
Explore journal overview, editorial leadership, indexing, articles in press, latest published work, and highlights from previous issues.
Research Article :
In principal, three endocrinesystems are involved in the regulation of carbohydrate metabolism in amphibians: the pancreatic system, the thyroid hormones and the adenohypophysis-interrenal axis [1]. Although this holds also true for mammals and fish, many pronounced species specificities make it rather unlikely, that development of carbohydrate regulation in vertebrates followed a phylogenetic stringent scheme. Each of the classes of vertebrates has developed different mechanisms for the regulation of carbohydrate metabolism, and major differences exist already within different species of each class [2]. Therefore, hormones exert many different effects on the carbohydrate metabolism in amphibians and mammals. Because of the typical life cycle of amphibians which includes a metamorphosis from larvae or tadpoles to adult animals, the stage-specific regulation of carbohydrate metabolism is an important prerequisite for normal amphibian development. Hormones like insulin, glucagon and thyroxine work together with corticosteroids to regulate the glycogen levels in liver and muscle and/or the blood glucose level in a very stage-specific manner [3]. It has been shown that the same hormones may not only act in different ways but also exert opposite effects during different developmental stages of amphibian animals [4-6]. However, the precise way in which this stage-specific regulation takes place and how the involved hormones interact with each other is still poorly understood. In this study, the South African clawed toad Xenopuslaevis (Daudin), an anuran amphibian, was taken as a model organism to investigate the hormonal regulation of glycogen metabolism in adult amphibians, because it was known from former studies to have rather stable glycogen content as found under various experimental conditions [7]. Under physiological conditions the liver glycogen content of adult Xenopus laevis toads ranges between 10 and 20% of the liver wet weight [8]. In naturally occurring populations of Xenopus laevis this value may be influenced to a great extent by a number of different environmental factors, such as seasonal changes, temperature, or food supply, as well as by the age and sex (gender roles) of the animals [9].Preliminary investigations of the glycogen metabolism in the adult toad have shown that prolonged starvation (up to 60 days), exposure to cold (1° C-4° C) and application of various hormones did not cause the glycogen content of the Xenopus liver to be reduced to values below 10% [10].It was the aim of this study to analyze systematically the effects of hormones and various substances relevant to the glycogen/glucose balance in adult Xenopus toads of both sexes. In addition, we examined ultrastructural changes in the hepatocytes induced by the different treatments. We also intended to elucidate the regulating mechanisms of liver glycogen turnover by histochemical detection of glycogen-relevant enzymes.
Materials
If not cited otherwise, the chemicals used were products of Merck and Fluka and were of analytical grade. Buffer solutions were always prepared with tri-distilled water.
Animals
Many of the male and female Xenopus laevis (Daudin), South African clawed toads, were either purchased from a breeding colony maintained by Dr. Ch H Thiebaud of the Institute for Experimental Zoology, University of Geneva (Switzerland), or were kindly donated by Prof. Dr. L Du Pasquier of the Basel Institute for Immunology (Basel, Switzerland). The majority of the animals used, however, were bred and reared in our own laboratory. The normal table of Nieuwkoop and Faber (1975) was used to determine the stages [11]. The animals were maintained in large plastic tanks with filtered tap water which was at 18° C - 20° C. The animals were kept under a light regimen of approximately 12-hr light/12-hr dark. The toads were fed chopped beef heart twice a week. The 2-4 years old animals weighed 30-50 g (males) and 60-90 g (females). One day before the start of each experiment feeding was stopped in order to eliminate dietary variations. Experiments were done with male and female groups of five animals each. The experiments were carried out in accordance with the guidelines of the Swiss Animal Care Decree, and were approved by the Cantonal Veterinary Office of Basel, Switzerland.
Hormonal treatments and biochemical analyses
The concentration for each hormone or substance is indicated in table 1. A volume of 0.1-0.5 ml of the hormone suspension (in 0.72% NaCl) was injected into the dorsal lymph sac of each animal. Control animals were injected with 0.1 ml of 0.72% NaCl alone. The duration of exposure and the number of injections are summarized in (Table 1). Before anesthesia by immersion in 1% MS-222 the toads were injected 1250 I.U. Liquemin (Heparin). Blood was collected by cardiac puncture, mixed immediately with 3.5% sodium citrate and centrifuged for 15 min at 3000 rpm. The supernatant was stored at 4°C prior to analysis. Liver and muscle glycogen contents were determined biochemically according to the method of Roe and Dailey (1966) [12]. The blood glucose was measured according to Schmidt (1961) using the glucose determination kit from Boehringer, Mannheim, Germany [13]. Blood lipids were determined according to Trinder (1969) using the lipid determination kits from Boehringer, Mannheim, Germany [14].
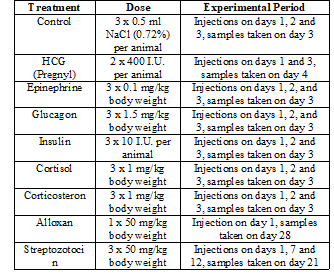
Table 1: Hormonal treatments, hormone doses and duration of experiments.
Electron microscopy
Samples of hepatic tissue and hind leg muscle were immersed into ice cold fixative (1% OsO4 in Soerensen phosphate buffer at pH 7.2 and 175 mOsmol). Fixation was carried out for 2 hr. After acetone dehydration the specimens were embedded in Epon. After polymerization 1.5 µm semithin sections were cut with glass knives and stained with 1% p-phenylendiamine for phase contrast microscopy. 60-90 nm ultrathin sections were cut with diamond knives and stained with 5% aqueous uranyl acetate and lead citrate [15]. Sections were examined with a Philips EM CM 100 operated at 80 kv. Images were digitally recorded and processed using the software Analysis (Soft Imaging System GmbH, Münster, Germany).
Histochemistry
Liver samples were frozen in liquid nitrogen and were stored at -80° C before use. 10 µm thin cryosections of unfixed material were cut at a cryostat temperature of -20° C on a cryostat (MICROM GmbH, Walldorf, Germany), thawed on cover slips, and air dried for several minutes.
Glucose-6-Phosphatase (G-6-Pase): After 5 min of air drying at room temperature, the sections were incubated in a medium containing lead nitrate as modified by Maly and Sasse (1983) [16,17]. After incubation and rinsing, the sections were mounted in glycerol jelly.
Glycogen phosphorylase: Glycogen phosphorylase activity was tested in sections which were air dried for 10 min after 1 h incubation (37° C) in a medium described by Takeuchi and Kuriaki (1955) and modified by Lindberg and Palkama (1972) [18,19]. After incubation, the sections were rinsed and stained in a sucrose-Lugol solution (10:1). Mounting was carried out in Lugol-glycerol jelly (2:5).
The enzyme activity was quantified by semiquantitative evaluation of the staining intensity, a method which despite its shortcomings seemed to be justified, since all the examined slides were prepared under identical conditions. Thus any general mistake should affect controls as well as experimental preparations.
Glyceraldehyde-3-Phosphate dehydrogenase:The activity of this NAD-dependent cytosolic enzyme was demonstrated by the methods of Henderson (1976) and De Schepper, et al. (1985) as modified by P. Maly (personal communication). Mounting was carried out in Mowiol [20,21].
Statistical analysis
The statistical significance of our results was ascertained by a two-tailed Students t-test. The values of 5 independent replicates were expressed as mean ± the Standard Error of the Mean (SEM), as indicated in the figures.
Biochemical analyses
Liver glycogen: The biochemical determination of the Xenopus liver glycogen showed that, with one exception, the glycogen content was neither reduced nor augmented considerably through the application of insulin, glucagon, epinephrine, cortisol, corticosterone, human chorionic gonadotropin, alloxan, or streptozotocin, as compared to the controls. The mean values of the liver glycogen contents of males ranged between 15% and 20% of the liver wet weight, whereas the liver glycogen contents of females ranged between 10% and 15%. We detected a slight reduction in the glycogen contents of males treated with alloxan, but this difference was not statistically significant.
Only human
chorionic gonadotropin which induces the vitellogenic response resulted in
a marked decrease of the liver glycogen stores in females. Their glycogen
content dropped to less than 5% as compared to about 15% in control females.
Contrasting this, the male liver glycogen content remained either stable or was
even increased to a certain extent (Figure
1).
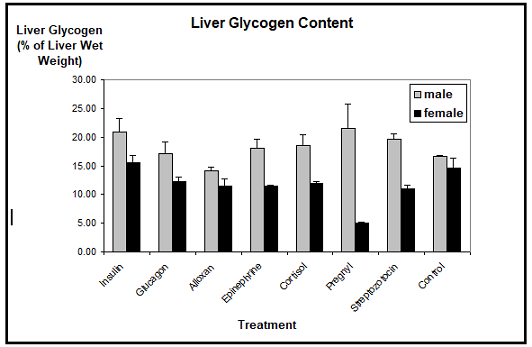
Muscle glycogen: The analysis of the muscle glycogen content did not reveal significant differences between the different hormonal treatments and the controls in Xenopus males and females (Figur 2). The mean values of the muscle glycogen contents of males ranged between 0.35% and 0.45% of the muscle wet weight, whereas the muscle glycogen contents of females ranged between 0.25% and 0.32%. Only alloxan treated males showed a slightly reduced muscle glycogen content, paralleling the findings for the liver glycogen content.
Blood glucose: The biochemical detection of blood glucose showed significant differences in blood sugar levels, not only between the different hormone applications and the controls, but also between the sexes (Figure 3). The average blood glucose level in control animals was about 50 mg/dl in males and about 40 mg/dl in females. In males, this value remained unchanged after application of insulin, glucagon, alloxan, HCG, or streptozotocin.
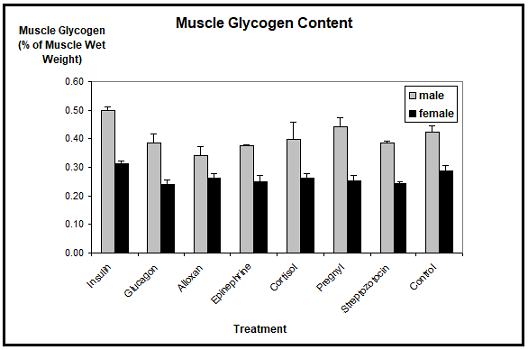
Only the injections of either epinephrine or cortisol resulted in an elevated blood glucose level, which however was only significant with epinephrine. The blood glucose level was in this case more than doubled. In females, insulin resulted in a marked decrease, and glucagon in a slight increase of the blood glucose concentration. Epinephrine (three times) and cortisol (two times) drastically elevated the blood glucose levels in females. Alloxan, HCG, and streptozotocin had no effect.
Note: * (** / ***) significantly different from control at p £ 0.05 (0.01/ 0.001).
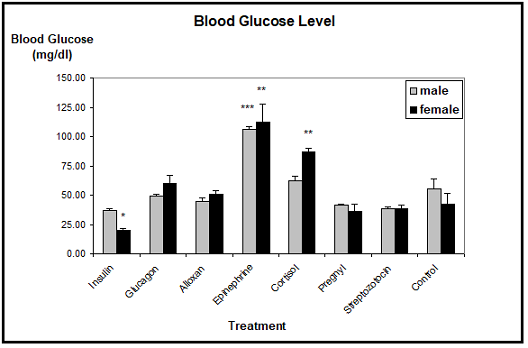
Blood lipid: The average level of blood triglycerides in control animals was about 15 mg/dl in males and about 18 mg/dl in females. The male blood lipid levels were only changed by insulin, epinephrine and cortisol, where triglycerides tended to be elevated. Unfortunately, these results were not found to be significant due to the rather high Standard Errors of the Mean. In females, insulin slightly decreased the blood triglycerides, whereas epinephrine, cortisol and streptozotocin increased the lipid level. However, only the mean value of the cortisol treated females proved to be significantly different from the controls (Figure 4).
Note: *significantly different from control at p £ 0.05.
Effects of hormones on the ultrastructure of hepatocytes
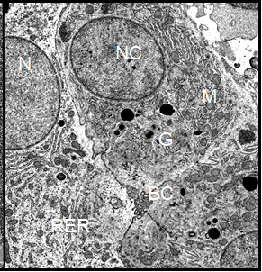
Light microscopic examination of p-phenylendiamine stained semithin sections showed that the liver parenchyma of Xenopus laevis was composed of cords of several polyhedral cells. The nuclei of the hepatocytes usually occupied the pole opposite from the bile canaliculus. Except for some basophilic substances consisting of RER cisternae, dictyosomes of the Golgi complex, and mitochondria in the cell periphery and in the peribiliary region, most of the cytoplasm was occupied by an amorphous, dark material which by ultrastructural examination was determined to be glycogen. We also observed large pigment aggregates embedded in the liver parenchyma. By examination of semithin sections we detected only slight differences in the cellular morphology of the hepatocytes taken from differentially treated Xenopus males and females. The primary purpose of this technique was to give a survey of the unusual structural organization of the Xenopus liver parenchyma (Figure 5). Electron microscopic examination of hepatocytes taken from Xenopus laevis male controls showed an ultrastructural picture which was characterized by its most prominent feature, the large amount of glycogen deposits. The hepatocytes contained more or less evenly distributed glycogen particles of the adult alpha type forming typical rosettes. The cells also contained some lipid droplets which were mostly present in groups of three to five individual droplets. Occasionally, some small and rounded mitochondria were visible at the periphery of the hepatocytes.
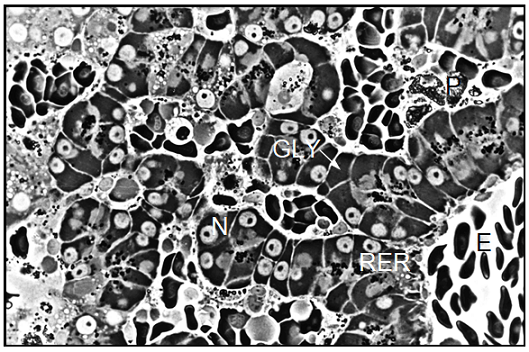
The parenchymal liver cells of male controls showed few signs of synthetic activity. This was reflected by the small amount of endoplasmic reticulum and of dictyosomes in these cells. The nuclei sometimes showed indentations and contained a lightly staining nucleolus. Surface invagination and densely packed chromatin indicated that these nuclei were metabolically inactive, showing limited production of nuclear ribonucleic acid (Figure 6). In general, the morphology of hepatocytes in Xenopus female controls was very similar to the ultrastructure of male cells. However, we found more cell organelles in the female liver cells, indicating a higher level of activity (Figure 7). In general, the morphology of parenchymal liver cells was neither significantly changed by the pancreatic hormones nor by the diabetes inducing substances alloxan and streptozotocin as compared to the cells from controls.
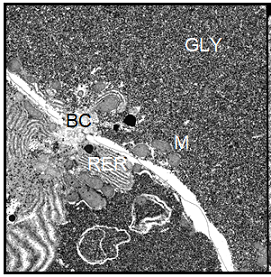
Note: BC=Bile Canaliculus, G=Golgi apparatus, GLY=Glycogen, L=Lysosomes, M=Mitochondrion, RER=Rough Endoplasmic Reticulum, LD=Lipid Droplet, N=nucleus.
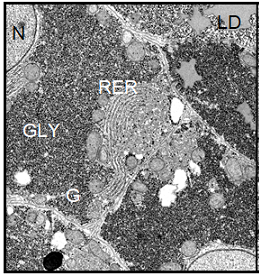
Figure 6:
(Left) Electron micrograph showing parenchymal liver cells from a Xenopus laevis control male. The
ultrastructural picture is characterized by the large amount of deposited
glycogen and represents a basic level of cellular and protein synthetic
activity. Magnification: ´ 4700.
The hepatocytes of insulin treated males contained slightly more glycogen than the cells of control males. Except for the lipid droplets, the content of all other cell components was slightly decreased, indicating that the synthetic activity in these cells was reduced. In contrast, the insulin treatment had little if any effect on the ultrastructure of female hepatocytes. The parenchymal liver cells of glucagon treated toads showed only few inconspicuous differences compared to the control. The cells still contained large areas filled with glycogen particles, and the amount of cell organelles reflected a basic level of cellular activity. Thus, the ultrastructural observation well supported our biochemical data, according to which the liver glycogen content was not decreased by the glucagon treatment.
In Xenopus toads treated with alloxan, slightly less glycogen was present than in the controls, again paralleling our biochemical data. The hepatocytes from streptozotocin treated toads showed no signs of an altered ultrastructure. It appears that neither the toxicity of the treatment nor the resulting diabetes mellitus was able to affect the morphology of Xenopus hepatocytes significantly. In contrast to the treatments described above, epinephrine clearly affected the ultrastructural picture in hepatocytes from Xenopus males and females. Whereas the liver glycogen content remained unchanged by the treatment, the amount of cell components involved in the protein synthetic machinery was strongly increased.
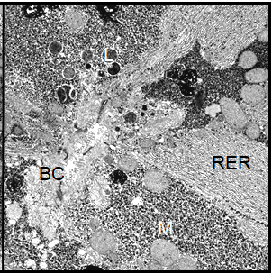
Also the cell nucleus gave the impression of increased activity, since the nuclear chromatin was less densely packed than in the controls; it appeared finely dispersed within the nucleus. In the parenchymal liver cells from cortisol treated toads we found the same characteristic morphological features as in those from the controls, i.e. high glycogen content and a basic level of cellular activity. In male Xenopus toads treated with human chorionic gonadotropin, the morphology of the hepatocytes was dominated by the very high content of glycogen particles. Apart from that, no visible morphological changes had occurred in these cells (Figure 8).
In contrast, we detected multiple dramatic effects of the HCG treatment on the female hepatocytes. The amount of stored glycogen was significantly decreased. The most prominent feature of these cells was the considerable proliferation of the granular endoplasmic reticulum and an increase of all cell components involved in protein synthesis. The cell nucleus showed a perfectly rounded shape and contained dispersed chromatin (Figure 9).
Histochemistry
Glucose-6-Phosphatase (G-6-Pase): In control males and females we observed only a moderate G-6-Pase activity. In the insulin treated toads of both sexes, however, the histochemical reaction was clearly reduced, whereas the glucagon treated animals showed a slight increase in G-6 - Pase activity. In the hepatocytes from toads treated with alloxan, G-6-Pase activity was strongly elevated. We also detected a clearly increased enzyme activity in epinephrine treated Xenopus, as well as in the cortisol treated toads. G-6-Pase activity remained unchanged in HCG treated males (Figure 10), but the hormone treatment apparently had a strongly enhancing effect on the enzyme activity in the female hepatocytes (Figure 11). The treatment with streptozotocin caused no visible differences in the G6Pase activity as compared to the control animals.
Glycogen phosphorylase: The histochemical analysis of the glycogen phosphorylase activity showed that, with the exception of HCG, the enzyme activity was generally much higher in males than in females. However, we detected no significant changes in the histochemical reactions from toads treated either with the pancreatic hormones or with the diabetes inducing substances alloxan and streptozotocin as compared to the controls. The enzyme activity was strongly enhanced in epinephrine treated males but not in the female toads, where the reaction was only slightly elevated. A similar picture was observed in cortisol treated animals, namely a clearly increased glycogen phosphorylase activity level in the male toads, and only a slightly elevated activity in the females. In males treated with HCG, the histochemical reaction was diminished, whereas in the HCG treated females a slightly elevated enzyme activity was detected.

Glyceraldehyde-3-Phosphate dehydrogenase: The
histochemical reaction of glyceraldehyde-3-phosphate dehydrogenase was slightly
stronger in the hepatocytes from control males than in those of the females. In
epinephrine treated males, the enzyme activity was unchanged, but was slightly
elevated in females. In contrast, the reactions were slightly increased in the
hepatocytes from both cortisol treated Xenopus
males and females.
While the hormonal regulation of the entire energy metabolism in mammals is well understood, there are controversial facts known about how hormones influence the carbohydrate metabolism in amphibians. Therefore, it was the main purpose of the present study to analyze the effects of various hormones on the glycogen/glucose balance in Xenopus as an important species of the anurans. Our results gained from the biochemical and histochemical analyses as well as from the ultrastructural examinations clearly showed that major differences indeed exist between the effects in mammals and those in amphibians. While in mammals a strong influence of the pancreatic hormones on carbohydrate metabolism has been described these hormones only weakly affected the glycogen/glucose balance in Xenopus [22,23]. The liver glycogen content was neither significantly augmented by insulin nor reduced by glucagon, and the blood glucose level was significantly affected only in insulin treated females, where the concentration was decreased.
The diabetes inducing substances alloxan and streptozotocin likewise showed no significant changes in the glycogen/glucose balance of the treated animals. It has been shown earlier in our laboratory that larval as well as young postmetamorphic Xenopus toads were influenced by the pancreatic hormones in a manner comparable to the reaction in mammals [10]. Therefore, we assume that the receptors for pancreatic hormones are either deactivated or no longer expressed with the onset of sexual maturity. In contrast to the weak responses to pancreatic hormones, we found strong effects of epinephrine and cortisol on the glycogen/glucose balance in Xenopus.
While the liver and muscle glycogen contents were not affected by the treatments, we detected an increase in the blood glucose concentration as well as elevated blood lipid levels in both sexes. The lacking response of liver and muscle glycogen to epinephrine, cortisol and corticosterone might reflect indeed a non-responsiveness of hepatic carbohydrate metabolism in this species. It might, however, be just as well that it reflects a systemic counterbalance reaction, e.g. through massive gluconeogenesis. In vivo experiments with Xenopus hepatocytes showed a marked response to epinephrine [2]. The same authors did, however, not investigate any of the other hormones used in our study. So one might speculate that the missing systemic regulation leads to a persisting expression of hormone receptors in cultured hepatocytes.
Our histochemical investigations revealed an increased G-6-Pase activity in epinephrine and cortisol treated males and females, an enhanced glycogen phosphorylase activity in males only, and a slightly elevated glyceraldehyde-3-phosphate dehydrogenase activity in females. On the ultrastructural level, the protein synthetic activity was strongly enhanced in epinephrine treated Xenopus toads of both sexes. From the finding that the blood glucose concentrations were elevated in epinephrine and cortisol treated toads without the liver and muscle glycogen contents being affected, the question arises as to where the additional blood glucose came from. By the histochemical reactions described above, we have tested the obvious possibility that gluconeogenesis was occurring.
Our results suggest that the mechanisms responsible for the regulation of the
glycogen/glucose balance differ in Xenopus
males and females. Since the glycogen phosphorylase activity was enhanced in
the males without the glycogen content being reduced, we must assume that the
occurring glycogenolysis is compensated for by simultaneous glycogen synthesis,
and that the liver glycogen turnover rate is generally increased in epinephrine
and cortisol treated males. In the females, we detected a strongly enhanced
G-6-Pase activity which apparently was not correlated to increased
glycogenolysis. In addition, the activity of glyceraldehyde-3-phosphate
dehydrogenase was slightly elevated in epinephrine and cortisol treated female
toads, which is also indicative of enhanced gluconeogenesis. From this data, it
seems reasonable to conclude that gluconeogenesis does occur at least in female
toads upon stimulation with epinephrine and cortisol. Also other authors have
discussed the possibility that conversion of protein and lipids to glucose may
take place in amphibia under the influence of catecholamines and
corticosteroids [1,5,24,25].
It is, however, not yet fully understood how these hormones control gluconeogenesis in amphibia. In Xenopus females treated with HCG, we observed a classic induction of the so called vitellogenic response which resulted in a marked reduction of the liver glycogen stores as well as in a drastically altered ultrastructure of the hepatocytes. It has been shown by several authors that vitellogenin, the egg yolk precursor protein, is synthesized in the hepatocytes, secreted into the bloodstream, and transported to the ovaries during the vitellogenic period [26-28]. The cytological changes observed in our TEM micrographs reflect the enhanced vitellogenin synthesis. In the HCG treated males, we detected neither a decrease in the liver glycogen content nor morphological changes in the hepatic ultrastructure. These findings suggest that the response to HCG described in females may be an indirect effect of this hormone by stimulation of ovarian estrogen secretion. Our suggestion is strongly supported by Nicholls, et al. (1968) who observed that injection of pregnant mare serum gonadotropin caused a vitellogenic response in intact Xenopus females but not in males or ovariectomized females [27].
Since Xenopus tadpoles and juveniles do react to hormonal stimulation the missing response in the adult animal certainly needs further investigation [3,10]. Pure counterbalance of glycogenolysis by gluconeogenesis alone cannot account for the glycogen stability. This would imply a high turnoverrate for glycogen. Glycogen as such does, however, not just disappear by means of dissolution. In mammals, at least, its turnover involves the proliferation of smooth endoplasmic reticulum [29]. So we believe that glycogen in Xenopus hepatocytes is stable on the basis of a deactivation or missing expression of the hormone receptors.
In the attempt to evaluate the results presented in our paper it appears to be
critical to differentiate well between different species of amphibia. Results
obtained from experiments with a member of the Ranidae family for instance
differ in many respects from results obtained from species of the family of
Pipidae like Xenopus, a fact which is
clearly demonstrated by a number of investigations [3,30].
Summary and Conclusions
To summarize the results presented in this paper, we found that the liver glycogen content in Xenopus laevis is very high and is not easily degraded by hormone application. The only situation where glycogen is significantly reduced is in females during HCG stimulated vitellogenesis. Since we assume that this effect is indirect and dependent on the action of estrogen, we suggest that this response might be specific for estrogens. The question of the meaning of this extreme liver glycogen stability arises, and what the importance of glycogen as an energy storage and reserve substance might be. From our data we conclude that in Xenopus laevis, the liver glycogen is apparently not the primary source for the glucose need of several tissues as is the case in mammals. Since the blood glucose levels in epinephrine and cortisol treated toads were elevated without the corresponding liver and muscle glycogen contents being affected, the additional glucose must have come from other sources.
Our histochemical results strongly suggest that gluconeogenesis from protein and/or lipid precursor substrates may be involved in the regulation of the glycogen/glucose balance in Xenopus. We propose that the preservation of high glycogen levels in favor of lipid or protein metabolism must be understood in the context of the amphibian lifestyle as oviparous and poikilothermic animals. The rapid mobilization of glycogen stores in Xenopus females during vitellogenesis apparently takes place in order to provide the carbohydrate supply in the oocytes. We believe that this process is a crucial prerequisite for the survival of this anuran species, since the embryo must live on the deposited energy reserves for a long time period. Another factor playing a role in the unusual glycogen metabolism in Xenopus is the ability of these poikilothermic amphibians to decrease their rate of glucose production under inconvenient conditions. In our opinion, this ability constitutes a fundamental difference between the homeothermic mammals and the poikilothermic amphibians which allows the latter to utilize relatively slow processes such as the transformation of protein and lipid to glucose as primary sources of glucose production. Moreover Xenopus laevis belongs to the group of amphibians which do not hibernate, but passes through a period of aestivation, which may also play a role when comparing different families of amphia with respect to carbohydrate metabolism.
We wish to thank Dr. P. Maly (Institute of Anatomy, Basel) for advice and
assistance with the histochemical assays, and Prof. Dr. L. Du Pasquier (Basel
Institute for Immunology) for kindly providing many of the Xenopus toads used in this study.
1. Hanke W and Neumann U. Carbohydrate metabolism in amphibian (1972) Gen Comp Endocrinol Suppl 3: 198-208.
2. Ade T, Segner H and Hanke W. Hormonal response of primary hepatocytes of the clawed toad, Xenopus laevis (1995) Exp Clin Endocrinol Diabetes 103: 21-27. https://doi.org/10.1055/s-0029-1211325
3. Hanke W. Die hormonale Regulation des Stoffwechsels bei Amphibien (1974) Fortschr Zool 22: 431-455.
4. Gray KM and Janssens PA. Gonadal hormones inhibit the induction of metamorphosis by thyroid hormones in Xenopus laevis tadpoles in vivo, but not in vitro (1990) Gen Comp Endocrinol 77: 202-211. https://doi.org/10.1016/0016-6480(90)90304-5
5. Hanke W and Leist KH. The effect of ACTH and corticosteroids on carbo-hydrate metabolism during the metamorphosis of Xenopus laevis (1971) Gen Comp Endocrinol 16: 137-148. https://doi.org/10.1016/0016-6480(71)90216-4
6. Wong KL and Hanke W. The effects of biogenic amines on carbohydrate metabolism in Xenopus laevis Daudin (1977) Gen Comp Endocrinol 31: 80-90. https://doi.org/10.1016/0016-6480(77)90194-0
7. Spornitz UM. Studies on the liver of Xenopus laevis, 1-The ultrastructure of the parenchymal cell (1975) Anat Embryol 146: 245-264. https://doi.org/10.1007/bf00302173
8. Spornitz UM. Studies on the liver of Xenopus laevis, III-The ultrastructure and the glycogen content of the developing liver (1978) Anat Embryol 154: 1-25. https://doi.org/10.1007/bf00317951
9. Merkle S. Sexual differences as adaptation to the different gender roles in the frog Xenopus laevis Daudin (1989) J Comp Physiol B 159: 473-480. https://doi.org/10.1007/bf00692419
10. Spornitz UM and Morson G. Sex- and age-dependent hepatic glycogen stability in Xenopus laevis (1992) Acta Anat 143: 168.
11. Normal Table of Xenopus laevis (Daudin), Nieuwkoop PD and Faber J (Eds.) (1975) North-Holland, Amsterdam.
12. Roe JH and Dailey RE. Determination of glycogen with the anthrone reagent (1966) Anal Biochem 15: 245-250. https://doi.org/10.1016/0003-2697(66)90028-5
13. Schmidt FH. Enzymatic determination of glucose and fructose simultaneously (1961) Klin Wochenschr 39: 1244.
14. Trinder P. The Effect of Nutritional Lipid Supplementation on Serum Lipid Levels and Effectiveness of Antitubercular Chemotherapy (1969) Ann Clin Biochem 6: 24.
15. Reynolds ES. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy (1963) J Cell Biol 17: 208-213. https://doi.org/10.1083/jcb.17.1.208
16. Chiquoine AD. The distribution of glucose-6-phosphatase in the liver and kidney of the mouse (1953) J Histochem Cytochem 1: 429-435. https://doi.org/10.1177/1.6.429
17. Maly IP and Sasse D. A technical note on the histochemical demonstration of G6Pase activity (1983) Histochemistry 78: 409-411. https://doi.org/10.1007/bf00496628
18. Takeuchi T and Kuriaki H. Histochemical detection of phosphorylase in animal tissues (1955) J Histochem Cytochem 3: 153-160. https://doi.org/10.1177/3.3.153
19. Lindberg LA and Palkama A. The effect of some factors on the histochemical demonstration of liver glycogen phosphorylase activity (1972) J Histochem Cytochem 20: 331-335. https://doi.org/10.1177/20.5.331
20. Henderson B. Quantitative cytochemical measurement of glycer¬aldehyde 3-phosphate dehydrogenase activity (1976) Histochem 48: 191-204.
21. De Schepper GG, Van Noorden CJ and Koperdraad F. A cytochemical method for measuring enzyme activity in individual preovulatory mouse oocytes (1985) J Reprod Fertil 74: 709-716. https://doi.org/10.1530/jrf.0.0740709
22. Eckert R and Randall JD. Tierphysiologie (1993) Georg Thieme Verlag, Deutschland 724.
23. Shafrir E, Bergman M and Felig P. The Endocrine Pancreas: Diabetes Mellitus (2nd Edn) Felig P, Baxter JD, Broadus AE and Frohman LA (Eds.) (1987) Endocrinology and Metabolism, McGraw-Hill Book Company, USA.
24. Janssens PA. Interference of metyrapone with the actions of cortisol in Xenopus laevis Daudin and the laboratory rat (1967) Gen Comp Endocrinol 8: 94-100. https://doi.org/10.1016/0016-6480(67)90117-7 Woof C and Janssens PA. Effects of fasting and cortisol administration on carbohydrate metabolism in Xenopus laevis Daudin (1978) Gen Comp Endocrinol 36: 346-359. https://doi.org/10.1016/0016-6480(78)90116-8
25. Follett BK, Nicholls TJ and Redshaw MR. The vitellogenic response in the South African clawed toad (Xenopus laevis Daudin) (1968) J Cell Physiol 72: 91-102. https://doi.org/10.1002/jcp.1040720408
26. Nicholls TJ, Follett BK and Evennett PJ. The effects of oestrogens and other steroid hormones on the ultrastructure of the liver of Xenopus laevis Daudin (1968) Zeitschrift für Zellforschung 90: 19-27. https://doi.org/10.1007/bf00496699
27. Wallace RA and Dumont JN. The induced synthesis and transport of yolk proteins and their accumulation by the oocyte in Xenopus laevis (1968) J Cell Physiol 72: 73-101. https://doi.org/10.1002/jcp.1040720407
28. Cardell RR. Smooth endoplasmic reticulum in rat hepatocytes during glycogen deposition and depletion (1977) Int Rev Cytol 48: 221-279. https://doi.org/10.1016/s0074-7696(08)61746-5
29. Janssens PA. Hormonal control of glycogenolysis and the mechanism of action of adrenaline in amphibian liver in vitro (1983) Gen Comp Endocrinol 49: 477-484.
Tel: +44 208 457 9080, E-mail: atardrs@yahoo.com
Citation
Atar-Zwillenberg RD, Atar M, Morson G and Spornitz MU. The role of hormones in the regulation of glycogen metabolism in the clawed toad Xenopus Laevis (Daudin) (2019) J Obesity and Diabetes 3: 17-24