Introduction
The pressure ulcer is a cutaneous lesion of ischemic origin linked to a compression of the soft tissues between a hard plane and the bony protrusions [1,2], met mainly in patients hospitalized in resuscitation or those which require a long term institutional care. The incidence of pressure ulcers varies widely depending on the clinical setting [3-7]. In fact, a multidisciplinary process improvement program was initiated at the University of Miami Hospital in 2009 to identify the prevalence of hospital-acquired pressure ulcers at the institution and to implement interventions to reduce the incidence of pressure ulcers. As a result, the rate of pressure ulcers dropped to 2.76% by the second quarter of 2012 and has remained steadily low at 1%–2% for nine consecutive quarters [3].
They impose a heavy burden not only on the patient, but on the entire health system. Reducing their frequency is an important component of the current goals for patient safety as evidenced by the Institute for Healthcare Improvement (IHI) [8-10], especially that they can be avoided or at least diminished, by simple actions and reflexes to adopt. In fact, prevention of pressure ulcers was one of the six new interventions adopted by The 5 Million Lives Campaign [10]. This is why it is important to define the factors that lead to its constitution and to codify the conduct to be held in both preventive and curative way. In Tunisia, the prevalence of pressure ulcers in a university hospital has been estimated at 5.3% [11]. However, for the risk factors associated with their occurrence, only univariate analysis was performed. The purpose of our work was to determine the incidence of pressure ulcers at Sahloul Hospital, to study its independent risk factors, and to evaluate assessment scales for pressure ulcers.
Methods
Setting, type and study population
We conducted a longitudinal observational incidence study at Sahloul hospital in Sousse over a 3-month period from October the 1st, 2015 to December 31st, 2015. This study is a pilot study and is voluntary we have contacted all long-stay departments of Sahloul Hospital and hosting patients who may be considered at risk of developing a pressure ulcer. Only departments of Anesthesia-Resuscitation, Internal Medicine, and Aesthetic and Restorative Surgery agreed to participate. Thus, we included all hospitalized patients in these at-risk services during the study period.
Data collection
Data collection was established through the observational analysis of patient data using an operating record filled at the patients bed after questioning and examination of the patient, consultation of his medical file and the interview with the care team. The data collected were related to patients socio demographic characteristics (age, socioeconomic level, associated co-morbidities, risk factors, etc.), the characteristics of the ulcer (location, size, associated signs, etc.), as well as the treatment received and/or in progress. We used the Mini Mental State Examination (M.M.S.E) to assess the cognitive functions [12]. Nutrition status was assessed via serum albumin level taking into account the of C-reactive protein levels. The undernutrtion was defined as albuminemia less than 35 g/l [13].
To evaluate the risk of pressure ulcer occurrence, we used three types of scales: Norton scale [14], Braden scale [15-17], and Water low scale [18]. For the classification of pressure ulcers, we used the staging of the National Pressure Ulcer Advisory Panel [19].
Statistical analysis
Statistical analysis was performed using SPSS software package (version 20.0, SPSS Inc, Chicago, IL, USA). Continuous variables were described as means ± standard deviations, and compared using the student t-test. Categorical variables were summarized with absolute and relative frequencies, and compared using the chi-squared test (or Fisher exact tests where appropriate). All variables with a significance level ≤ 20%, in this analysis, were included in the multi-variate analysis. The latter was performed according to the stepwise binary logistic regression model. Significance was considered at P-value<0.05.
Results
Patients characteristics
During the study period, 150 patients were enrolled in the three departments. Their average age was 46.1 ± 20.2 years with extremes ranging from 4 to 86 years old. A male predominance was noted (n=99, 66%). More than half of the patients (58.7%) were hospitalized in the department of anesthesia-resuscitation, 32.6% were hospitalized in the department of internal medicine, and 8.7% in the department of reconstructive surgery.
The mean length of stay was 8.3 ± 4.5 days. Most common medical histories were diabetes (15.4%) and high blood pressure (14.7%). The most common underlying disease was polytrauma (n=52, 34.7%). Out of 150 enrolled patients, 51 had superior function disorders, 30 had deep sensitivity disorders and 10 had tactile and thermo-algesic sensitivity disorders. On the other hand, 76 patients (50.7%) had under nutrition during their hospitalization, and 22 patients (15%) had a fluid intake<1.5 liters/24 hours. A nutritional compliment was administered to 55 patients (36.7%). We recorded 4 cases of complications of venous thrombosis of the lower limbs. Only 14 patients (9.3%) benefited from compression stockings, and 58 patients (38.7%) benefited from rehabilitation. Half of the patients benefited from a change of position every 3 hours.
Risk assessment scales of pressure ulcer (Table 1)
In our study, the mean Braden score was 15 ± 6.8 (6-23). One in four patients had a very high risk of pressure ulcers (Braden score ≤ 7). The mean Norton score was 12.6 ± 5.3 (5-19). In addition, 46% of patients had a high risk of pressure ulcers (Norton score<12). The mean Waterloo score was 11 ± 7.1 (2-29). In addition, 12.7% of patients had a high risk of pressure ulcers (> 20).
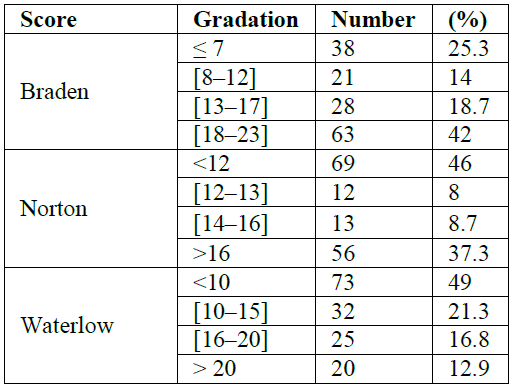
Table 1: Distribution of patients according to different scales.
Incidence of pressure ulcer
In our study, 56 episodes of pressure ulcers were observed in 29 patients, which correspond to a patient incidence of 19.3% (95% CI: [13-25.3]), and a pressure ulcer incidence of 37% (95% CI: [29.3 - 44.7]). The mean time to onset of pressure ulcers was 6.5 ± 2.5 days with extremes ranging from 2 to 10 days. The most frequent locations were heels (41%) and buttocks (30.4%), as shown in Table 2. In our study, 20 patients had a Stage IV of pressure ulcer, which corresponds to 69% of patients and 83.9% of identified pressure ulcers (Table 3).
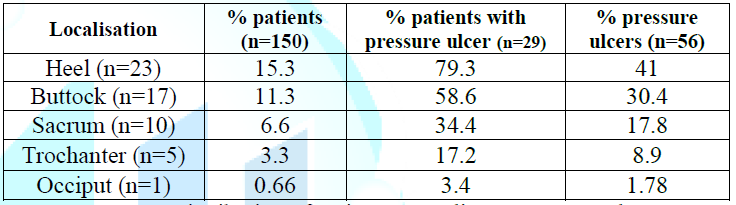
Table 2: Distribution of patients according to pressure ulcer localization.
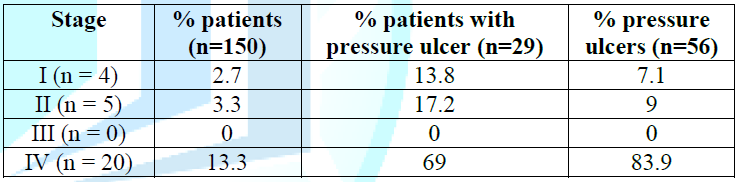
Table 3: Distribution of patients according to pressure ulcer stage.
Risk factors of pressure ulcer
In univariate analysis, several factors were associated with the occurrence of pressure ulcer (Table 4). However, in multivariate analysis, only polytrauma (p=0.048, RR=2.88, 95%CI=[1.09-8.2]) and disorders of high functions (p<10-3, RR=11.17, 95%CI=[3, 58-35,1]) were identified as independent risk factors of the occurrence of pressure ulcers. In our study, the Braden scale had the best sensitivity/specificity pair and a better diagnostic orientation (Y=0.95) (Table 5).
Discussion
In our series, 29 patients were affected which corresponds to a patient incidence of 19.3% (95% CI: [13-25.3].) In total, 56 episodes of pressure ulcers were observed in these patients, which corresponds to a pressure ulcer incidence of 37% (95% CI: [29.3-44.7]), and polytrauma (OR=2.88, p=0.048) and functional disorders. (OR=11.17, p<10-3) were the independent risk factors for pressure ulcers.
Incidence of pressure ulcers
Our rate is in the range reported in the literature: In Europe, the incidence varies considerably from one country to another and the results reported were a source of controversy in the same country. Indeed, Lahmann et al., [20], based on two different data collection systems AQUA Institute and Kinexus, reported incidences of 1.3% and 6.7% respectively. They concluded that the first was probably underestimated. Another recent German study, for a larger sample, was even lower than the previous ones (0.78%) [21]. In Nigeria, alarming figures have been reported [22, 23].
Table 4: Univariate analysis of factors associated with pressure ulcer.
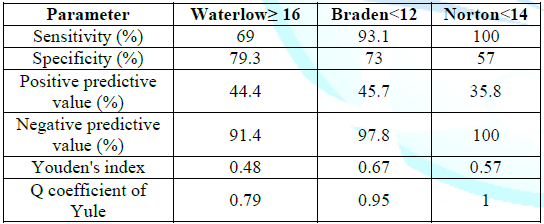
Table 5: Evaluation of the three scales.
The authors attributed these very high incidences to the insufficient knowledge of caregivers about preventive measures, the lack of a clear strategy and policy and the lack of means to fight against this skin pathology. Thus, the incidence rate varies considerably from one region of the world to another and from one country to another (0.04%- 87.5%). In Spain, the incidence has increased from 16% [24] in 2001 to 26.7% in 2006 [25]. This difference could be related to the nature of the patients recruited, the methodological differences, the nature of hospitals and the duration of patient follow-up (5 months vs 12 months). The situation is more alarming in developing countries [22, 23]. At the end of this literature review, our rate remains relatively high although it is much lower than those reported in developing countries. Several explanations find their places:
(i) Our study was conducted in high-risk services to develop pressure ulcers
(ii) The workload is important in these services with a nurse-to-patient ratio well below that recommended
(iii) Poor perception of pressure ulcer disease
(iv) In our hospital, like the rest of the country, we do not have standardized protocols for prevention of pressure ulcers despite the latest efforts to develop them
(v) The lack of an adequate risk management approach in our establishment
(vi) The lack of a national policy aimed at improving the quality of care despite the creation of the National Institutes of Health Accreditation (INASante).
Factors associated with the occurrence of pressure ulcers
In our study, polytrauma (OR=2.88, p=0.048) and higher function disorders (OR=11.17, p<10-3) were the independent risk factors for pressure ulcers. More than 100 factors associated with pressure ulcers have been identified in the literature [4]. With regard to gender, the majority of studies did not show a significant association with the occurrence of pressure ulcers [11]. However, it is currently known that advanced age increases the risk of pressure ulcer because it is strongly correlated with low body mass index and undernutrition [26, 27]. Two studies have shown that the reduction of mobility was predictive of the presence of pressure ulcers [28].
Indeed, any pathology leading to immobility of the patient could favor the occurrence of pressure ulcers such as those mentioned in our study: polytrauma and disorders of higher functions. These latter two are among the list of intrinsic risk factors used by the Royal College of Nursing (United Kingdom) [29]. It includes: disorders of consciousness and/or motility, malnutrition, urinary and fecal incontinence, the quality of the skin, the decrease of circulatory flow, arterial hypotension, sensory and motor neuropathy, the psychological condition and lack of motivation to participate in care, and age. Several arguments have been put forward to explain these different results: the difference in methodology, the variables studied, as well as the measurement tools used on the one hand and the demographic specificities and characteristics of the populations studied on the other. The populations studied come from different services and are disparate. The size of the samples varies from the scale of a unit of care to that of groups of hospitals. The information is collected either by direct observation, retrospective examination of patients files, or by sending questionnaires to care teams. Thus, these differences do not facilitate comparison of studies.
Evaluation of scales
In our study, the Braden scale had the best sensitivity-specificity pair. In fact, the risk factors specific to some situations or particular diseases must be taken into account individually and continuously. Risk scales will always be used where they have shown efficacy in clinical practice as a complementary tool. For example, Norton or Braden scales have been developed in acute care hospital settings, as well as in nursing homes, Waterlow scale and other scales in the field of paraplegiology, and are mainly used in these centers. Only the Norton and Braden scales are reported in the Agency for Health Care Policy and Research guideline and are therefore considered appropriate for determining the risk of pressure ulcers. The Braden scale is considered in the literature to be sufficiently reliable, because of its sensitivity and specificity, to be used in routine practice to determine if a patient is at risk for developing a pressure sore or not at risk [11, 30]. The sensitivity being 100% and the specificity of 64-90%, the scale was then considered reliable. Pancorbo-Hidalgo et al., [17], by assessing the sensitivity and specificity of the most used scales, concluded that the Braden scale has sufficient validity and a balance between its sensitivity (75%) and its specificity (67.5%), compared to the Norton and Waterlow scales.
In general, the selected risk assessment system must be adapted to the patient. Assessment should be made not only at the patients admission, but also during changes in the patients clinical condition [15]. It must allow the rapid establishment of prevention means. These scales should be seen as complementary to clinical judgment and not as an isolated tool, independent of other clinical factors. A reasonable risk assessment should therefore include the use of a risk scale associated with a clinical assessment of skin condition [31].
Limits
The main limitations of our work are the small size and the heterogeneous nature of our population between the different departments. On the other hand, some characteristics are specific to our context, the lack of means and availability comes in chief line.
Conclusion
Our study allowed us to collect specific data of Sahloul hospital, which, moreover, coincide on several points with those of the studies carried out on an international level. The predictive factors for the occurrence of pressure ulcers found in our study, which are polytrauma and disorders of the higher functions, push us to give them a greater value in the evaluation in order to better prevent the risk.
References
1. Jahnigen D, Baxter C and Bodenbender R. Pressure ulcers prevalence, cost and risk assessment: Consensus development conference statement (1989) Decubitus 2: 24-30.
2. Anaes P. Editor treatment of pressure ulcers in adults and elderly subjects, Consensus Conference Proceedings 2001.
3. Amparo C, Debbies A, Hope S, Fortunata J, Jennifer AL, et al. Improving outcomes by implementing a pressure ulcer prevention program (PUPP): Going beyond the Basics (2015) Healthcare 3: 574-585. https://doi.org/10.3390/healthcare3030574
4. Clinical guideline [CG179], Pressure ulcers: Prevention and management. NICE 2014.
5. Schoonhoven L, Bousema MT and Buskens E. The prevalence of pressure ulcers in hospitalised patients in a university hospital in The Netherlands: A prospective inception cohort study (2007) Int J Nurs Stud 44: 927-935. https://doi.org/10.1016/j.ijnurstu.2006.02.011
6. Xiao-Lin Z and Fan-Jie M. A care bundle for pressure ulcer treatment in intensive care units (2015) INSS 2: 340-347. https://doi.org/10.1016/j.ijnss.2015.10.008
7. Bours GJ, De Laat E, Halfens RJ and Lubbers M. Immobility-a major risk factor for development of pressure ulcers among adult hospitalized patients: A prospective study (2004) Scand J Caring Sci 18: 57-64. https://doi.org/10.1046/j.0283-9318.2003.00250.x
8. Duncan KD. Preventing pressure ulcers: The goal is zero (2007) Jt Comm J Qual Patient Saf 33: 605-610.
9. Armstrong DG, Ayello EA, Capitulo KL, Fowler E, Krasner D, et al. New opportunities to improve pressure ulcer prevention and treatment: implications of the CMS inpatient hospital care present on admission indicators/hospital-acquired conditions policy: A consensus paper from the International Expert Wound Care Advisory Panel (2008) Adv Skin Wound Care 21: 469-478. https://doi.org/10.1097/01.ASW.0000323562.52261.40
10. McCannon CJ, Hackbarth AD and Griffin FA. Miles to go: An introduction to the 5 Million Lives Campaign (2007) Jt Comm J Qual Patient Saf 33: 477-484.
11. Ben Mbarka F, Ben Jeddou K, Khalfallah M, Jarraya D, Jarraya H, et al. Prevalence and risk factors of pressure ulcers in a Tunisian hospital (2017) Tunis Med 95: 494-499.
12. Folstein MF, Folstein SE and McHugh PR. Mini-mental state: A practical method for grading the cognitive state of patients for the clinician (1975) J Psychiatr Res 12: 189-198. https://doi.org/10.1002/(SICI)1099-166(199805)13:5<285AID-GPS753>3.0.CO2-V
13. Patry C and Raynaud-Simon A. Undernutrition: What prevention strategies? (2010) Gerontology and society 33: 157-170. https://doi.org/10.3917/gs.134.0157
14. Norton D. Calculating the risk. Reflections on the norton scale (1989) Adv Skin Wound Care 2: 24-31.
15. Teot L and Dealey C. How to prevent bedsores: Media Flashes, 1997.
16. Jiang Q, Li X, Qu X, Liu Y, Zhang L, et al. The incidence, risk factors and characteristics of pressure ulcers in hospitalized patients in China (2014) Int J Clin Exp Pathol 7: 2587-2594.
17. Pancorbo-Hidalgo PL, Garcia-Fernandez FP, Lopez-Medina IM and Alvarez-Nieto C. Risk assessment scales for pressure ulcer prevention: a systematic review (2006) J Adv Nurs 54: 94-110. https://doi.org/10.1111/j.1365-2648.2006.03794.x
18. Waterlow J. Pressure sores: a risk assessment card (1984) Nursing times 81: 49-55.
19. Cherry GW. The European Pressure Ulcer Advisory Panel: a means of identifying and dealing with a major health problem with a European initiative (2006) Springer 183-187. https://doi.org/10.1007/1-84628-134-2_20
20. Lahmann N, Dassen T and Kottner J. Frequency of pressure ulcers in german hospitals (2012) Gesundheitswesen 74: 793-797. https://doi.org/10.1055/s-0031-1299778
21. Eberlein-Gonska M, Petzold T, Helaß G, Albrecht DM and Schmitt J. The incidence and determinants of decubitus ulcers in hospital care-an analysis of routine quality management data at a university hospital (2013) Dtsch Arztebl Int 110: 550-556. https://doi.org/10.3238/arztebl.2013.0550
22. Idowu OK, Yinusa W, Gbadegesin SA and Adebule GT. Risk factors for pressure ulceration in a resource constrained spinal injury service (2011) Spinal Cord 49: 633-647. https://doi.org/10.1038/sc.2010.175
23. Iyun AO, Malomo AO, Oluwatosin OM, Ademola SA and Shokunbi MT. Pattern of presentation of pressure ulcers in traumatic spinal cord injured patients in University College Hospital, Ibadan (2012) Int Wound J 9: 206-213. https://doi.org/10.1111/j.1742-481X.2011.00877.x
24. Manzano F, Navarro MJ, Roldán D, Moral MA, Leyva I, et al. Pressure ulcer incidence and risk factors in ventilated intensive care patients (2010) J Crit Care 25: 469-476. https://doi.org/10.1016/j.jcrc.2009.09.002
25. Yepes D, Molina F, León W and Perez E. Incidence and risk factors associated with the presence of pressure ulcers in critically ill patients (2009) Med Intensiva 33: 276-281. https://doi.org/10.1016/S0210-5691(09)72195-3
26. Sharp CA and McLaws ML. Estimating the risk of pressure ulcer development: is it truly evidence based? (2006) Int Wound J 3: 344-353. https://doi.org/10.1111/j.1742-481X.2006.00261.x
27. Doley J, Mallampalli A and Sandberg M. Nutrition management for the patient requiring prolonged mechanical ventilation (2011) Nutr Clin Pract 26: 232-241. https://doi.org/10.1177/0884533611405536
28. Nonnemacher M, Stausberg J, Bartoszek G, Lottko B, Neuhaeuseret M, et al. Predicting pressure ulcer risk: a multifactorial approach to assess risk factors in a large university hospital population (2008) J Clin Nurs 18: 99-107. https://doi.org/10.1111/j.1365-2702.2008.02425.x
29. Royal College of Nursing. Pressure ulcer risk assessment and prevention (2001) Royal College of Nursing, UK.
30. Bergstrom N, Braden B, Kemp M, Champagne M, Ruby E. Multi-site study of incidence of pressure ulcers and the relationship between risk level, demographic characteristics, diagnoses, and prescription of preventive interventions (1996) J Am Geriatr Soc 44: 22-30. https://doi.org/10.1111/j.1532-5415.1996.tb05633.x
31. Sving E, Idvall E, Högberg H and Gunningberg L. Factors contributing to evidence-based pressure ulcer prevention. A cross-sectional study (2014) Inter J Nurs Stud 51: 717-725. https://doi.org/10.1016/j.ijnurstu.2013.09.007
Citation: Ghali H, Chouket R, Ben Cheikh A, Khefacha S, Dhidah L, et al. Incidence and risk factors of pressure ulcers in a Tunisian university hospital (2019) Nursing and Health Care 4: 25-28
*Corresponding author: Hela Ghali, Department of Prevention and Security of Care, University Hospital Sahloul, Tunisia, Tel: +216 2430 1810, E-mail: yasminebenrejeb@hotmail.com
Keywords
Pressure ulcer, Incidence, Risk factors, Tunisia


 PDF
PDF