Introduction
High Sympathetic (S) tone and CAN; defined as critically low resting Parasympathetic [P] tone, P<0.10 beats per minutes 2 [bpm2] have been associated with ACS, CHF, malignant ventricular arrhythmias, and increased mortality [1-8]. Good P-tone is cardio-protective [9]. Despite knowing this, we dont use (P&S) measures routinely, if at all, to help risk stratify and medically manage our patients. Historically, the reason for this may be due to the difficulty generalizing non-invasive autonomic measures to typical clinical populations. Heretofore, non-invasive measures of autonomic activity, including those based on beat-to-beat cardiac activity, are all measures of only total autonomic activity, forcing assumption and approximation to theorize P&S activity. However, weve found the user-friendly accurate, relatively inexpensive, easily mastered ANX 3.0 P&S monitor (formerly ANSAR Medical Technologies, Inc., now TMCAMS, Inc., Atlanta, GA, USA) assessment quite valuable. The P&S Monitor is based on technology developed, validated and verified by the first joint Bio-Medical Engineering program group from MIT (Massachusetts Institute Technology) and Harvard [10-14]. This article briefly reviews some of the studies we have completed as well as mentioning ongoing and future trials.
High S-tone and low P-tone at rest is relative and assessed
by (SB: SB=[resting S]/[resting P; normal is 0.4
Symptoms and P&S responses to stimuli help to differentiate catecholaminergic from angiotensin effects, for example. High SB may be medication induced such as with excessive utilization of inhaled beta-2 agonists for pulmonary disorders [16-18]. Beta-adrenergic and alpha-adrenergic blockers, angiotensin-blockers [ACE-Is (Angiotensin Conversion Enzyme Inhibitor) and ARB (Angiotensin Receptor Blocker)] are known to reduce resting S-tone. Implanted cardiac devices and cardiac rhythm therapy also effect the sympathetics by forcing them to entrain to the therapy. Low SB, indicating (resting) Parasympathetic Excess (PE) is associated with depression, syncope, excess-gut motility, and is relieved with very low-dose anti-cholinergics. P-tone is a measure of the net, cumulative result of both nicotinic and muscarinic receptors. When treating the symptoms of these disorders, titrating to normalize SB is a goal.
There are also proper dynamic or challenge P&S balances. Challenge imbalances may lead to resting imbalances and complicating or confounding resting imbalances. For example, upon assuming a head-up posture (e.g., standing) the proper dynamic balance is a slight decrease in P-tone quickly followed by a modest increase S-tone. This defeats the effect of gravity causing a shift in blood to the lower extremities and vasoconstricts the lower vasculature to support standing. A decrease in S-tone at this time (Sympathetic Withdrawal-SW) is associated with orthostatic dysfunction and may cause secondary, high, resting BP as a compensatory response to the decrease in BP associated with orthostatic dysfunction. Typically, the high resting BP (Blood Pressure) is considered the primary and treated as such, yet the patients become more lightheaded and then become non-compliant.
This is because the medication induced lower resting pressure, which results in poor diastolic coronary and brain perfusion caused by the decline in standing BP, and the patients body defeats the therapy to maintain proper perfusion. Low prolonged coronary diastolic pressure (and associated perfusion) due to SW with prolonged, high systolic pressure, as measured by high resting BP, may lead to heart failure. P&S monitoring helps to document these complications and guide therapy. Another possible imbalance from the stand example is a challenge PE [15]. PE may also lead to secondary SE and confound the treatment of high BP, for example. Challenge PE is associated with difficult to control BP, blood glucose, various hormone levels, increased weight, difficult to describe pain syndromes (including chronic refractory pain syndrome (CRPS)), unexplained arrhythmia (palpitations), seizures, temperature dysregulation (both response to heat or cold and sweat responses), and symptoms of depression or anxiety, fatigue, exercise intolerance, sex dysfunction, sleep or GI disturbance, lightheadedness, cognitive dysfunction or Brain Fog or frequent headache or migraine [19]. Challenge PE may also be treated with very low-dose anti-cholinergics, or if heart disease, high BP, or some other form of SE, PE may be treated with the double cocktail: carvedilol whose central alpha effect lowers PE. A better understanding of P&S pathophysiology provides more information to reduce morbidity and mortality risk and improve patient outcomes [15].
Methods
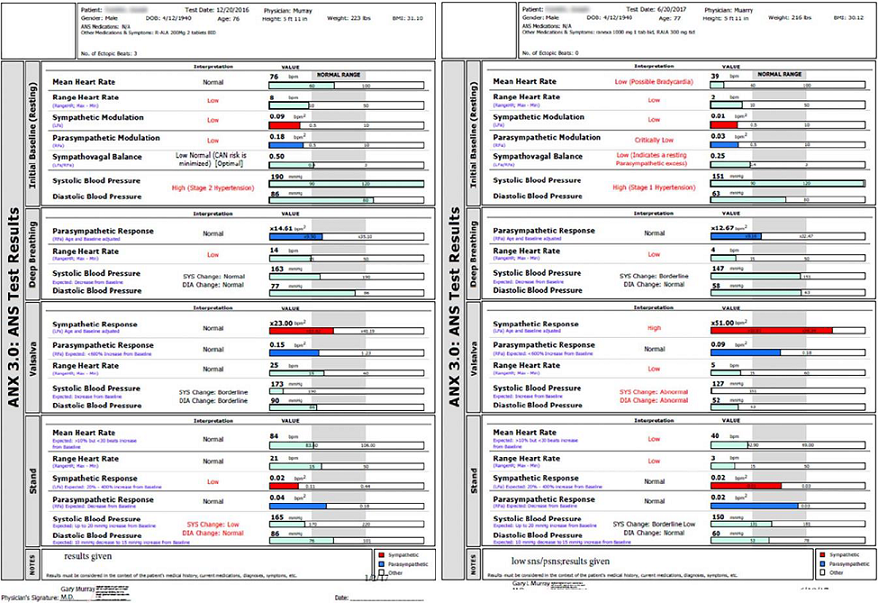
The ANX 3.0 P&S function monitor (hereafter designated P&S Monitor) computes simultaneous, independent measures of P&S activity based on continuous time-frequency analyses of Heart Rate Variability (HRV) with concurrent time-frequency analyses of Respiratory Activity (RA). The following variables were recorded (although not all are detailed in the results section): 5 min. seated resting BP and P&S activity (measured as Respiratory Frequency area [RFa] and Low-Frequency area [LFa] respectively); Exhalation/Inhalation (E/I) ratio and RFa were computed in response to 1 min. of deep breathing (paced at 6 breaths/min); Valsalva ratio and LFa & RFa were computed in response to a short series of Valsalva maneuvers (10 to 15 sec. each); and HR, BP, LFa, RFa and 30:15 ratio were computed in response to 5 min. of head-up postural change (quick stand followed by quiet 5 min. standing) [10-14].
Sympathovagal Balance is computed as LFa/RFa (reported means are averages of ratios, not ratio of averages). P-activity (RFa) was defined as the spectral power within a 0.12 Hz-wide window centered on the Fundamental Respiratory Frequency (FRF) in the HRV spectrum. FRF was identified as the modal peak from the time-frequency analysis of RA. Effectively, FRF is a measure of vagal outflow as it affects the heart, as in Respiratory Sinus Arrhythmia (RSA). S-activity (LFa) was defined as the remaining spectral power, after computation of RFa in the low-frequency window (0.04-0.15 Hz of the HRV spectrum) [10-14]. The 30:15 ratio is the ratio of the 30th R-R interval after a quick head-up postural change (standing) to the 15th R-R interval after standing. The 30:15 ratio reflects the reflex bradycardia after standing that is dependent on sympathetic vasoconstriction. The Valsalva ratio is the ratio of the longest R-R interval to the shortest R-R interval during a 15 sec. Valsalva maneuver. The E/I ratio is the ratio of the heart beat interval during peak exhalation over that during peak inhalation during paced breathing. The E/I ratio is a threshold measure of more or less Vagal (P) tone, as are the 30:15 and Valsalva ratios.
In the first study, statistics, including means, standard deviations, and student t-tests, were performed under SPSS v 14.1. Student t-tests were performed as 2-tailed with equal variance. Significance values were determined on the null hypothesis that the pre- and post-treatment P&S values were equal. In the second study, continuous data were assessed for normality with normally distributed data and analyzed using Student t-tests. Non-normally distributed data were assessed using a Mann-Whitney test. Dichotomous data were analyzed using the Chi-square test or Fishers-Exact Test. We determined that 50 patients per group were needed to have a sufficient sample size using an alpha of 0.05, difference of means of 6 units and expected standard of deviation of 15 units with a power of 80%. All statistics were performed under SPSS v 1.4. Student t-tests were performed as two-tailed with equal variance. Significance values were determined on the null hypothesis that pre- and post- treatment values were equal. In our third study, Receiver Operating Characteristic (ROC) analysis was determined. SB>2.5 and (Left Ventricular Ejection Fraction) LVEF<0.34 best predicted major cardiac events (MACE: acute coronary syndromes, acute CHF, malignant ventricular arrhythmias, cardiovascular death). The p-value of a SB>2.5 vs. LVEF<0.34 or reversible defect(s) on Myocardial Perfusion Imaging (MPI) was computed by uncorrected chi-square test. In our fourth study, continuous data were assessed for normality with normally distributed data using Student t-tests and non-normally distributed data using a Mann-Whitney U test. Dichotomous data were analyzed using the chi-square test or Fishers exact test. A p-value of 0.05 or less was considered significant. Student t-tests as two-tailed with equal variance. Significance values were determined on the null-hypothesis that the pre- and post-treatment values are equal. All patients signed informed consents.
Results
Congestive Heart
Failure
In
CHF, S is increased due to enhanced stimulatory input, increased adrenal
catecholamine output, as well as reduction of restraining influences, including
reduced vagal input, although beta-1 Adrenergic Receptors (AR) are down
regulated due to chronic stimulation. Beta- 2 AR, muscarinic and nicotinic
receptor function remains intact. Patients responding to Cardiac Resynchronization
Therapy (CRT) demonstrate improved P&S function, whereas non-responders
do not [20-23].
In our first study, 54 ACC/AHA (American College Cardiology/American Heart Association) guideline-treated chronic CHF patients [54% HFrEF (Heart Failure Reduced Ejection Fraction), 46% HFpEF (Heart Failure Preserved Ejection Fraction)] were randomized to adding Ranolazine (RANCHF-Ranolazine-Treated Heart Failure) vs. continued usual care (NORANCHF-No Ranolazine- Treated Heart Failure) [24]. Demographics between these groups matched well; the mean beta blocker dose was higher in the NORANCHF cohort. 59% of the patients in each group initially had high SB, CAN or both. At 1 year, 94% of RANCHF patients improved P&S measures; 88% normalized high SB and corrected CAN. Only 50% of NORANCHF patients improved (p=0.056).
Individually, only 18% of NORANCHF patients normalized high SB vs. 83% of RANCHF (p=0.013). Four NORANCHF patients (15%) demonstrated SB responses that became abnormally high. At 1 year, resting P-activity was 0.50 bpm2 in RANCHF patients vs. 0.38 bpm2 in NORANCHF (p=0.004). Improvement of P&S measures in RANCHF patients were independent of Brain Natriuretic Peptide (BNP) and impedance cardiogram results, suggesting a direct effect of RAN on P&S function. This was confirmed by similar improvements in P&S measures in a 30 patient control group without known cardiac disease that had initial CHF-like profiles.
In our second study, 109 ACC/AHA guideline-treated (New York Heart Association) NYHA class 2-4 chronic CHF patients (84 HFrEF, 25 HFpEF; 54 RANCHF, mean follow-up 24.5 mo.; 55 NORANCHF, mean follow-up 22.8 mo., were matched for age, gender, and history [25]. 98% of patients took a beta blocker (slightly higher dose in NORANCHF); HFpEF RANCHF patients had more patients with HTN and chronic renal insufficiency. 70% of RANCHF patients increased LVEF an average 11.3 units (p=0.018 for HFrEF RANCHF, initial mean LVEF 0.30); LVEF in NORANCHF patients decreased 1 unit from initially 0.30. RAN MACE (cardiac death, acute CHF, VT/VF) occurred in 31.5% vs. 38.2% MACE in NORANCHF (an 18% reduction). Again, RAN improved P&S measures. SB decreased in RANCHF (p=0.019) while increasing in NORANCHF patients (p=0.039). In the total population, final SB was 3.5 in MACE patients vs. 2.28 in patients without. This led us to do our third study. In our third study (unpublished data) we followed 483 patients for a mean of 4.92 yr. (127 with CAD risk factors, 224 with CAD, 132 with chronic CHF). We compared SB>2.5 to reversible myocardial imaging defect(s) or LVEF<0.34 as a predictor of MACE (ACS, acute CHF, VT/VF, cardiac death). SB independently outperformed them (p=0.001) with a sensitivity of 0.59, OR=7.03 (CI: 4.59-10.78), specificity of 0.83, PPV=0.64, and NPV=0.80. 31% of patients had a SB>2.5. There were 3 patterns of high SB (P&S measures taken every 6 mo.): acute, chronic, and intermittent. An acutely high SB (20%) is the most ominous.
In our fourth study, in a cohort of 109 patients with low standing S-response (known as Sympathetic Withdrawal as opposed to the normal increase in S-activity with stand), 29 were found with Neurogenic Orthostatic Hypotension (NOH, fall in Standing BP (sBP) of at least 20/10 mmHg), and 60 with Neurogenic Orthostatic Intolerance (NOI, fall in sBP of -6 to -19 mmHg) [26]. Both groups were given (r) alpha lipoic acid, at a mean dose range of 993-1500 mg/d. A third, control, group included 20 patients with either NOH or NOI. All patients were followed for a mean of 2.28 yr. 66% of NOH patients responded (standing change in BP ranged from -28/-10 mmHg to 0/+2 mmHg [p=0.0129 for systolic, p=0.0456 for diastolic pressure changes]). 67% of NOI patents responded as well (standing change in BP ranged from -9/+1 mmHg to +6/+2 mmHg; [p ≤ 0.001 for systolic, ns for diastolic, pressure changes]). The control group had no changes in BP. If maintaining a diastolic BP at least 60 mmHg to preserve coronary perfusion were taken into account, 88% of patients would be responders. Although all patients treated with (r)ALA increased their S-response to stand, responsiveness depended upon the resting S-tone: those with the lowest resting S-tone (indicating advanced autonomic dysfunction) responded the least.
Sympathovagal Balance
High
S-activity contributes to MACE through hemodynamic stress, coronary
vasoconstriction, cardiac electrical instability, endothelial dysfunction, and
LDL (Low Density
Lipoprotein) cholesterol oxidation. Alternatively, MACE acutely increases
S-activity and responsiveness. Therefore identifying high SB should help
predict the risk of developing MACE as well as diagnosing its presence.
Logically, normalizing SB will help to prevent MACE and reduce its mortality
and morbidity.
SB>2.5 increases the odds of suffering MACE seven-fold. Dutifully prescribing ACC/AHA and (Joint National Committee) JNC 8 guidelines for prescribing beta blockers for chronic HFrEF, CAD, and hypertension is insufficient to insure optimal SB, which likely plays a significant role in the continued disturbing rates of MACE in our patients. In fact, death rate per 100,000 treated hypertensives is increasing. Now we may and should measure SB, and adjust pharmacologic therapy accordingly. Ranolazine reduces SB and MACE in chronic CHF, likely by its effect on cardiac sodium channel 1.5 and P&S sodium channel 1.7 [24,25]. We were the first to report Ranolazine reduces ACS in CAD [27].
Sympathetic
Withdrawal and Orthostatic Dysfunction
Orthostatic
hypotension occurs in 10-30% of the elderly, associated with significantly
increased mortality and morbidity. Resting P&S activity falls with aging.
Chronic disease accelerates the aging effect. The P-nervous system (comprised
primarily of the Vagus Nerve outside the brain) is more exposed, and therefore,
more susceptible to insult, including increased oxidative stress that occurs
with age. As a result, the P&S nervous systems become uncoupled, with
P-activity declining faster than S-activity. This imbalance leads to autonomic
dysfunction and ultimately autonomic neuropathy. A first sign of autonomic
dysfunction is orthostatic dysfunction, including SW which typically precedes
any decline in BP upon standing. SW (as in NOH) is a leading cause of orthostatic dysfunction.
We typically, pharmacologically, treat symptomatically with Midodrine,
Fluodrocortisone, Desmopressin, or occasionally with expensive Droxydopa
(Northera) or other drugs. (R)ALA, an over-the-counter powerful antioxidant
supplement (ALA is produced in the body and production declines with age),
treated the cause of NOH and NOI successfully in 66% of patients by increasing
S-responses with stand (relieving SW). Hopefully, treating the cause will slow
this diseases progression and reduce mortality and morbidity, as well as
treatment complications such as supine/sitting high BP and fluid overload.
P&S activity should be measured in all orthostatic patients without venous
stasis or medication-related orthostatic dysfunction. A concern of orthostatic
dysfunction is the possibility of low coronary perfusion. If coronary diastolic
BP is below 60 mmHg then the heart is hypo-perfused, as is the brain. As a
result, an adrenaline storm is released and systolic pressure is increased.
This increases cardiac demand and cardiac stress.
A resulting increase in systolic pressure over 130 mmHg (resting) may produce pulse pressures (>70 mmHg) that are associated with poor prognoses. Prolonged, this condition may precipitate, and certainly may exacerbate, heart failure. In this way SW may be associated with heart failure. Without a means of recognizing SW, therapy would typically be directed to reducing systolic pressure and thereby pulse pressure. However, this may exacerbate the orthostatic dysfunction and exacerbate coronary hypo-perfusion, as well as lead to non-compliance, or unstable or difficult to manage BP as the body attempts to maintain coronary and brain perfusion. Similarly, if the orthostatic drop in BP is treated as the primary, as with vasopressors, it may further increase systolic pressure, increasing pulse pressure, thereby exacerbating heart failure as well.
Cardiovascular
Autonomic Neuropathy
CAN is
defined as very weak, resting P-activity; regardless of resting S-activity. CAN
is a normal part of the aging process. It simply means that the typical elderly
person has a higher morbidity and especially mortality risk than the typical
younger person. However, this is not to dismiss CAN. As soon as CAN is
demonstrated, a full cardiac work-up is recommended, if for no other reason
than to establish a baseline. The other reason not to dismiss CAN is that it
should always be risk stratified. CAN is risk stratified by SB. Given that a
little more (resting) P-activity is cardio-protective, low-normal SB
(0.4
· If CAN with high SB is demonstrated, consider sympatholytics based on history, titrated against normalizing SB and thereby normalize mortality risk. Choice of sympatholytic treatment is based on patient history. For example, if BP is high then consider anti-hypertensives, or if BP is normal to low or HR is high, then consider beta-blockers. As with diabetics, Carvedilol is often the preferred beta-blocker for CAN with high SB.
· If CAN with low SB is demonstrated, consider low-dose anti-cholinergics (very low-dose anti-depressants-low-dose to minimize morbidity risks), depending on other medical history, titrate to normalize SB and thereby normalize mortality risk. Choice of anti-cholinergic is based on patient history. For example if BP is high then consider very low-dose SSRI, or if BP is normal to low then consider very low-dose SSRI or tri-cyclic.
· If CAN is present with normal resting SB with a recent cardiac work-up, then mortality risk is normal, and the resting autonomic state of the patient is well managed. Any (resting) abnormality may be due to end-organ dysfunction. If there has not been a recent cardiac work-up, then one is recommended.
For chronic patients, CAN has been found to carry the same 50% increase in the five-year mortality rate as in diabetics [29,30]. More recently, some data suggests that CAN represent a 50% increase in the two-year mortality rate. In addition to geriatric patients, CAN may be normal for post-MI, post-CABG, and CHF patients, as well as other chronic diseases. CAN is associated with other risk factors including:
· Low ejection fraction
· Poor cardiac output
· Arrhythmias
· Cardiomyopathies including chronic heart failure
· Poor circulation, coronary artery disease with or without angina
· Greater mortality and
· Greater morbidity including silent myocardial infarction and early cardiac death.
Often, very low P-activity (CAN) leads to the need for an implanted cardiac device [1,31-44].
Discussion
P&S Monitoring was chosen for two reasons. First, P&S Monitoring includes spectral analyses based on the time-frequency analysis technique of Continuous Wavelet Transforms (CWT), rather than the frequency-only analysis technique of the Fast Fourier Transforms (FFT). Although including short-term FFT is accurate for stationary signals, it results in a compromise in time and frequency resolution because fixed length windows are analyzed. Therefore, the FFT (including the short-term FFT) involves are two weak assumptions: the P&S signals are not stationary (even at rest or during quiet standing) and the time-frequency compromise is not static in addition to the fact that it is a compromise. The P&S-tone values from P&S Monitoring are computed from nonstationary, continuous, independent RA and HRV signals. CWT permits automatic adjustment of the window length to the features of the signal. As a result, time-frequency resolution is superior to all prior HRV studies [11-14]. Second, P&S Monitoring is the only non-invasive technique that (mathematically) independently and simultaneously quantifies P-activity without assumption and approximation. Other autonomic measures based on beat-to-beat cardiac activity (e.g., HRV, beat-to-beat BP and Pulse Wave Velocity measures) assume that P-activity is always located within the 0.15-0.40 Hz frequency range (a wide window to improve the capture of P-activity). Instead, P&S Monitoring measures a second, independent measure of P&S activity: RA using impedance plethysmography. The first measure is of the heart (HRV); the second measure is of the lungs (RA).
While it is true that RSA is generated from RA (via pulmonary baroreceptors and the Vagus Nerve), measuring RA is not a direct measure of P-activity. RA is a second independent measure of the autonomic nervous system and therefore fully satisfies the algebraic requirement necessary to fully characterize a system with two independent components. With these two independent measures as verified and validated by the MIT/Harvard team, P&S Monitoring localizes and quantifies P-activity, and thereby S-activity, over the period of observation without the need for assumption and approximation. Conceptually, the P&S Monitoring process, in effect, measures RSA even when it is not possible to visualize it from the cardiogram. Given that RSA is purely parasympathetic in etiology, conceptualizing the measurement of RA as a measure of RSA helps to understand the process that provides a direct measure of P-activity. The process is based on the measure of the FRF [11-14]. For example, if the patients respiratory rate (FRF) is slow, P could be contained within the low frequency range (0.04-0.15 Hz), e.g., S-range of HRV. The low frequency range represents S-activity as modulated by P-activity [10,11]. Slow respiration leads to higher low frequency HRV activity misinterpreted as increased S-response unless FRF is determined. For the first time, simultaneous time-frequency analysis of HRV and RA accurately identifies P, unscrambling S & P activity. This technological breakthrough allowed us to correctly measure SB and CAN.
Conclusions
P&S abnormalities, including high SB, CAN, and low S-responses to standing (head-up postural change) are common. They cause and contribute to the mortality, morbidity, and cost of medical care, including for CAD, chronic CHF and NOH. Despite our ability to easily diagnose and address these P&S abnormalities, we seldom, if ever, do. Our patients deserve better.
Future Trials
Hypertension
We
have begun to investigate P&S abnormalities contributing to hypertension,
considering the question When is high blood pressure a symptom and better
treated as secondary? By 2021, worldwide, 1.5 billion or 1/3 of the worlds
population will be hypertensive. Currently, only 35% of patients are
clinic-controlled, 30% have masked-uncontrolled high BP (MUCH) Masked Uncontrolled
Hypertension, and after 1.5 yrs. of treatment, 25% return to uncontrolled
status. Often, we find high BP to be compensatory to decreases in BP upon
head-up posture, such as with SW in NOH or NOI. Apparently this is to help
maintain coronary and brain perfusion. This form of hypertension is often
relieved organically once SW and thereby orthostatic dysfunction is relieved
[45]. Another P&S finding associated with high BP that does not seem to
respond to standard therapy is associated with high S-activity secondary to
high P-activity. Typical therapy seems to exacerbate the high P-activity,
thereby forcing higher S-activity (since P-activity establishes the threshold
around which S-responds). As a result, BP becomes more labile or the patient
seems unresponsive.
It may be that treating the abnormal P-excess and normalizing P-activity, may organically normalize the S-response and thereby normalize BP. We plan to compare P&S-assisted therapy to JNC 8 (Figure 1).
Coronary Disease
We
plan a multicenter, randomized, prospective study: Management of Outpatients
using Sympathovagal Balance Trial (MOST) to compare P&S-guided therapy to
usual care (Figures 2,3). Our hypothesis generating findings include the
observation that SB>2.5 seems to be a better predictor of MACE (ACS, acute
CHF, VT/VF, cardiac death) when compared with reversible myocardial imaging defect(s)
or LVEF<0.34 in the same patients.
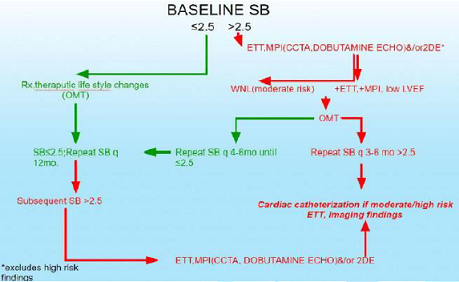
Figure 2: Stable PTS with risk factors for CAD.
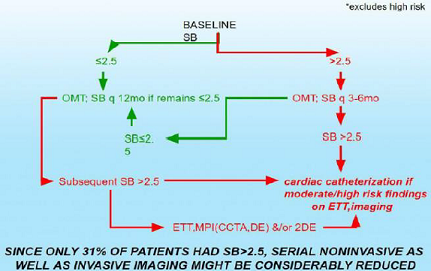
Figure 3: Stable CAD, CHF PTS Baseline ETT, MPI (CCTA, DE) &/or 2DE*.
References
1. Vinik A and Ziegler D. Diabetic cardiovascular autonomic neuropathy (2007) Circulatn 115: 387-397. https://doi.org/10.1161/circulationaha.106.634949
2. Tomaselli G and Zipes D. What causes sudden death in heart failure? (2004) Circ Res 95: 754-763. https://doi.org/10.1161/01.res.0000145047.14691.db
3. Maser R, Mitchell B, Vinik A and Freeman R. The association between cardiovascular autonomic neuropathy and mortality in individuals with diabetes: a meta-analysis (2003) Diabetes Care 26: 1895-1901. https://doi.org/10.2337/diacare.26.6.1895
4. Watanabe J, Shinozaki T, Shiba N, Fukahori k, Koseki Y, et al. Accumulation of risk markers predicts the incidence of sudden death in patients with chronic heart failure (2006) Eur J Heart Fail 8: 237-242. https://doi.org/10.1016/j.ejheart.2005.08.003
5. Curtis B and OKeefe J. Autonomic tone as a cardiovascular risk factor: the dangers of chronic fight or flight (2002) Mayo Clinic Proc 72: 45-54. https://doi.org/10.4065/77.1.45
6. McCance A, Thompson P and Forfar J. Increased cardiac sympathetic nervous activity in patients with unstable coronary heart disease (1993) Eur Heart J 14: 751-757. https://doi.org/10.1093/eurheartj/14.6.751
7. Manfrini O, Morgagni C, Pizzi C, Fontana F and Bugiardini R. Changes in autonomic nervous system activity: spontaneous versus balloon-induced myocardial ischemia (2004) Eur Heart J 25: 1502-1508. https://doi.org/10.1016/j.ehj.2004.03.019
8. Akuttsu Y, Kaneko K, Kodama Y, Suyama J, Shinozuka A, et al. Significance of cardiac sympathetic nervous system abnormality for predicting vascular events in patients with idiopathic paroxysmal atrial fibrillation (2010) Eur J Nucl Med Mol Imaging 37: 742-749. https://doi.org/10.1007/s00259-009-1322-7
9. Abe M, Iwaoka M, Nakamura T, Kitta Y, Takano H, et al. Association of high levels of plasma free dopamine with future coronary events in patients with coronary artery disease (2007) Circ J 71: 688-692. https://doi.org/10.1253/circj.71.688
10. Aysin B, Colombo J and Aysin E. Comparison of HRV analysis methods during orthostatic challenge: HRV with respiration our without? (2007) 29th Int Conf IEEE EMBS Lyon, France. https://doi.org/10.1109/iembs.2007.4353474
11. Akselrod S, Gordon D, Ubel FA, Shannon DC, Berger AC, et al. Power spectrum analysis of heart rate fluctuation: a quantitative probe of beat-to-beat cardiovascular control (1981) Sci 213: 220-222. https://doi.org/10.1126/science.6166045
12. Akselrod S, Gordon D, Madwed J, Snidman N, Shannon D, et al. Hemodynamic regulation : Investigation by spectral analysis (1985) Am J Physiol 249: H867-H875. https://doi.org/10.1152/ajpheart.1985.249.4.h867
13. Akselrod S, Eliash S, Oz O and Cohen S. Hemodynamic regulation in SHR: investigation by spectral analysis (1987) Am J Physiol 253: H176-H183. https://doi.org/10.1152/ajpheart.1987.253.1.h176
14. Akselrod S. Spectral analysis of fluctuations in cardiovascular parameters: a quantitative tool for the investigation of autonomic controls (1988) Trends Pharmacol Sci 9: 6-9. https://doi.org/10.1016/0165-6147(88)90230-1
15. Colombo J, Arora RR, DePace NL and Vinik AI. Clinical Autonomic Dysfunction: Measurement, Indications, Therapies, and Outcomes (2014) Springer Science, New York, USA.
16. Salpeter SR. Cardiovascular safety of β2-adrenoceptor agonist use in patients with obstructive airway disease: a systematic review (2004) Drugs Aging 21: 405-414. https://doi.org/10.2165/00002512-200421060-00005
17. Cazzola M, Matera MG and Donner CF. Inhaled β2-Adrenoceptor Agonists: Cardiovascular Safety in Patients with Obstructive Lung Disease (2005) Drugs 65: 1595-1610. https://doi.org/10.2165/00003495-200565120-00001
18. Salpeter, Shelley R. et al. Cardiovascular Effects of β-Agonists in Patients with Asthma and COPD: A Meta-Analysis (2004) CHEST 125: 2309-2321. https://doi.org/10.1378/chest.125.6.2309
19. Tobias H, Vinitsky A, Bulgarelli RJ, Ghosh-Dastidar S and Colombo J. Autonomic nervous system monitoring of patients with excess parasympathetic responses to sympathetic challenges-clinical observations (2010) US Neurology 5: 62-66. https://doi.org/10.17925/usn.2010.05.02.62
20. Triposkiadis F, Karavannis G, Skoularigis J, Louridas G and Butler J. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications (2009) J Am Coll Cardiol 54: 1747-1762.
21. Bibevski S and Dunlap M. Evidence for impaired vagus nerve activity in heart failure (2011) Heart Fail Rev 126: 129-133. https://doi.org/10.1007/s10741-010-9190-6
22. Martignani C, Diemberger J, Nanni C, Biffi M, Ziacchi M, et al. Cardiac resynchronization therapy and cardiac sympathetic function (2015) Eur J Clin Invest 45: 792-799. https://doi.org/10.1111/eci.12471
23. Del Mazumder D, Kass D, ORourke B and Tomaselli G. Cardiac resynchronization therapy restores sympathovagal balance in failing heart by differential remodeling of cholinergic signaling (2015) Circ Res 116: 1691-1699. https://doi.org/10.1161/circresaha.116.305268
24. Murray G and Colombo J. Ranolazine improve autonomic balance in heart failure when added to guideline-driven therapy (2014) Heart International 9: 1-7. https://doi.org/10.5301/heartint.5000215
25. Murray G and Colombo J. Ranolazine preserves and improves left ventricular ejection fraction and autonomic measures when added to guideline-driven therapy in chronic heart failure (2014) Heart International 9: 66-73. https://doi.org/10.5301/heartint.5000219
26. Murray G and Colombo J. (r) Alpha Lipoic Acid is a safe, effective pharmacologic therapy of chronic orthostatic hypotension associated with low sympathetic tone (2019) Thieme Medical Publishers 333 Seventh Avenue, New York, USA. https://doi.org/10.1055/s-0038-1676957
27. Murray G and Colombo J. Ranolazine therapy reduces non-ST-segment elevation myocardial infarction and unstable angina in coronary disease patients with angina (2016) Int J Angiology 25: 159-164. https://doi.org/10.1055/s-0036-1572364
28. Umetani K, Singer DH, McCraty R and Atkinson M. Twenty-four hour time domain heart rate variability and heart rate: relations to age and gender over nine decades (1998) J Am Coll Cardiol 31: 593-601. https://doi.org/10.1016/s0735-1097(97)00554-8
29. Vinik AI, Maser RE, Mitchell BD and Freeman R. Diabetic autonomic neuropathy (2003) Diabetes Care 26: 1553-1579. https://doi.org/10.2337/diacare.26.5.1553
30. Maser R, Mitchell B, Vinik AI and Freeman R. The association between cardiovascular autonomic neuropathy and mortality in individuals with diabetes: a meta-analysis (2003) Diabetes Care 26: 1895-1901. https://doi.org/10.2337/diacare.26.6.1895
31. DePace NL, Mears JP, Yayac M and Colombo J. Cardiac autonomic testing and diagnosing heart disease: A clinical perspective (2014) Heart International 9: 37-44. https://doi.org/10.5301/heartint.5000218
32. DePace NL, Mears JP, Yayac M and Colombo J. Cardiac autonomic testing and treating heart disease: A clinical perspective (2014) Heart International 9: 45-52. https://doi.org/10.5301/heartint.5000216
33. Bullinga JR, Alharethi R, Schram MS, Bristow MR and Gilbert EM. Changes in heart rate variability are correlated to hemodynamic improvement with chronic CARVEDILOL therapy in heart failure (2005) J Card Fail 11: 693-699. https://doi.org/10.1016/j.cardfail.2005.06.435
34. Fatoni C, Raffa S, Regoli F, Giraldi F, La Rovere MT, et al. Cardiac resynchronization therapy improves heart rate profile and heart rate variability of patients with moderate to severe heart failure (2005) J Am Coll Cardiol 46:1875-1882. https://doi.org/10.1016/j.jacc.2005.06.081
35. Fathizadeh P, Shoemaker WC, Woo CCJ and Colombo J. Autonomic activity in trauma patients based on variability of heart rate and respiratory rate (2004) Crit Care Med 32: 1300-1305. https://doi.org/10.1097/01.ccm.0000127776.78490.e4
36. Peng‐sheng C, Chung‐chuan C, Tan AY, zhou S, Fishbein MC, et al. The mechanisms of atrial fibrillation (2006) J Cardiovasc Electrophysiol 17: S2-S7. https://doi.org/10.1111/j.1540-8167.2006.00626.x
37. Copie X, Lamaison D, Salvador M, Sadoul N, DaCosta A, et al. Heart rate variability before ventricular arrhythmias in patients with coronary artery disease and an implantable cardioverter defibrillator (2003) Ann Noninvasive Electrocardiol 8: 179-184. https://doi.org/10.1046/j.1542-474x.2003.08302.x
38. Alter P, Grimm W, Vollrath A, Czerny F and Maisch B. Heart rate variability in patients with cardiac hypertrophy-relation to left ventricular mass and etiology (2006) Am Heart J 151: 829-836. https://doi.org/10.1016/j.ahj.2005.06.016
39. Debono M and Cachia E. The impact of cardiovascular autonomic neuropathy in diabetes: is it associated with left ventricular dysfunction? (2007) Auton Neurosci 132: 1-7. https://doi.org/10.1016/j.autneu.2006.11.003
40. Just H. Peripheral adaptations in congestive heart failure: a review (1991) Am J Med 90: 23S-26S.
41. Nakamura K, Matsumura K, Kobayashi S and Kaneko T. Sympathetic premotor neurons mediating thermoregulatory functions (2005) Neurosci Res 51: 1-8. https://doi.org/10.1016/j.neures.2004.09.007
42. Manfrini O, Morgagni G, Pizzi C, Fontana F and Bugiardini R. Changes in autonomic nervous systemactivity: spontaneous versus balloon-induced myocardial ischaemia (2004) Eur Heart J 25: 1502-1508. https://doi.org/10.1016/j.ehj.2004.03.019
43. Vinik AI, Maser RE and Nakave AA. Diabetic cardiovascular autonomic nerve dysfunction (2007) US Endocrine Dis 2: 66-74.
44. Clarke B, Ewing D and Campbell I. Diabetic autonomic neuropath (1979) Diabetologia 17: 195-212.
45. Arora RR, Bulgarelli RJ, Ghosh-Dastidar S and Colombo J. Autonomic mechanisms and therapeutic implications of postural diabetic cardiovascular abnormalities (2008) J Diabetes Sci Technol 2: 568-571. https://doi.org/10.1177/193229680800200416
*Corresponding author:
Gary L Murray, The Heart and Vascular Institute, 7205 Wolf River Blvd, Germantown, TN, 38138, USA, Tel: 901-507-3100, Fax: 901-507-3101, E-mail: drglmurray@hotmail.com
Citation:
Murray LG and Colombo J. Routine measurements of cardiac parasympathetic and sympathetic nervous systems assists in primary and secondary risk stratification and management of cardiovascular clinic patients (2019) Clinical Cardiol Cardiovascular Med 3: 27-33.
Keywords
Cardiac parasympathetic, Cardiac sympathetic, Coronary diseases, Heart failure.


 PDF
PDF