Introduction
One of the most important goals of root canal treatment is to eliminate bacteria from the infected root canal systems through instrumentation and use of disinfecting agents [1,2]. Biofilm can be defined as a sessile multicellular microbial community characterized by cells that are firmly attached to a surface and enmeshed in a self-produced matrix of Extracellular Polymeric Substance (EPS) [3,4]. Elimination from anatomical complexity of the root canal system is a challenging procedure. Thus; the use of a potent antibiofilm strategy has become mandatory to be able to resist endodontic infection [5,6].
Sodium hypochlorite in concentrations from 0.5%-6% is the most commonly recommended root canal irrigant. Aqueous solution of sodium hypochlorite is a dynamic balance of sodium hydroxide and hypochlorous acid, which on interaction with microorganisms and organic tissue causes chloramination, amino acid neutralization, and saponification reactions leading to strong antibacterial and tissue-dissolving effects [7]. Calcium hydroxide is the most commonly used intracanal medication during root canal procedures [8]. Its antibacterial property is generally related to the release of hydroxyl ions, which produces the lethal effects on bacterial cells including protein denaturation and damage to the bacterial cytoplasmic membranes and DNA [9]. However, the antimicrobial activity of sodium hypochlorite and calcium hydroxide can be inactivated by dentin, exudate from the periapical area, and microbial biomass [10]. In addition, both sodium hypochlorite and calcium hydroxide do not always eliminate E. faecalis biofilms from the root canal system.
Recently, Photodynamic Therapy (PDT) as an antibiofilm strategy is based on the use of a nontoxic dye (photosensitizer agent), that when activated by using a low energy light lead to production of free radicals such as singlet oxygen [11]. Singlet oxygen generated is highly reactive and is known to target various bacterial sites such as cell wall, nucleic acid, lipids membrane and proteins membrane, which promote bacterial cell death. Therefore, it has potentiality to enhance the disinfection efficacy of the conventional chemo-mechanical preparation [12]. Nanoparticles are insoluble particles that are ranging from 1 to 100 nm in size which combines biology principles with physical and chemical procedures to generate nano-sized particles with specific functions [13]. Different nanoparticles have been introduced as an irrigant or medicament to control the bacterial biofilm in root canal system. Pomegranates (Punica granatum L.) which have a long history of antibacterial use dating back to biblical times [14].
Egyptians used pomegranates to treat a number of different infections. It was utilized as a traditional remedy for thousands of years under the Ayurvedic system of medicine, with extracts from the rind of the fruit and bark of the tree being effective against diarrhea and dysentery. The Pomegranates in nanosized form have been applied in many health care fields because of their broad-spectrum bactericidal and virucidal properties [15]. So, the aim of this study was to evaluate the antibiofilm efficacy of nanoherbal medicament and photodynamic therapy using Confocal Laser Scanning Microscope.
Materials and Methods
A total of one hundred non-carious recently extracted human single rooted teeth with fully formed root apices were collected from patients with ages ranging between 20 and 45 years old. Also to confirm the presence of type I root canal morphology in each tooth according to Vertuccis classification. The teeth that had caries, deep cracks, attritions, fractures or restorations would be excluded from the study.
The dentin section blocks were prepared and standardized to be (4×4×1 mm) (Length × Width × thickness) according to Haapasalo and Orstavik D. technique through the following steps: Demarcations were done at Cementoenamel Junction (CEJ) and at the last 3 mm of the roots. The crowns of demarcated teeth were cut off at the level of CEJ by using a diamond disc mounted in a straight hand piece under water coolant [16]. Root canal patency for each sample was done using a size #10 and #15 K file. Instrumentation was done using Revo-S rotary NiTi file system according to the manufacturers instructions. Rotary files were mounted in a torque limited control motor at a torque 1.8 Ncm and speed 400 rpm as recommended by the manufacture starting from SC1 (25 taper 0.06) to AS40 (40 taper 0.06).
Irrigation was done using side vented needle gauge 30 mounted on 3 ml plastic syringe of 5.25% Sodium hypochlorite (NaOCl) between each instrumentation during the cleaning and shaping procedure. The apical 3 mm of the roots were sectioned off and the roots were longitudinally sectioned in bucco-lingual direction along the midsagittal plane into two semicylindrical halves and the cementum was removed from the root surface using Isomete saw at 1000 rpm under water cooling. Each samples of the roots were shaped and refined to be (4 × 4 × 1 mm) (Length × Width × thickness) respectively using Isomete saw at 1000 rpm under water coolant.
Sterilization of the samples
Each dentin section block was placed in 1.5 mL Eppendorf tube filled with Brain Heart Infusion (BHI) broth and sealed within sterilization pouches then underwent sterilization using autoclave at 12 °C for 20 minutes. After completion of sterilization, the Eppendorf tubes opened in a sterile air laminar flow cabinet and a sterile paper points were inserted into the Eppendorf tubes for 1 minute until it completely saturate and absorbed the broth media, then the paper point was transferred and spread on brain heart infusion agar plate The plate was incubated for 24 hours at 37°c and 100% humidity for bacterial count. After 24 hours the plate was inspected and checked for no inhibition zones around the incubated paper point indicating a sterility of the samples before inoculating the multispecies microorganisms.
Preparation of the microorganisms
Cultivation of Standard strains: E. faecalis American Type Culture Collection (ATCC 29212) and Staphylococcus epidermidis (Staph. epidermidis) American Type Culture Collection (ATCC 12228) were done and each Eppendorf tube contain sterile dentin section block was opened in a sterile air laminar flow cabinet. Eppendorf tubes were filled with 0.5 ml of E. faecalis suspension plus 0.5 ml of Staph epidermidis suspension using a sterile micropipette tips for each organism. Eppendorf tubes were closed tightly and shacked well then incubated for 21 days at 37°C in incubator.
Confirmation of multispecies biofilm formation
After incubation period, the samples were examined for confirmation of multispecies biofilm formation on dentin section blocks. Three randomly samples were selected after finishing of sterilization and before bacterial inoculation. Also, others three randomly samples were selected after 21 day after incubation of dentin section blocks with multispecies microorganisms. These samples were inspected to confirm the presence/absence of multispecies bacterial biofilm formation using Scanning Electron Microscope (SEM) as shown in (Figure 1a and Figure 1b).

Figure 1a: Image showing dentin section block immediately after sterilization.

Figure 1b: Image showing dentin section block after 3 weeks of biofilm inoculation.
Preparation of irrigant solutions and medicaments
Preparation of nano pomegranate extract: The herbal extracts are turned into nano herbals by nano emulsion O/W (oil on water) which was done using two-step procedure, Initially coarse emulsion was prepared by mixing the paraffin oil (1 ml)+(1 gram extract) and surfactant Tween 80 (2 ml) followed by addition of distilled water (7 ml) in the ratio 1:2:7 using magnetic stirrer at 600 rpm for 10 min at 25°c. Then, prepared coarse emulsion was further sonicated using sonicator with a maximum power output of 750 W under ice bath to avoid rising in temperature. The sonication process was carried out for 10 minutes.
Characterization of transformation to nanoherbals: Nanoemulsion was characterized by Transmission Electron Microscopy (TEM) at magnification 20000 and it showed nanoemulsion spherical in shape with average size of 50 nanometer (Figure 2).
Rose Bengal dye (Photosensitizer agent): Stock solution of Rose Bengal dye (Sigma-Aldrich, St Louis, MO. USA) was prepared by dissolving 20 mg RB powder in 20 ml deionized water to obtain a final concentration 10 μM/mL of RB dye after vortexed for 3 min at 37°C. The stock solution of Rose Bengal dye was stored at 4°C when not needed.

Characterization of Rose Bengal (RB) dye (Spectrophotometric analysis of RB dye solution): The absorption spectra of diluted solution of the prepared RB dye was recorded within 550 to 750 nm using double beam UV-Visible Spectrophotometer** and the maximum absorption peak was recorded at 650 nm (Figure 3).
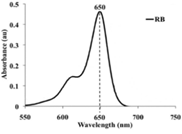
Figure 3: A diagram showing UV-absorption spectrum of RB showed maximum absorption peak at 650 nm.
Grouping of the samples
Group (1)-Medicated with nano punica granatum herbal extract medicament: Each sample was medicated with 1 ml of nano punica granatum herbal extract medicament within the Eppendorf tube till its fully covered and incubated in the incubator at 37°C for 7 days at humidity 100%.
Group (2)-Medicated with Calcium hydroxide medicament: Each sample was medicated with 1 ml of Calcium hydroxide medicament (Urbical promedica Germany) within the Eppendorf tube till it is fully covered and incubated in the incubator at 37°C for 7 days at humidity 100%.
Group (3)-Final rinse using Rose Bengal dye (RB) and activated with diode laser (Photodynamic therapy): Each sample was rinsed with 1 ml of Rose Bengal dye solution (10 μM) concentration within the Eppendorf tube and protected from ambient light then left for 15 minutes (prior to irradiation) for allowing the dye to interact with biofilm. Irradiation was performed using a diode laser (Lasotronix device) 635 nm for 5 minutes at energy fluence 30 J/cm2.
Group (4)-Final rinse using Sodium Hypochlorite (NaOCl): Each sample was rinsed with 1 ml of Sodium Hypochlorite (NaOCl) solution 5.25% concentration within the Eppendorf tube then left for 15 minutes.
Group (5)-Control group: There is no final rinse was used in this group. Each sample was left within the Eppendorf tube without any treatment (Negative control group) until time of evaluation.
Evaluation using confocal laser scanning microscopy
For the analysis of antimicrobial activity using confocal laser scanning microscopy, the samples were removed from the nanoherbal gel and irrigant solutions then washed for 1 min by deionized water to remove the remnants of the nanoherbal medicaments and irrigant solutions then stained by (Florocine Diacetate FDA and Propodium Iodid PI) to detect Live/Dead bacteria in the bacterial biofilm and left for 15 min then the samples were removed from the stains solutions, placed on slide, covered by cover slide and examined under confocal microscope then, the data were collected, tabulated, and then statistically analyzed.
Statistical analysis
The mean and standard deviation values were calculated for each group. Viable counts of antibacterial activity were transformed to their log10 values. Data were confirmed to be normally distributed. All Data were explored for normality using Kolmogorov-Smirnov and Shapiro-Wilk tests. Data showed parametric (normal) distribution. One way ANOVA followed by Tukey post-hoc test was used to compare between different groups for non-related samples in parametric data. Pair-wise sample t-test was used to compare between dependent samples. The significance level was set at P ≤ 0.05. Statistical analysis was performed with IBM® SPSS® Statistics Version 20 for Windows.
Results
With regard to the antimicrobial effectiveness of tested irrigant and medicaments, the highest mean percentage of dead bacteria was found in nano punica granatum (Group 1) (88.38 ± 5.45) followed by Rose Bengal dye (RB) (Photodynamic therapy) (Group 3) (75.73 ± 5.15) followed by sodium hypochlorite 5.25% (Group 4) (59.43 ± 8.14) followed by calcium hydroxide paste medicated (Group 2) (38.73 ± 5.15). The least mean percentage of dead bacteria was found in Control group (Group 5) (7.25 ± 1.12). With a statistically significant difference between groups where (p<0.001) as shown in Table 1 and Figure 4.


Analysis of multispecies biofilm images by CLSM: Figures 5-10 showing 3D reconstruction of multispecies bacterial biofilm representing live (green) and dead (red) bacteria for different groups.

Figure 5: Group (1) Nano punica granatum.
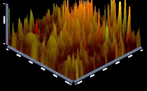
Figure 6: Group (2) Calcium hydroxide.
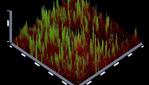
Figure 7: Group (3) Rose Bengal Dye (RB dye).

Figure 8: Group (4) Sodium hypochlorite (NaOCl).

Figure 9: Group (5) Control Group.
Discussion
Root canal disinfection is one of the main clinical challenges in endodontic therapy due to the physical limitations of irrigation within a closed system, anatomical root complexities and bacterial accumulation in the forms of bacterial biofilms [17,18]. Many methods have been utilized to efficiently disinfect the root canal system including different irrigant solutions with different activation method [19]. The level of biofilm maturation is an essential factor for biofilm resistance to antimicrobial agents [20]. For the biofilm development; the bacterial suspension was changed every 3 days to avoid the bacterial endotoxins accumulation that might have led to bacterial death. This allowed the bacterial multiplying without being dampened by their byproducts [21].
The most potent antibacterial compounds in pomegranate is ethanol, water, methanol, and acetone extracts of P. granatum that have shown strong antimicrobial properties against gram-positive and gram negative non-oral microorganisms also, contribute synergistically as mixtures to bring about the effects, including anthocyanins (pelargonidin-3-galactose and cyanidin-3-glucose) and flavonols (quercetin and myricetin) [22]. Although many intracanal medicaments were introduced to be used to disinfect root canal, calcium hydroxide remains the most wildly used as it exerts antibacterial effects in the root canal system as long as a high pH is maintained [23]. The Rose Bengal dye was selected as a photosensitizer agent for Photodynamic therapy due to its antibacterial activity against various gram positive and gram negative bacteria [24-26].
Also, RB dye is an anionic PS that has been shown excellent binding with cationic polymers such as chitosan nanoparticles. The concentration of anionic RB dye that used in the study was 10 μM. At this concentration, the singlet oxygen that produced was enough to induce significant biological activity against bacterial biofilm and this concentration also was founded to be below the cytotoxic limit based on the Shrestha and Kishen study [24]. Confocal Laser Scanning Microscope was used in this study because of its ability to determine the microbial biofilms at different levels of the infected root canal. Also, it provides detailed information about the presence, distribution, viability status, and depth of penetration of biofilm inside dentinal tubules in accurate 3D reconstructions images. The viability status of bacterial biofilm (live/dead bacteria) was determined throughout use of fluorescence-based live/dead staining technique. This technique were performed with two fluorescent dyes that was allowed two color discrimination of the population of living cells from the dead cells population (green/red colored). FDA and PI stains were used to stain live cells and dead cells, respectively [27].
FDA is a non-fluorescent permeable dye, when cross the cellular membrane of living cell convert into fluorescein dye (green in color) by interaction with intracellular Esterases (produced by metabolic activity of microorganisms). PI is a fluorescent impermeable dye that cannot cross the cellular membrane of living cells. It reaches the nucleus by passing throughout disordered areas of dead cell membranes and staining the nuclei (red in color) by intercalate with the DNA double helix of the cell. Thus, live bacterial cells are fluorescent green, whereas dead bacteria with damaged membranes are fluorescent red in color [28].
In the present study, the control group (group 5) showed the least mean percentage of dead bacteria when compared to other groups. The percentage of dead bacteria was found to be 7.25 % which is the normal for any untreated bacterial population. This result is in agreements with other researches done in this filed [29,30]. This can be explained by the fact of the absence of disinfecting agent in the control group that has lethal effect on the multispecies bacterial biofilm. With regard to pomegranate nano-herbal (group 1) it showed the highest significant absolute mean score of dead bacterial cells and these results were coincided with results reported by shoko T. et al. these findings may be attributed to that pomegranate extracts contains phenolic, anthocyanin, flavonols and organic acids. In accordance to our study the antimicrobial properties of P. granatum have been recently noticed [31,32].
The ethanol, water, methanol and acetone extracts of P. granatum have shown strong antimicrobial properties against gram-positive and gram negative nonoral microorganisms [33]. However A few studies have evaluated the antibacterial properties of this plant on oral bacteria [34]. The effect of water extract of P. granatum flower (petal) on oral microbial pathogens was investigated, which showed its greatest antimicrobial effect on Streptococcus sanguinis. Also in accordance to the present study, Gulube and Patel investigated pomegranate on biofilm formation, the crude extract of pomegranate killed cariogenic Streptococcus mutans at high concentrations [35]. At sub-bactericidal concentrations, it reduced biofilm formation.
Irshad R. et al. revealed that pomegranate nanoparticles are found to have high antibacterial activity along with biocompatibility [36]. This high antibacterial activity can be referred to its small size and large surface area. With regard to calcium hydroxide medicament (group 2) the multispecies biofilms on dentin section blocks were destroyed after 7 days of treatment, whereas several live bacteria still remained on the dentin sections. Possibly the high alkalinity of calcium hydroxide was neutralized by the dentin and biofilm matrix of multispecies biofilms [37,38] attributing to the reduced antibacterial effect of calcium hydroxide.
With regard to RB dye group (group 3), the result of dead bacteria could be attributed to the fact of RB dye is an anionic photosensitizer (negatively charged photosensitizer) that does not interact electrostatically with the negatively charged bacterial cell membranes, resulting in the membrane barriers of the bacterial cells limit the simple diffusion of RB dye into the bacteria cells. This leaded to reduced penetration of the Rose Bengal dye in to bacterial biofilm within dentinal tubules [39]. Also, this result is in agreements with other researches done in this field [40,41].
With regard to sodium hypochlorite group (group 4), the result of dead bacteria could be attributed to the fact of sodium hypochlorite in concentration 5.25% demonstrated antibiofilm effect against E. faecalis biofilm. The proportion of live bacteria in the 5.25% sodium hypochlorite group could be measured by viability staining and CLSM because sodium hypochlorite destroyed E. faecalis biofilm so quickly, leaving very little residual biofilm on dentin sections for analysis [42]. Time of tested materials application was done in accordance to clinical resemblance and it is expected to affect the results of the present study.
Conclusion
Nano punica granatum herbal extract medicament (gel/cream) and Rose Bengal dye irrigation (Photodynamic therapy) could be considered potent antibiofilm strategies for disinfection of the root canal system.
References
1. Rocas I and Siqueira J. Identification of bacteria enduring endodontic treatment procedures by a combined reverse transcriptase-polymerase chain reaction and re verse-capture checkerboard approach (2010) J Endod 36: 45-52. https://doi.org/10.1016/j.joen.2009.10.022
2. Torabinejad M, Handysides R, Khademi A and Bakland L. Clinical implications of the smear layer in endodontics: a review (2002) Oral Surg Oral Med Oral Pathol Oral Radiol Endod 94: 658-66. https://doi.org/10.1067/moe.2002.128962
3. Kayaoglu G and Ørstavik D. Virulence factors of Enterococcus faecalis: relationship to endodontic disease (2004) Crit Rev Oral Biol Med 15: 308-320. https://doi.org/10.1177/154411130401500506
4. Stewart P and Costerton J. Antibiotic resistance of bacteria in biofilms (2001) Lancet 358: 135-138. https://doi.org/10.1016/s0140-6736(01)05321-1
5. Waltimo T, Trope M, Haapasalo M and Ørstavik D. Clinical efficacy of treatment procedures in endodontic infection control and one year follow-up of periapical healing (2005) J Endod 31: 863-866. https://doi.org/10.1097/01.don.0000164856.27920.85
6. Peters O, Laib A, Gohring T and Barbakow F. Changes in root canal geometry after preparation assessed by high-resolution computed tomography (2001) J Endod 27: 1-6. https://doi.org/10.1097/00004770-200101000-00001
7. Estrela C, Estrela CR, Barbin EL, Spano JCE, Marchesan AM, et al. Mechanism of action of sodium hypochlorite (2002) Braz Dent J 13: 113-117. https://doi.org/10.1590/s0103-64402002000200007
8. Lee M, Winkler J, Hartwell G, Stewart J, Caine R, et al. Current trends in endodontic practice: emergency treatments and technological armamentarium (2009) J Endod 35: 35-39. https://doi.org/10.1016/j.joen.2008.10.007
9. Siqueira JF Jr and Lopes HP. Mechanisms of antimicrobial activity of calcium hydroxide: a critical review (1999) Int Endod J 32: 361-369. https://doi.org/10.1046/j.1365-2591.1999.00275.x
10. Haapasalo HK, Siren EK, Waltimo TM, Òrstavik D and Haapasalo MPP. Inactivation of local root canal medicaments by dentine: an in vitro study (2000) Int Endod J 33: 126-131. https://doi.org/10.1046/j.1365-2591.2000.00291.x
11. Hamblin M and Hasan T. Photodynamic therapy: a new antimicrobial approach to infectious disease (2004) Photochem Photobiol Sci 30: 436-450. https://doi.org/10.1039/b311900a
12. George S and Kishen A. Influence of photosensitizer solvent on the mechanisms of photoactivated killing of Enterococcus faecalis (2008) Photochem Photobiol 84: 734-740. https://doi.org/10.1111/j.1751-1097.2007.00244.x
13. Cushing B, Kolesnichenko V and OConnor C. Recent advances in the liquid-phase syntheses of inorganic nanoparticles (2004) Chem Rev 104: 3893-3946. https://doi.org/10.1021/cr030027b
14. Farmahan H. Pomegranate, in Recent Trends in Horticulturein the Himalayas (2004) India.
15. Negi GJ and Jena B. Antimicrobialactivities of pomegranate, in Pomegranates: Ancient Roots to Modern Medicine (2006) CRC Press, USA.
16. Haapasalo M and Ørstavik D. In vitro infection and disinfection of dentinal tubules (1987) J Dent Res 66: 1375-1379. https://doi.org/10.1177/00220345870660081801
17. Gregorio C, Arias A, Navarrete N, Rio V, Oltra E, et al. Effect of Apical Size and Taper on Volume of Irrigant Delivered at Working Length with Apical Negative Pressure at Different Root Curvatures (2013) J Endod 39: 119-124. https://doi.org/10.1016/j.joen.2012.10.008
18. Tay C, Quah S, Lui J, Yu V and Tan K. Matrix Metalloproteinase Inhibitor as an Antimicrobial Agent to Eradicate Enterococcus faecalis Biofilm (2015) J Endod 41: 1-6. https://doi.org/10.1016/j.joen.2015.01.032
19. Kishen A. Advanced therapeutic options for endodontic biofilms (2010) Endod Topics 22: 99-123. https://doi.org/10.1111/j.1601-1546.2012.00284.x
20. Stojicic S, Shen Y and Haapasalo M. Effect of the source of Biofilm Bacteria, Level of Biofilm Maturation, and Type of Disinfecting Agent on the susceptibility of Biofilm Bacteria to Antibacterial agents (2013) Endod 39: 473-477. https://doi.org/10.1016/j.joen.2012.11.024
21. Stoodley L and Stoodley P. Evolving concepts in biofilm infections (2009) Cell Microbiol 11: 1034-1043. https://doi.org/10.1111/j.1462-5822.2009.01323.x
22. Reddy M, Gupta S, Jacob M, Khan S and Ferreira D. Antioxidant, antimalarial and antimicrobial activities of tannin-rich fractions, ellagitannins and phenolic acids from Punica granatum L (2007) Planta Medica 73: 461-467. https://doi.org/10.1055/s-2007-967167
23. Cavallito CJ and Bailey J. Allicin, the Antibacterial Principle of Allium sativum: Isolation, Physical Properties and Antibacterial Action (1944) J Am Chem Society 66: 1950-1951. https://doi.org/10.1021/ja01239a048
24. George S, Hamblin M and Kishen A. Uptake pathways of anionic and cationic photosensitizers into bacteria (2009) Photochem Photobiol Sci 8: 788-795. https://doi.org/10.1039/b809624d
25. Dahl T, Midden W and Neckers D. Comparison of photodynamic action by Rose Bengal in gram-positive and gram-negative bacteria (1988) Photochem Photobiol 48: 607-612. https://doi.org/10.1111/j.1751-1097.1988.tb02870.x
26. Guo Y, Rogelj S and Zhang P. Rose Bengal-decorated silica nanoparticles as photosensitizers for inactivation of gram-positive bacteria (2010) Nanotechnol 21: 651-659. https://doi.org/10.1088/0957-4484/21/6/065102
27. Arite E, Marle J and Cate J. Conofocal microscopy study of undisturbed and chlorhexidine-treated dental biofilm (2001) J Dent Res 80: 1436-1440. https://doi.org/10.1177/00220345010800051001
28. Zapata R, Bramante C, Moraes I, Bernardineli N, Gasparoto T, et al. Confocal Laser Scanning Microscopy Is Appropriate to Detect Viability of Enterococcus faecalis in Infected Dentin (2008) J Endod 34: 1198-1201. https://doi.org/10.1016/j.joen.2008.07.001
29. Shen Y, Qian W, Chung C, Olsen I and Haapasalo M. Evaluation of the effect of two Chlorhexidine preparations on biofilm bacteria in vitro: a three-dimensional quantitative analysis (2009) J Endod 35: 981-985. https://doi.org/10.1016/j.joen.2009.04.030
30. Shen Y, Stojicic S, Qian W, Olsen I and Haapasalo M. The synergistic antimicrobial effect by mechanical agitation and two chlorhexidine preparations on biofilm bacteria (2010) J Endod 36: 100-104. https://doi.org/10.1016/j.joen.2009.09.018
31. Shoko T, Soichi T, Megumi M, Eri F, Jun K, et al. Isolation and identification of an antibacterial compounds from grape and its application to food (1999) Nippon Nogeikagaku Kaishi 73: 125-128.
32. Webster P, Webster S, Rich K and McDonald K. Ultrastructural preservation of biofilms formed by non-typeable Hemophilus influenza (2004) Biofilms 1: 165-182. https://doi.org/10.1017/s1479050504001425
33. Romero R, Schaudinn C, Kusanovic J, Gorur A, Gotsch F, et al. Detection of a microbial biofilm in intraamniotic infection (2008) Am J Obstet Gynecol 198: 135-135. https://dx.doi.org/10.1016%2Fj.ajog.2007.11.026
34. Diaspro A. Confocal and two -Photon Microscopy: Foundations, Applications, and Advances (2002) Wiley-Liss press, United States.
35. Gulube Z and Patel M. Effect of Punica granatum on the virulence factors of cariogenic bacteria Streptococcus mutans (2016) Microb Pathog 98: 45-49. https://doi.org/10.1016/j.micpath.2016.06.027
36. Irshad R, Tahir K, Li B, Ahmad A, Siddiqui A, et al. Antibacterial activity of biochemically capped iron oxide nanoparticles: A view towards green chemistry (2017) J Photochem Photobiol B 170: 241-246. https://doi.org/10.1016/j.jphotobiol.2017.04.020
37. Stuart CH, Schwartz SA, Beeson TJ and Owatz CB. Enterococcus faecalis: its role in root canal treatment failure and current concepts in retreatment (2006) J Endod 32: 93-98. https://doi.org/10.1016/j.joen.2005.10.049
38. Evans M, Davies JK, Sundqvist G and Figdor D. Mechanisms involved in the resistance of Enterococcus faecalis to calcium hydroxide (2002) Int Endod J 35: 221-228. https://doi.org/10.1046/j.1365-2591.2002.00504.x
39. Soukos N, Socransky S, Mulholland S, Lee S and Doukas AG. Photomechanical drug delivery into bacterial biofilms (2000) Pharm Res 17: 405-409.
40. Shen Y, Stojicic S, Qian W, Olsen I and Haapasalo M. The synergistic antimicrobial effect by mechanical agitation and two chlorhexidine preparations on biofilm bacteria (2010) J Endod 36: 100-104. https://doi.org/10.1016/j.joen.2009.09.018
41. Kishen A, Upadya M, Tegos G and Hamblin M. Efflux pump inhibitor potentiates antimicrobial photodynamic inactivation of Enterococcus faecalis biofilm (2010) Photochem Photobiol 86: 1343-1349. https://doi.org/10.1111/j.1751-1097.2010.00792.x
42. Stojicic S, Shen Y, Qian W, Johnson B and Haapasalo M. Antibacterial and smear layer removal ability of a novel irrigant, QMiX (2012) Int Endod J 45: 363-371. https://doi.org/10.1111/j.1365-2591.2011.01985.x
*Corresponding author
Mohamed Ahmed Wakwak, Lecturer of Operative Dentistry, Faculty of Dental Medicine, Al-Azhar University, Makram Ebid, Nasr City, Egypt, E-mail: drwakwak2006@azhar.edu.eg
Citation
Farghaly MA and Wakwak AM. Confocal laser scanning electron microscope assessment of different antibiofilm strategies of root canal system disinfection (an experimental study) (2019) Dental Res Manag 3: 68-73.
Keywords


 PDF
PDF