Background
The World Health Organization recommends parasitological confirmation of malaria either by microscopy or RDT before initiating treatment with ACT, and this is documented in the Uganda National Malaria treatment guidelines [1,2]. The level of malaria endemicity, the urgency of diagnosis, the experience of the physician and cost of the technique are some of the factors that influence the choice of the malaria-diagnostic technique to use [3]. In turn, the technique and quality of diagnosis determine the treatment options, treatment (health) outcomes, and level of resource use. Ideally, an acceptable diagnostic technique should be both cost-effective and provide results that are consistently accurate and timely in order to have a direct impact on treatment [4].
There are three methods of detecting malaria parasites in peripheral blood; microscopy, antigen detection using Rapid Diagnostic Tests and Polymerase Chain Reaction. Microscopy is a recommended method for routine malaria diagnosis because it allows the identification of different malaria-causing parasites ( Plasmodium falciparum , Plasmodium vivax , Plasmodium malariae and Plasmodium ovale ) and quantification of parasite density to monitor response to treatment [5]. The Ministry of Health in Uganda recommends the use of RDT at Health Centre II & community levels, while microscopy is used at Health Centre level III and IV, and hospitals [6]. Microscopy itself is not a magic bullet; cases of misdiagnosis leading to inappropriate treatment still exist in Uganda. As a result, the practice of treating all febrile infections with anti-malaria drugs remains an outstanding challenge [7]. This creates need for appropriate malaria diagnostic strategies that will promote efficient use of resources, reduce costs on the management of malaria and address challenges of presumptive treatment of malaria [8,9]. Microscopy is the major malaria diagnostic technique used in hospital settings in Uganda. Microscopy uses either Fields or Giemsa staining techniques. Fields staining technique is most commonly used in health centres and hospitals although the Uganda Ministry of Health recommends use of Giemsa stain [6].
Mulago National referral hospital like other health facilities mainly uses Fields staining technique for laboratory diagnosis of malaria. Acute Care Unit, a major pediatric ward at Mulago National referral hospital has a malaria prevalence ranging from 30-35%, malaria being the leading cause of complications and death among children less than five years admitted at this ward. Due to the high malaria prevalence, over 75% of children admitted to acute care unit are tested for malaria using Fields staining technique prior to admission therefore the type of management and treatment offered to these children is influenced by the first malaria test results. However, both the accuracy of results and cost of microscopy are determined by the type of stain used. Fields stain takes a short time to results. Giemsa stain has better staining properties and recommended for Quality assurance purposes. Given the limited resources and the need to ensure proper treatment of children admitted to Mulago National Referral Hospital, it is important to determine the cost effectiveness of the staining techniques so as to decide which one is appropriate to take on. This study set out to determine and compare the cost effectiveness of Giemsa and Fields staining techniques in parasitological confirmation of malaria among children under five years received at the Acute Care Unit of Mulago National Referral Hospital in order to inform policy and implementation.
Methods
Study design and setting
This was a cross sectional cost effectiveness analysis study carried out at the Acute Care Unit of Mulago National Referral Hospital. Acute Care Unit is a 24 hour emergency ward and reception center for all pediatric nonsurgical patients. It receives children aged up to 12 years but majority (about 75%) are below five years. . Upon stabilizing their medical condition, they are transferred to general pediatric wards for continuation of care. Acute Care Unit admits 40-50 children daily, has a laboratory which operates 24 hours daily. Malaria is the leading cause of morbidity and mortality among children admitted at Acute Care Unit, Mulago National Referral Hospital and over 90% of these children undergo a parasitological confirmation test for malaria before and/or during care. Routine malaria diagnosis at the Acute Care Unit is by microscopy using Fields staining technique. On average, 20–30 children less than five years are tested for malaria daily. There are seasonal variations in the prevalence of malaria in the ward. The prevalence of malaria among these children ranges between 20% and 35%.
Study population
The study population was made up of 243 children below five years of age presenting at Acute Care Unit laboratory for malaria test before admission.
Sample size determination and sampling
The study sample size was obtained using Buderers method [10] for calculation of power in diagnostic tests based on a standard 2 by 2 table for comparing diagnostic tests. This method was used because determination of effectiveness was based on sensitivity and specificity of Fields and Giemsa staining techniques. Two sample sizes were obtained based on the need for adequate sensitivity and for adequate specificity [10]. Using this method, the number of patients needed for adequate sensitivity was 243 while that for adequate specificity was 351. Given the time and resources available the study used 243 children to determine effectiveness of the staining techniques. Study participants were selected using consecutive sampling, a good sampling method when determining effectiveness or accuracy of diagnostic tests [11]. The study only included children with caregivers consent. Enrollment and determination of effectiveness was done from April 25 to June 15, 2014.
Sampling procedure
Study participants were selected using consecutive sampling. This method has been recommended for use in determining effectiveness or accuracy of diagnostic tests (Knottnerus and Muris, 2003). Eligible children were enrolled into the study as they presented at the acute care unit laboratory for the initial malaria test prior to admission.
Inclusion criteria
All children below five years who were received at acute care unit Mulago National Referral Hospital and presented for the initial malaria test prior to admission were included in the study.
Exclusion criteria
Children below five years who reported to acute care unit Mulago National Referral Hospital for the initial malaria test without caregivers consent were not included in the study.
Cost data collection
The costs of the staining techniques included; direct medical and direct non-medical costs incurred during malaria diagnosis using either Fields or Giemsa staining techniques. Direct medical costs were precisely related to the staining method. They included; laboratory technologists time, cost of reagents, equipment and supplies. Direct non-medical costs incurred in the process of testing were not directly related to the staining method. They included: costs for utilities like water, electricity and laboratory space. The ingredient approach was used for costing each staining technique. This approach involved identification, quantification and valuation of all inputs for the staining techniques in order to obtain unit costs. This was done for the different steps in the staining process which included; smear preparation, smear drying and examination. Costs were obtained in aggregate form and broken down to obtain unit costs in terms of cost per blood smear. These unit costs were summed up to obtain a unit cost for each staining technique. This study included costs incurred while staining of blood smears in either staining techniques only. Costs for steps that were similar in both techniques were excluded from the analysis because they equally incurred in both staining techniques and cannot cause differences in the outcome. These included costs for; blood sample collection, blood smear preparation, smear drying and examination. Overhead costs were assumed to be equally incurred in both staining techniques and were therefore excluded from the analysis. These included cost of; reagent storage, building maintenance, cost of hospital/ laboratory administration, cost of reagent preparation and storage, transportation, cleaning and taxes. Costs of the Polymerase Chain Reaction test were not included in the analysis since this test was only used as a gold standard.
The costs incurred during staining in either technique included; capital costs, reagent costs, labor costs and utility costs. Capital costs included cost of laboratory space and equipment. Reagent costs included costs of all reagents that were used during staining. Labor costs included; Labor laboratory technicians time and cost of utilities (water and electricity) used during staining.
Data on costs was obtained in aggregate form and then disaggregated to come up with unit costs. Cost data was obtained from the market wholesale price, the administration of Mulago National Referral Hospital, and National Medical Stores and General Medical Stores. The United States dollar was used in this study because it is a widely used currency in most Cost Effectiveness Analyses and for aiding comparison. The costs were collected in Uganda shillings and converted to the United States dollars at the existing exchange rate during study period of 1United States dollar to 2550 Uganda shillings (www.oanda.com).
Measurement of effectiveness
Blood samples for determination of effectiveness of Fields and Giemsa staining techniques were collected from 243 children below five years of age who presented at Acute Care Unit laboratory for malaria diagnosis during the study period. Effectiveness of the staining techniques was determined by the number and proportion of correctly diagnosed cases as applied in other studies [12-15]. As applied in a similar study [12], Polymerase Chain Reaction method was used as the gold standard for this study using nested Polymerase Chain Reaction technique for Plasmodium falciparum because Plasmodium falciparum accounts for over 95% of malaria infections in Uganda. Probabilities for stain effectiveness were the calculated positive and negative predictive values of the two staining techniques as compared to Polymerase Chain Reaction method.
Cost effectiveness Analysis model
The decision tree analytic model using Tree Age software was used for the cost effectiveness analysis. Effectiveness probabilities and values together with the providers costs incurred by either technique were used to populate the model in order to determine cost effectiveness of the two staining techniques. The payoffs for the correctly diagnosed cases were the number of correctly diagnosed cases, true positive and true negative cases for both staining techniques while cases that were not correctly diagnosed (false positive and false negatives cases) had a payoff of zero. In both staining techniques, the cost payoffs were the unit costs for the staining technique.
Sensitivity analysis
Use of different sources of costs created uncertainty. In order to address this uncertainty, sensitivity analysis was carried out on unit costs of each staining technique. The costs were halved and doubled in order to get the lower and upper limits of sensitivity ranges respectively. One-way sensitivity analysis was used since only one variable was varied.
Ethical Approval
Ethical approval was obtained from the Makerere University School of Public Health Institutional Review Board. Permission to conduct the study in Mulago National Referral Hospital was also obtained from the Mulago National Referral Hospital Research and Ethics Committee (Protocol MREC539).
Quality Assurance and Control
All study activities were coordinated and supervised by the principle investigator (PI) who is a laboratory technologist with experience in research. The research team comprised of one nurse and three laboratory technologists, all with more than ten years of experience in their fields. These were briefed about their activities prior to data collection by the PI. The PI actively participated in ensuring quality data collection and documentation. Results for Giemsa and Fields stains were filed and kept separately. PCR tests were conducted in a separate and highly specialized research laboratory by highly qualified laboratory technologists. Information on blood samples was checked for consistency with that on the study forms. We ensured uniformity in labeling on the forms and blood samples. Rounding off of costs during disaggregation was avoided.
Results
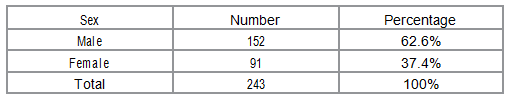
Table: Demographic characteristics of study participants
Demographic characteristics of study participants
Costs
Various costs incurred when using either Fields or Giemsa staining technique were identified and sorted into three broad categories; reagent costs, labor costs and equipment costs as indicated in Table 1. Unit costs were US $ 0.030 and US $ 0.769 for Fields and Giemsa staining techniques respectively. The percentage of reagent costs as a proportion of total unit costs of the staining techniques were 5.19% and 0.446% for Fields and Giemsa staining techniques respectively. Labor costs comprised of 89.63% and 99.469% of the unit costs of Fields and Giemsa staining techniques respectively. The corresponding percentage of equipment costs as a proportion of total unit costs for the staining techniques were 5.18% and 0.085% respectively.
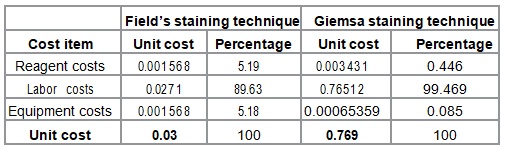
Table 1: Unit costs for the staining techniques.
Effectiveness
Effectiveness of each staining technique was determined by the number and proportion of cases correctly diagnosed using Giemsa and Fields staining techniques with PCR method as the gold standard (Table 2). Effectiveness probabilities for populating the decision tree analytic model were calculated and are presented in Table 3. The numbers of correctly diagnosed cases were 227 and 230 for Fields and Giemsa staining techniques respectively. The corresponding proportion of correctly diagnosed cases was 93.4% and 94.7% respectively. Effectiveness probabilities were calculated using epidemiological principles for determination of diagnostic accuracy based on 2X2 table [16]. The probabilities included; the positive and negative predictive values, and their complementary probabilities. The calculated probabilities together with the number of correctly diagnosed cases were populated in the analysis model to obtain expected values for either staining techniques. The expected values were the basis for comparison of effectiveness and cost effectiveness.
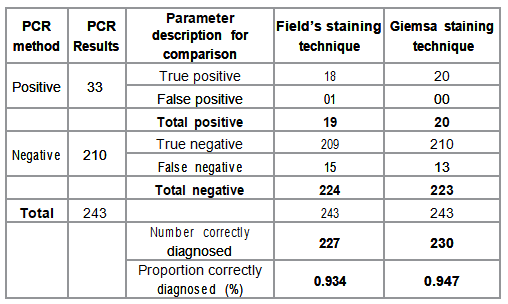
Table 2: Stain effectiveness as compared with PCR method.
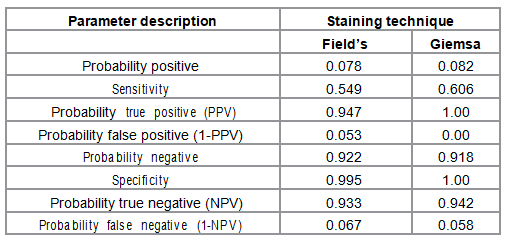
Table 3: Effectiveness Probabilities for the staining techniques.
Cost effectiveness analysis
Cost effectiveness analysis using both costs and number of correctly diagnosed cases The cost effectiveness analysis indicated that Fields staining technique was more costs effective than Giemsa staining technique (Figure 1). Figure 2 provides a graphical presentation of the cost effectiveness analysis. Since Giemsa had higher effectiveness and higher costs than Fields staining technique, the graph further emphasizes the importance of the decision tree analysis as a way of determining the more cost effective staining technique.
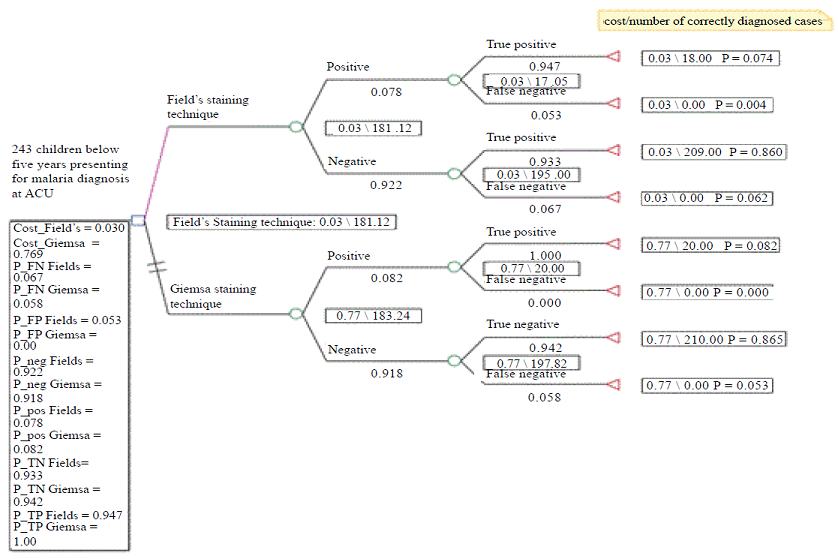
Figure 1: Decision Tree for Cost Effectiveness Analysis.
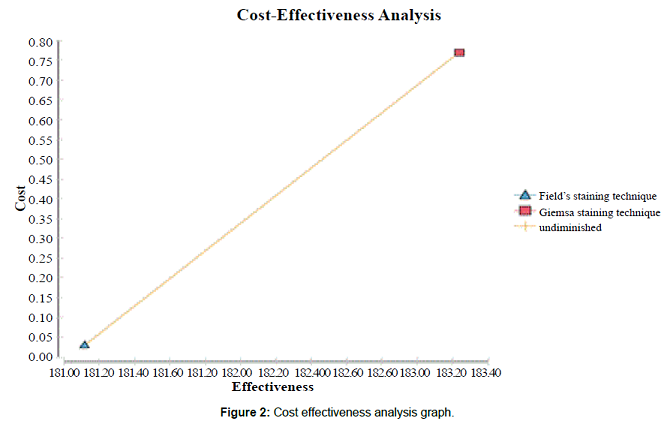
Figure 2: Cost effectiveness analysis graph.
Incremental cost effectiveness analysis ratio ( ICER )
Table 4 indicates the cost effectiveness rankings obtained from the cost effectiveness analysis. These include; costs, effectiveness and provided the incremental cost effectiveness ratio for staining techniques based on the expected values generated in the decision tree analysis. The incremental costs and incremental effectiveness were 0.74 US $ and 2.12 correctly diagnosed cases respectively while the ICER was 0.35 US $ per additional correctly diagnosed case.
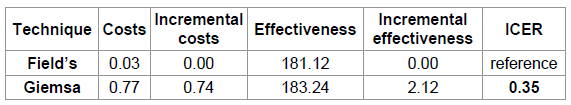
Table 4: Cost effectiveness rankings.
Sensitivity analysis
We conducted one-way sensitivity analysis using TreeAge. This was done by varying the costs (Table 5) of Fields and Giemsa staining techniques on assumption that other parameters remained constant. In both instances, Giemsa staining technique remained more cost effective than Fields staining technique and the ICER remained was not affected. The ICER remained constant at 0.35 even with varying these costs.
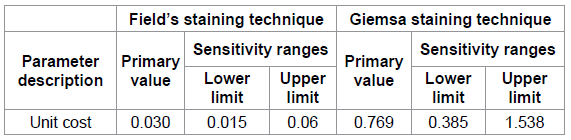
Table 5: Sensitivity Analysis Ranges.
Discussion
The World Health Organization and Uganda Ministry of Health recommends microscopy using Giemsa staining technique for the parasitological confirmation in the diagnosis of malaria [6]. Microscopy using Giemsa staining technique provides quality test results and is considered to be more effective than Fields staining technique. However, its cost effectiveness is still debated. Most health facilities in Uganda that still use Fields staining technique argue that Giemsa has a high time to results compared to Fields stain which increases its cost.
Costs of staining techniques
Unit costs for Fields and Giemsa staining techniques were 0.030US $ and 0.769 US $ respectively. This implied the cost of Giemsa staining technique was 25.6 times higher than that for Fields staining technique. Staining with Giemsa takes 32 minutes compared to Fields that takes 1 minute and 8 seconds. Labor costs were the highest cost drivers in both staining techniques constituting of 89.63% and 99.469% of unit costs for Fields and Giemsa staining techniques respectively. This finding is similar to what was found in previous studies on costs of malaria diagnosis using microscopy which also reported labor costs as the highest cost driver [12, 14, 15].
There was a slight difference (0.001863 US $ ) in reagent costs incurred in the staining techniques although the proportion of reagent costs was higher for Fields staining technique compared to that for Giemsa staining technique. Reagent and equipment costs for the Fields staining technique were almost equal at 5.19% and 5.18% of the unit cost respectively. Proportions of reagent and equipment costs were higher for Fields staining technique (5.19% and 5.18%) than for Giemsa staining technique (0.085% and 0.446%). This is because unlike Giemsa, Fields staining technique requires more reagents and equipment but reagents needed in Fields staining technique are cheaper than those for Giemsa staining technique. Fields staining technique was found to be more cost saving and this partly accounts for its preference in high malaria diagnosis workload in resource limited settings Effectiveness of the staining techniques Giemsa staining technique was more effective than Fields staining technique. The number of correctly diagnosed cases was 227 and 230 for Fields and Giemsa staining techniques respectively. The corresponding proportion of correctly diagnosed cases was 93.4% and 94.7% respectively. There was a slight difference in number and proportion of correctly diagnosed cases of malaria by the two staining techniques.
The sensitivity of Fields and Giemsa staining techniques were 54.9% and 99.5% respectively while sensitivity was 60.6% and 100% for Fields and Giemsa staining techniques respectively. This results do not very different from those of a similar study that reported 47.2% and 46.1% sensitivity and 93.4% and 97.2% specificity for Fields and Giemsa respectively [17].
The positive predictive values were 0.947 and 1.00 while corresponding negative predictive values were 0.933 and 0.942 for Fields and Giemsa staining techniques respectively. The expected effectiveness value for Giemsa staining technique was 183.24 while that for Fields staining technique was 181.12. This indicates that Giemsa was slightly more effective than Fields staining technique.
Cost effectiveness of Giemsa compared to Fields staining technique
Fields staining technique was more cost effective than Giemsa staining technique. This is because the cost of Giemsa staining technique is higher than Fields staining technique; yet, Giemsa staining technique has a slightly higher effectiveness than fields staining technique. The incremental cost effectiveness ratio was 0.35 $ per additional correctly diagnosed case of malaria. This implies that based on the findings of this study, every additional correctly diagnosed case of malaria obtained by moving from Fields to Giemsa staining technique cost the provider 0.35 US $ which is 8.2 times higher than the unit cost of Fields staining technique.
Sensitivity analysis
Results of the sensitivity analysis indicated that the Incremental Cost Effectiveness Ratio was not affected by varying the costs of the staining technique. Based on the expected values from the cost effectiveness analysis it remained constant at 0.35. The results of the sensitivity analysis indicated that the cost effectiveness analysis model was robust.
Study Limitations
The use of number and proportion of correctly diagnosed cases of malaria as the outcome posed a limitation for this study. This is an intermediate outcome that is assumed to be linked to improved final outcome, recovery from disease. The link between correctly diagnosing a case, optimal clinical management of the patient, and a satisfactory health outcome may be difficult to prove without a close patient follow up. Within the scope of this study, it was not possible to estimate the link between incorrectly diagnosed cases of malaria and the final clinical outcome. This is because no patient follow up was made hence further research will be required in this area. Another limitation of this study was use of the providers perspective. However, in this study the provider perspective was used because we only considered the costs of providing these staining techniques, although there are some indirect costs incurred by the consumer which were not included.
Confounding Factors
This study had minimal confounding. The possible source of confounding for this study could have been the difference in malaria slide preparation and microscopy slide reading. This was addressed by using qualified and highly experienced (over 10 years experience) laboratory technologists in laboratory diagnosis of malaria in a busy hospital setting.
Conclusion
Drawing from findings of this study, Field’s staining technique was more cost effective than Giemsa staining technique. It provided a higher number of correctly diagnosed cases of malaria at a lower cost than Giemsa staining technique. With Uganda statistics of 2013 indicating a Gross Domestic Product (GDP) per capita of 1365.13 US dollars, an of ICER of 0.35 US dollars indicates that Field’s staining technique is affordable. This study therefore recommends the use of Field’s staining technique for routine microscopy for the parasitological confirmation of malaria diagnosis limited resource settings like the Acute Care Unit at Mulago National Referral Hospital and Uganda at large, and in other low income countries. Implementation of the Ministry of Health’s recommendation to use Giemsa staining technique should be promoted when adequate resources have been made available to support it. This study highlights the need to incorporate cost effectiveness analyses in decision making process to inform policy and implementation.
Competing Interests
The authors declare that they have no competing interests
Authors’ Contributions
NJ participated in the inception, design, implementation of the research, analysis and interpretation of findings as well as writing the manuscript. NSL participated in the inception and design of the research and also conducted PCR assays. YA was involved in drafting the manuscript through providing critical review and gave final approval of the version to be published. CM participated in the analysis, interpretation of data and was involved in drafting the manuscript. SOB participated in analysis, interpretation of data, drafting the manuscript and gave final approval of the version to be published.
Authors’ Information
NJ, currently Research Associate at Makerere University School of Public Health was a student of Master of Health Services Research at Makerere University at the time of conducting the research. NSL is the Laboratory Director of the Makerere University-University of San Francisco California Molecular Laboratory under the Infectious Diseases Research Collaboration. YA is a senior Epidemiologist at the Uganda Malaria Surveillance Project. CM is an assistant Lecturer under the Department of Health Policy, Planning and Management at the School of Public Health, Makerere University. SOB is a Senior Lecturer under the Department of Health Policy, Planning and Management at the School of Public Health, Makerere University
Acknowledgement
This research was made possible by Uganda Malaria Clinical Operational and Health Services (COHRE) Training Program at Makerere University, Grant #D43-TW00807701A1, from the Fogarty International Center (FIC) at the National Institutes of Health (NIH). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of FIC or NIH.
References
1. Ministry of Health M.O.H, (2008) Guidelines for treatment of malaria.
2. WHO (2012) World Malaria report.
3. Tangpukdee N, Duangdee C, Wilairatana P, Krudsood S. Malaria diagnosis: a brief review. (2009) Korean J Parasitol 47: 93-102.
4. WHO (2013) WHO Bulletin.
5. Bronzan RN, McMorrow ML, Kachur SP. Diagnosis of malaria: challenges for clinicians in endemic and non-endemic regions. (2008) Mol Diagn Ther 12: 299-306.
6. Ministry of Health M.O.H (2013) Uganda National Guidelines for implementation of Parasite Based Diagnosis of Malaria, Uganda.
7. Ndyomugyenyi R, Magnussen P, Clarke S. Diagnosis and treatment of malaria in peripheral health facilities in Uganda: findings from an area of low transmission in south-western Uganda. (2007) Malar J 6: 39.
8. Nankabirwa J, Zurovac D, Njogu JN, Rwakimari JB, Counihan H, et al., Malaria misdiagnosis in Uganda--implications for policy change. (2009) Malar J 8: 66.
9. Namagembe A, Ssekabira U, Weavr MR, Blum N, Burnett S, et al., Improved clinical and laboratory skills after team-based, malaria case management training of health care professionals in Uganda. (2012) Malar J 11: 44.
10. Buderer NM. Statistical methodology: I. Incorporating the prevalence of disease into the sample size calculation for sensitivity and specificity. (1996) Acad Emerg Med 3: 895-900.
11. Knottnerus J, Muris J. Assessment of the accuracy of diagnostic tests: the cross-sectional study. (2003) Journal of clinical epidemiology 56:1118-1128.
12. Batwala V, Magnussen P, et al., Cost-effectiveness of malaria microscopy and rapid diagnostic tests versus presumptive diagnosis: implications for malaria control in Uganda. (2011) Malar J. 10: 372.
13. Shillcutt S, Morel C, et al., Cost-effectiveness of malaria diagnostic methods in sub-Saharan Africa in an era of combination therapy. (2008) Bulletin of the World Health Organization, 86: 101-110.
14. Uzochukwu BS, Obikeze EN, Onwujekwe OE, Onoka CA, Griffiths UK. Cost-effectiveness analysis of rapid diagnostic test, microscopy and syndromic approach in the diagnosis of malaria in Nigeria: implications for scaling-up deployment of ACT. (2009) Malar J 8: 265.
15. Chanda P, Castillo-Riquelme M, Masiye F. Cost-effectiveness analysis of the available strategies for diagnosing malaria in outpatient clinics in Zambia. (2009) Cost Effectiveness and Resource Allocation. 7: 5.
16. Szklo M, Nieto FJ, Miller D. Epidemiology: beyond the basics. (2001) American Journal of Epidemiology. 153: 821-822.
17. Batwala V, Magnussen P, Nuwaha F. Are rapid diagnostic tests more accurate in diagnosis of Plasmodium falciparum malaria compared to microscopy at rural health centres. (2010) Malar J 9: 349.
*Corresponding author
Juliana Namutundu, Makerere University, School of Public Health, P.O.BOX 7072, Kampala, Uganda, Tel: +256 41 4533332 E-mail: namutundu@yahoo.com
Citation
Namutundu J, Lubwama NS, Adoke Y, Mayora C, Baine SO (2016) Cost Effectiveness of Giemsa versus Field’s Staining Technique: Implications for Malaria Diagnosis among Children in a Busy Hospital Setting in Uganda. NHC 106: 26-32


 PDF
PDF