Introduction
The advent of the motorvehicle nearly one century ago has greatly altered modern life. While modern industry and technology continue to develop, it is easy to enjoy the benefits of these improvements rather than consider their consequences. Motor vehicles, the most commonly used form of transportation, are highly regarded for their convenience. However, motor vehicles produce whole body vibration (WBV). Though motor vehicles have been greatly improved in recent years, WBV does still exist and its potentially devastating aftermath must be understood. In 2011, there was an estimated 5,338,000 police-reported motor vehicle traffic crashes. According to a recent study by Charlie Klauer of the Virginia Tech Transportation Institute Center for Vulnerable Road User Safety, fatigue is the cause of 20 percent of all U.S. automobile crashes. Our study on motor vehicle-induced-whole body vibration (MVWBV) challenges the previous notion of driver fatigue [1,2]. These results suggest that “driver fatigue” I actually brain impairment and dysfunction caused by MV-WBV. To further this study, we hypothesize that MV-WBV can produce accumulated brain injuries that compromise drivers cerebral function, such as judgment and reactive capabilities, and can be the cause of motor vehicle accidents. Long term MV-WBV is an important risk factor of cerebrovascular diseases and stroke. On wide literature review, it is evident that truck drivers have a higher incidence rate of hypertension, cerebrovascular diseases and stroke [3-5]. Prevention of early neuronal injury from MV-WBV can reduce late chronic brain diseases. In our previous study on handarm vibration injury, the 4F-peptide was studied, and its preliminary preventative effects from hand-arm vibration injury were described [6]. The goal of this study is to understand the pathological damage to cerebral capillaries and neurons from MVWBV and to validate the 4F-peptide preventative effect from MV-WBV injury. A study of simulated animal vibration is an effective way to understand this pathologicalprocess and damage as rats are similar to humans in anatomy and physiological and biological features, which has been demonstrated quite effectively in the past [7-9].
Materials and Methods
Ethics statement
For the care and use of laboratory animals, all protocols in this study conformed to the National Institutes of Health (NIH) guidelines and received approval from the Biomedical Resource Center (BRC) and the Institutional Animal Care and Use Committee (IACUC) at the Medical College of Wisconsin (AUA- 2363). After the animals arrived, they were allowed to acclimate for 7 days prior to exposure. Animals were housed in a central animal care facility with 12-h light cycles and given food and water adlibitum.
Animal groups
56 Sprague-Dawley male rats (weight 250-300g) were divided into seven groups (N=8): (1) 8-week normal control: had no treatment; (2) 8-week vibration group (exposed to whole body vibration at 30 Hz and 0.5g acceleration for 4 hours/ day, 5 days/week for 8 weeks); (3) 8-week vibration group with preconditioning 4F peptide; (4) 8-week vibration group with post conditioning 4F peptide; (5) 8-week vibration group with pre and post conditioning 4F peptide; (6) 12-week normal control group; and (7) 12-week vibration group (exposed to the same vibration, 5 days/week for 12 weeks). At the end point, all rats were evaluated by brain histo-pathological studies.
Vibration set-up
The rats in all control and vibration groups were placed individually in poly vinyl chloride (PVC) tubes. Rats voluntarily entered the tubes. The rats did not exhibit stress at all as seen in our previous rat experiments. The tubes were taped to a vibrating platform and the tails were taped alongside the tube on the platform (Figure 1). Vibrations were performed without any sedative or anesthesia. The electromagneticvibration motor (Bruel and Kjaer (B&K), type 4809, Denmark) was driven by a sine wave signal from a function generator (Simpson 420, Elgin, IL). The acceleration was set with a power amplifier (B&K type 2706). Frequency and acceleration were calibrated prior to beginning the study using an HP 1201 B oscilloscope and a B&K 4384 accelerometer connected to a B&K Integrating Vibration Meter, type 2513, with linear vertical oscillations of 30 Hz and 0.5g acceleration (4.9 m/s2 r.m.s. acceleration). These vibration parameters were selected after wide literature review and consultations with US and international human-WBV associations; therefore the vibration parameters chosen simulated the most common motor vehicle driving situations [10-17]. The reliability of this animal model for vibration studies has been identified and compared to other vibration animal models demonstrating that this model is psychologically stressfree by our systematic and continued studies described in our published paper [10].
Conditioning rats with 4F (human apolipoprotein A-I molecule mimetic)
The 4F (Amino acid sequence: Ac-DWFKAFYDKVAEKFKEAFNH2) was synthesized by the Blood Center of Wisconsin. The peptide was reconstituted in normal sterile saline. The preconditioning group received subcutaneous injections of 4F (3mg/kg) every day approximately 30 minutes prior to the daily exposure. Rats were weighed daily to adjust the dosage of 4F as required. The rationale for the administering paradigm of 4F to the rats was as follows: oral dosing in the food or water would not provide enough accuracy; therefore, we administered 4F as a subcutaneous injection to verify the exact amount given to each rat every time. The dosing concentration of 3mg/kg has been shown to be quite effective in reducing the effects of vibration injury as found by our previous study. It is similar to the concentration used in human studies and is at the lower end of other published in vivo studies 4F [6]. The published 4F half-life times vary from 6 to 12 hours. Our hypothesis for the timing of injections is that preconditioning might be most important because the initial WBV effect is vasospasm and 4F is a strong vasodilatorand neuralprotector, which prevents myelin damage as shown in our previous published work on vibration [6]. Having a high circulating concentration of 4F at the onset of vibration might prove to be the best prevention approach. In addition, a recently completed clinical trial titled: “4-EVER: a Trial Investigating the Safety of 4F Endovascular Treatment of Infra-Inguinal Arterial Stenotic Disease” (ClinicalTrials.gov Identifier: NCT01413139) has shown positive results.
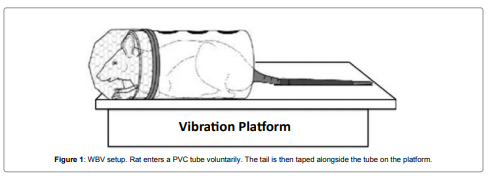
Figure 1: WBV setup. Rat enters a PVC tube voluntarily. The tail is then
taped alongside the tube on the platform.
Tissue processing for Histo-pathological study
Light microscopic (LM) and transmission electron microscopic (TEM) studies on the brain cortices were performed after vibration had completed. At the end time point, the rats were anesthetized using isoflurane gas inhalation. The rat skull was carefully opened using a microsurgical saw under a surgical microscope. On the right side, the right brain cortex (1 mm thickness) was carefully harvested using a #11 blade without any traumatic intervention. Specimens were immediately immersed into a 2.5% glutaraldehyde solution in phosphate-buffered saline. After fixation, they were processed routinely and plastically embedded. Semi-thin 0.5μm transverse sections were cut and stained with toluidine blue for the LM study. Ultra-thin (50-70 nm) sections of the brain cortex, including capillaries, stained with salts of uranyl acetate and lead citrate were prepared for the TEM study. The left cortex tissue was harvested in the same way for H & E (hematoxylin and eosin) stain. The brain membrane was kept intact to avoid artifact neuronal injury and immediately immersed in the 10% formalin in 0.1 M phosphate-buffered saline at pH 7.4. The tissues were then processed for routine paraffin embedding. Blocks were stored at 4°C until H & E staining for cortex neuron injury analysis.
Measurement of capillary dilation degree
In the semi-thin sections, the capillary dilation degree wascalculated as a ratio. The inner endothelial circumference (EC)and the outer pericyte circumference (OC) were measured usingMeta Vue Software (Molecular Devices, Sunnyvale, Ca). Each circumference was manually traced in Meta Vue, and the lengthof each parameter was then calculated by the program. Thecapillary dilation ratio was calculated by dividing EC/OC for eachcapillary. A lumen size change is indicated by the squeezing of theendothelium with an unchanged OC. This is a proven accuratemeasurement [18,19]. A lower mean EC/OC Ratio indicates constricted lumen, and the capillary spasm in the acute WBV stagemay become structural constriction in the chronic WBV stage.
Neuronal pathological analysis
There were two types of histo-pathologically prepared sections for study: 1) semi-thin sections were observed for cerebral microvascular changes and 2) H & E stained sections were observed and analyzed for neuron changes.
Results
General behavior and welfare of all animals were monitored daily. No animal showed abnormal behaviors of stress such as shaking, clenching, biting, postural arching, or clawing. Health inspections included checking for secretions of eyes/nose, porphyrin discharge (chromodacryorrhea), and hematuria. No animal presented any of these ill appearances nor exhibited symptoms of persistent diarrhea, weight loss, light avoidance, lethargy, head waving (inner ear infection), or congested breathing. The vital signs, such as heart rate, respiratory rate, and oxygen saturation of all rats remained stable. Their eating and drinking habits did not change. The growth rate of all rats was within the normal range. This intense observation has shown that this animal model is stress-free, which was further confirmed by our previously published work [6,10,19]. In our subsequent studies, only the WBV factor was tested.
Pathological features in different vibration terms
In both the 8- and 12-week controls (Figure 2A and 2C), the capillaries displayed well-opened oval-shaped lumens with clearly intact basement membranes (BM) of the endothelial cells and cell membranes of the pericytes. The endothelium cell layer of the capillary also maintained a uniform thickness. All of these pathological features suggest no brain impairment or dysfunction. With WBV, noticeable damage was sustained by cerebral capillaries. In the 8-week vibration group the brain capillaries had significant characteristic changes in all the rats compared to a normal control: the endothelium layer of the capillary walls was not of uniform thickness and was partially damaged and broken, the lumens were severely constricted, and edema was present around the capillaries and all neuropils. The capillary walls were also thicker (Figure 2B and 2D). All these features indicated that capillary sclerosis had occurred, and the blood perfusion of the whole brain was dramatically compromised. Each of these effects was more pronounced in the 12-week vibration group (Figure2D) as compared to the 8-week vibration group (Figure 2B). The nueropil backgrounds of both the 8- and 12-week vibration groups are less intense than in the normal controls and vacuoles can be seen; all of these changes are more prominent in the 12-week vibration group, as many vacuoles can be seen in the neuropils (Figure 2D). These characteristic changes indicate extensive chronic edema occurred in the 12-week vibration group. The capillary dilation ratio of the 8-week vibration group was 0.595, and this was smaller than the 8-week control group (0.690); this comparison was statistically significant (p=0.003). A similar comparison pattern was also visible in the vasodilation ratios of the 12-week control vs. vibration groups (0.721 vs. 0.572, respectively) and this was also statistically significant (p<0.0001).The capillary dilation ratio of the 12-week vibration group was 0.572, which was less than the capillary dilation ratio of the 8-week vibration group (0.595), suggesting that longer periods of WBV cause more damage to brain capillaries and lead to increased cerebral impairment by thickening capillary walls and shrinking lumen size to a greater extent. Compared to the 8 week vibration group (Figure 2B), the 12-week vibration capillary (Figure 2D) has a more uneven endothelium layer of the capillary wall. Therefore, more damage occurs to cerebral capillaries and brain impairment occurs to a greater extent with longer periods of WBV. The rats that were conditioned with the 4F peptide before and/ or after vibration had a larger degree of capillary dilation than the 8-week control and 8-week vibration groups. The endothelium layer of these capillaries was also of a uniform thickness and was not damaged or broken. Compared to the 8-week control capillary.
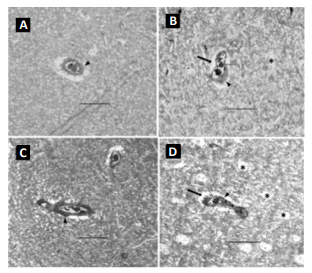
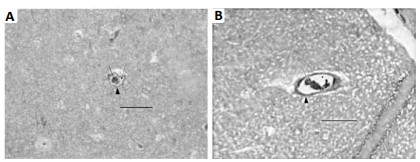
Figure 3: Cross-section of cerebral capillaries. A: 8-week vibration with pre-conditioning 4F peptide. B: 8-week vibration with postconditioning 4F peptide. In both pictures, the solid arrow points to red blood cells. The dashed arrow is pointing to the lumen, which is not constricted in these capillaries. The solid arrowhead is pointing to the capillary wall; in these capillaries, the endothelial layer is of a uniform thickness around the entire capillary. Bar = 50 µm.
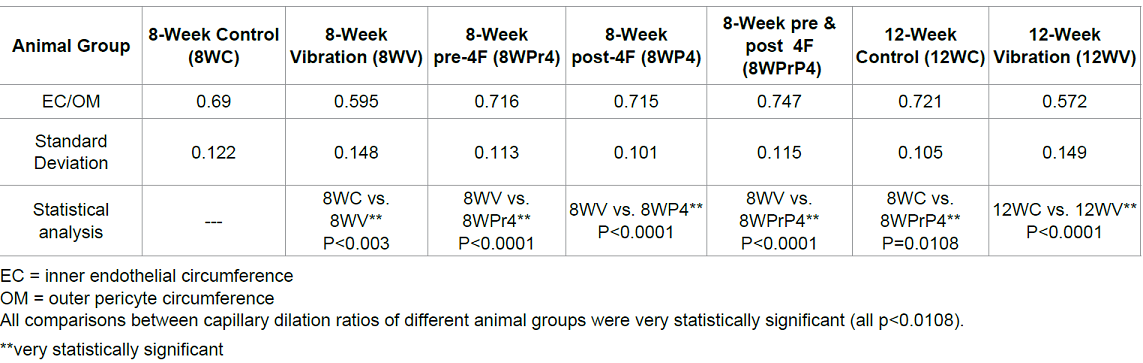
Table 1: Capillary dilation ratio (EC/OM).
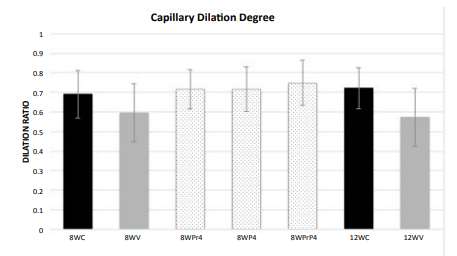
Figure 4: Capillary Dilation Ratio
of each animal group. 8WC: 8-week control; 8WV: 8-week vibration; 8WPr4: 8-week
pre-conditioning 4F peptide; 8WP4: 8-week post-conditioning 4F peptide; 8WPrP4:
8-week pre- and post-conditioning 4F peptide; 12WC: 12-week control; 12WV:
12-week vibration.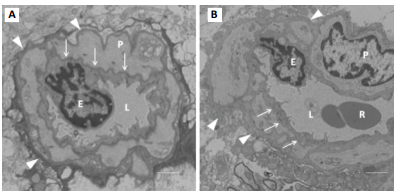
Figure 5: TEM section of cerebral
capillary. P = pericyte cell, E = endothelial cell, R = red cell, L= lumen. A
was from 4-week control group: Arrows indicate basement membrane (BM) that was
a clear intense black line; B was from 4-week vibration group: Its BM was
invisible or disappeared. The arrowheads indicate cell membrane of pericyte
cells that was also clear and intense black structure in A, while in B this
became a coarse, thicker gray structure. The whole capillary wall apparently
presented as thicker and more fibrotic in B than in A. Bar = 1 µm.
Capillaries, as vascular end units, are directly relevant to tissue perfusion, providing oxygen to meet the bodys needs. This study focused on the capillary changes that result from WBV. From our previous study, we found that in shorter periods of vibration (one to two weeks), there is no obvious capillary damage. However, significant damage does occur to capillaries when vibration periods equal or exceed four weeks [20]. As seen in figure 5A (from our previous study), the basement membrane of the 4-week control group is clearly defined. However, in the 4-week vibration group (figure 5B) the basement membrane has nearly disappeared, the cell membranes of the pericyte cells have become coarse and thick, and the entire capillary wall is now thicker and more fibrotic. Therefore, noticeable cerebral damage is sustained after the 4-week vibration period. The insidious nature of the WBV injury coupled with its discretely identifiable sequelae should be seriously regarded. Acute WBV causes cerebral vascular spasm, leading to constriction and ultimately to decreased cerebral blood flow. Neurons begin to sustain impairment. Drivers usually feel fatigued and drowsy during long hours of driving. We believe this brain dysfunction (impairment) is from WBV, which decreases judgment and reactive capability suggesting that it is an important contribution to “driver fatigue.” The exact biological mechanism of WBVinduced vascular and neuronal damage is still unknown. Locally, vibration force both stimulates sympathetic nerve fibers to release norepinephrine and increases the smooth muscle sensitivity to norepinephrine, a twofold amplification of norepinephrine efficacy [21]. The smooth muscle spasm may also be caused by direct stimulation from the vibration shearing force: endothelial cells are physically injured during vasoconstriction by tight pinching between the folds of the internal elastic membrane, which causes bulging into the lumen [22]. The damage to neurons and peripheral nerves is from direct shearing force and ischemia. Relative to the average rat lifespan of 3 years, the average human lifespan in the US is 78 years, which is about 26 times that of the rat lifespan. 8- and 12-week vibration periods in the rats are equivalent to approximately 4.5 and 6 years of vibration to the human respectively. Four and a half years for most occupational drivers is not a very long period, rendering a significant comparable timeline in our 8-week rat models. Furthermore, our pathological study has shown this injury is a gradually cumulative process, as our measured changes at 12 weeks were significantly greater than 8 weeks. These morphological lesions such as thickening and destruction of capillary walls may permanent brain lesions and other secondary diseases, such as chronic hypertension, cerebrovascular diseases, and stroke in the human model. This may suggest that damaging processes such as WBV may play a role in other cerebral diseases and pathologic processes much more extreme than mere drivers fatigue, such as dementia, Alzheimers disease, or even Parkinsons disease. Early preventative approaches, such as the use of protective peptide 4F, could be utilized and potentially stymie these destructive processes. Such future studies, perhaps in human models, would be greatly beneficial to understanding the pathophysiology of these mysterious cerebral disorders.
Acknowledgment
U.S. Army Medical Research & Materiel Command (USAMRMC) and the Telemedicine & Advanced Technology Research Center (TATRC), Fort Detrick, MD, [Grant number W81XWH111069].
Author Disclosure Statement
No competing financial interests exist for all authors.
References
- Yan JG, Zhang LL, Agresti M, Matloub HS, Sanger JR. Mental Judgment Impairment from Whole Body Vibration: Experiment in Rats. (2012) Proceedings of the 4th American Conference on Human Vibration. Hartford Connecticut, USA.
- Yan JG, Zhang LL, Agresti M, Matloub HS, Yan Y, et al. Neural Systemic Impairment from Whole Body Vibration. (2013) Program and Abstract Book of the 5th International Conference on Whole Body Vibration. Academic Medical Center, Amsterdam, Holland.
- Savage R, Billing D, Furnell A. Whole-body vibration and occupational physical performance: a review. (2015) Int Arch Occup Environ Health. 1-17.
- Wolfgang R, Burgess-Limerick R. Whole-body vibration exposure of haul truck drivers at a surface coal mine. (2014) Appl Ergon 45:1700-1704.
- Park MS, Fukuda T, Kim TG, Maeda S. Health risk evaluation of whole-body vibration by ISO 2631-5 and ISO 2631-1 for operators of agricultural tractors and recreational vehicles. (2013) Ind Health 51: 364-370.
- Rowe DJ, Yan JG, Zhang LL, Pritchard KA Jr, Kao DS, et al.. The preventive effects of apolipoprotein mimetic D-4F from vibration injury-experiment in rats. (2011) Hand (N Y) 6: 64-70.
- Bertelli JA, Mira JC, Gilbert A, Michot GA, Legagneux J. Anatomical basis of rat brachial plexus reconstruction. (1992) Surg Radiol Anat 14: 85-86.
- Bertelli JA, Taleb M, Saadi A, Mira JC, Pecot-Dechavassine M. The rat brachial plexus and its terminal branches: an experimental model for the study of peripheral nerve regeneration. (1995) Microsurgery 16: 77-85.
- Yan JG, Eldridge MP, Dzwierzynski WW, Yan YH, Jaradeh S, et al.. Intraoperative electrophysiological studies to predict the efficacy of neurolysis after nerve injury-experiment in rats. (2008) Hand (N Y) 3: 257-262.
- Yan JG, Zhang LL, Yan Y, Sanger JR, Jensen ES, et al. Improved animal model for vibration injury study. (2010) Scand J Lab Anim Sci 37:159-169.
- Malchaire DJ, Piette A, Mullier I. Vibration exposure on forklift trucks. (1996) Ann Occup Hyg 40: 794–791.
- Lewis CH, Griffin MJ. A comparison of evaluations and assessments obtained using alternative standards for predicting the hazards of whole body vibration and repeated shocks. (1998) J Sound Vib 215: 915–926.
- Toward MGR, Gunston TP, Griffin MJ. Evaluation of whole-body vibration exposure in British industry. (2005) Institute of Sound and Vibration Research, University of Southampton.
- Paschold HW. Whole-body vibration: an emerging topic for theSH&E profession. (2008) Professional Safety 52-57.
- Huang Y, Griffin M J. Nonlinearity in apparent mass and transmissibility of the supine human body during vertical wholebody vibration. (2009) J Sound Vib 324: 429–452.
- Paschold HW, Mayton AG. Whole-body vibration: building awareness in SH&E. (2011) Professional Safety 30-35.
- Griffin MJ, Seidel H. Encyclopedia of occupational health and safety. (2011) Geneva: International Labor Organization.
- Curry BD, Govindaraju SR, Bain JL, Zhang LL, Yan JG, et al. Nifedipine pretreatment reduces vibration-induced vascular damage. (2005) Muscle Nerve 32: 639-646.
- Matloub HS, Yan JG, Kolachalam RB, Zhang LL, Sanger JR, et al. Neuropathological changes in vibration injury: an experimental study. (2005) Microsurgery 25: 71-75.
- Yan JG, Zhang LL, Agresti M, LoGiudice J, Sanger JR, et al. Neural systemic impairment from whole-body vibration. (2015) J Neurosci Res 93: 736-744.
- Olsen N, Fjeldborg P, Brchner-Mortensen J. Sympathetic and local vasoconstrictor response to cold in vibration induced white finger. (1985) Br J Ind Med 42: 272-275.
- Curry BD, Bain JL, Yan JG, Zhang LL, Yamaguchi M, et al.Vibration injury damages arterial endothelial cells. (2002) Muscle Nerve 25: 527-534.
Keywords
Cumulative brain injury; Brain dysfunction; Whole-Body Vibration


 PDF
PDF