Fe-Co) alloy at 1200 K, 1225 K and 1250 K.
Phase diagrams of the binary systems obtained by thermo-calc software and databases are shown in Fig3. In phase diagrams different phases can occur at different temperatures and at various concentrations. Comparison with referent data shows good accordance with available phase diagrams in literature.

Figure 3: Phase diagram of Fe-Co alloy.
In the above Table 1 pressure and mass are constant. However other thermodynamic properties i.e. Gibbs free energy, activity, enthalpy and volume are change at different temperatures. In table it is clear that increasing the temperatures then activity increases and total Gibbs energy decreases, which shows proportionality between temperature and thermodynamic parameters. The highest negative value of Gibbs energy is observed at 1250 K showing stability of (Fe-Co) binary alloys system; the enthalpy proportionally increases with temperature due to the fact that the heat content of the system increases with temperature. At the three temperatures (1200 K, 1225 K and 1250 K) there (FCC_Al#l) phase can occurs, which is the austenite phase shows solid solution of the alloy (Fe-Co). At 1250 K we observed maximum enthalpy, which shows the capability of system at high temperature and heat provide high stability of alloys.
Conclusion
A thermodynamic property of Ferric base alloy system (Fe-Co) alloy has been found by Thermo-Calc software. It is indicated that thermo-calc software play important role in the design of structure and functional of Fe base alloy. The thermodynamic calculations in binary alloy (Fe- Co) at 1200 K, 1225 K and 1250 K were determined. The binary system has negative values for Gibbs energy. The total Gibbs energy decreases with increasing temperature. At 1250 K, highest negative value of Gibbs energy in (Fe-Co) alloy system is observed which shows that the system is more stable, which increasing the level of stability, hardness, wear resistance, corrosion resistance and other doping characteristics of alloy. Activity values of the components are less than unity and show negative deviation from the Raoults law. Increase the temperature then activity increases and deviation in activity decreases, which indicates that there is strong interaction between the atoms of (Fe-Co). At 1250 K it has better accordance with Raoults law in Fe-Co binary system, which is important for engineering technology and metallurgy.
A Phase diagrams for the investigated binary system is calculated by the use of Thermo- Calc software and database. Obtained results presented one good base for the further thermodynamic analysis and developing in this group of innovative Fe base alloys. The obtained and shown results for the (Fe-Co) system provide complete thermodynamic optimization of this system.
References
1. J Hertz. Josiah Willard Gibbs and Teaching Thermodynamics of Material (History) (1992) J Phase Equilibria 13: 450-458. https://doi.org/10.1007/BF02665759
2. GW Elmen. Magnetic material and appliance (1929) US patent No. 1: 739-752.
3. Ellis WC and Greiner ES. Equilibrium relations in the solid state of their on-cobalt system (1941) Trans AS 415-433.
4. Normanton AS, Bloomfield PE, Sale FR and Argent BB. A calorimetric study of iron-cobalt alloys (1975) Metal Sci 9: 510-517. http://dx.doi.org/10.1179/030634575790444658
5. Ohnuma I, Enoki H, Ikeda O, Kainuma R, Ohtani H, et al. Phase equilibria in the Fe-Co binary system (2002) Acta Mater 50: 379-393. https://doi.org/10.1016/S1359-6454(01)00337-8
6. Fingers RT and Rubertus CS. Application of high temperature magnetic materials (2000) IEEE Trans Magnetics 36: 3373-3375. https://doi.org/10.1109/20.908805
7. Andersson JO, Helander T, Hoglund L, Shi P and Sundman B. Thermo-Calc & DICTRA, computational tools for materials science (2002) Calphad 26: 273. https://doi.org/10.1016/S0364-5916(02)00037-8
8. Sundman B, Jansson B and Andersson JO. The Thermo-Calc databank system (1985) CALPHAD 9: 153-190. https://doi.org/10.1016/0364-5916(85)90021-5
9. Xiong W, Hedstrom P, Selleby M, Odqvist J and Thuvander M. An Improved Thermodynamic Modeling of the Fe-Cr System Down to Zero Kelvin Coupled with Key Experiments (2011) Calphad 35: 355. http://dx.doi.org/10.1016/j.calphad.2011.05.002
10. Saunders N, Kucherenko S, Li X, Miodownik AP and Schille J. A new computer program for Predicting materials properties (2001) J Phase Equilibria 22: 463-469. https://doi.org/10.1361/105497101770333036
11. Hallstedt B, Dupin N, Hillert M, Hoglund L, Lukas H, et al. Thermodynamic models for crystalline phases. Composition dependent models for volume, bulk modulus and Thermal expansion (2007) CALPHAD 31: 28-37. https://doi.org/10.1016/j.calphad.2006.02.008
12. Gheribi A and Charrand P. Application of the CALPHAD method to predict the thermal conductivity in dielectric and semiconductor crystals (2012) CALPHAD 39: 70-79. https://doi.org/10.1016/j.calphad.2012.06.002
13. Kostov A, Friedrich B and Zivkovic D. THERMODYNAMIC CALCULATIONS IN ALLOYS Ti-Al, Ti-Fe, Al-Fe and Ti-Al-Fe (2008) J Mining and Metallurgy 44: 49-61. DOI:10.2298/JMMB0801049K
14. Waseem Ullah Shah, Syed Mehmood Shah, Matiullah Khan, Dil Faraz Khan, Athanasios G. Mamalis, et al. Thermodynamic Study Of Binary Alloy System (Co-Cr) Using Calphad Method (2018) Surface Review and Letters. 25: 1850049. https://doi.org/10.1142/S0218625X1850049X
Keywords
CALPHAD; (Fe-Co) binary alloys; Thermo-Calc software; Phase diagram; Activity curve; Gibbs curve, Thermodynamic calculation.


 PDF
PDF