Introduction
There is a need to recognize and respect the fact that each person is a unique in this world with its gifted qualities and genetic endowments. Every human-being fortunately or unfortunately needs to be aware of the fact that the present human body shape, size and figure is going to give up one day or another day and what will stay in the world, are the novel deeds, enlightening thoughts, genuine and truthful contributions and distinguished achievements during the life span. Human health and disease are the relative terms which depend on the healthy and balanced up keep against the odd surrounding conditions. When the human populations are subject to dwelling in varied geography, ecology, different environmental conditions, climatic fluctuations and changes, disease susceptibility, persistence of diseases, or bio-social inherent pressures, the natural selection may alter the genetic allele frequencies in one population relative to another [1,2]. Positive natural selection the phenomenon that accounts for the increase in the prevalence of advantageous traits in a population has played an important role in human development and evolution as a species. Large differences in allele frequencies between populations, thus, are the signals in the genome that have undergone selection.
Other signals of recent positive selection include the long haplotypesand reduced allelic variation in the regions around the selected variants. Therefore, the characterization of signatures of positive selection in genes, that are of adaptive significance in human populations, have greater medical relevance for identifying the functionally significant variants that play important roles in health and disease scenario of the host [1,2]. Moreover, different persons may differ in genetic constitution and its response to an infectious disease for example, the malaria. Malaria has been one of the most prevalent and successful parasitic diseases widely spread throughout the globe. Plasmodium parasites, due to multiple factors, have complex biology, high polymorphism, increasingly high resistance to anti-malarial drugs in many endemic regions of the world. As a result of interaction between malaria parasites and human species have led to fixation of several inherited alterations in many populations so that some of the underlying mechanisms confer protection against malaria [1,2]. Natural selection supports such positively involved struggle for existence of the fittest of all.
Cardinal Malaria Situation
Malaria is a parasitic disease highly widespread in tropical and subtropical regions of the world. This disease is most commonly found in poor countries, having less developed systems for health infrastructureand inadequate control and comprehensive preventive strategies. High rates of morbidity and mortality can be attributed to the lack of timely available diagnostic facilities, due to financial constraintsand geographical and transport barriers, access to effective treatment due to insufficient supply of quality medicineand the growing parasite resistance to anti-malarial drugs such as chloroquine, pyrimethamine, etc. The immune response induced in humans by parasitic infection of malaria is a complex one and it varies depending on genetic make-up of the host, age, epidemiological factors, level of malaria endemicity, parasite stage, parasite species, availability of diagnostic facilities and quality of treatmentand repeated parasitic infection. Both innate and acquired immunity processes are invoked vigorously during the infection.
The World Malaria Report 2017 presents a comprehensive state of play in global progress in the fight against malaria up to the end of 2016. In 2016, an estimated 216 million cases of malaria occurred worldwide. Most malaria cases in 2016 were in the World Health Organization (WHO) African Region (90%), followed by the WHO South-East Asia Region (7%) and the WHO Eastern Mediterranean Region (2%) and the like. Plasmodium falciparum is the most prevalent malaria parasite in sub-Saharan Africa, accounting for 99% of estimated malaria cases in 2016. In 2016, there were an estimated 445,000 deaths from malaria globally. About 2 million confirmed malaria cases and 1,000 deaths are reported annually, although 15 million cases and 20,000 deaths are estimated by WHO South East Asia Regional Office. India contributes 77% of the total malaria in Southeast Asia. India was fourth with 7 percent of deaths, after Nigeria (30%), the Democratic Republic of Congo (14%), Burkina Faso (7%) and so on [3]. Malaria is one of the leading causes of death worldwide and genes involved in malaria resistance are the most important for the natural selection in human populations. In 1949, Haldane suggested that infectious disease could be a strong selective force in human populations [4]. Evidence for the strong selective effect of malaria resistance includes the high frequency of a number of detrimental hemato-genetic diseases (including the different hemoglobinopathies, thalassemiasand red cell enzymopathy), caused by the effects of these malaria resistance variants. In view of this pathetic scenario, it is justifiable that population genetics could be useful to determine the amount and pattern of natural selection in human population isolates [5-12].
Human Red Cell Genetic Variants and Plasmodium Falciparum Malaria
Red cell variants that modulate malaria risk can serve as models to identify clinically relevant mechanisms of pathogenesisand thus define parasite and host targets for next-generation therapies. Multiple red cell variants are known to confer protection from malaria. From a biological point-of-view, these insights highlight the co-evolution of host and parasite and serve as a model of balancing selection. From a clinical perspective, these relationships represent a naturally occurring model of protection from severe, life threatening malaria, which can be used to isolate the mechanisms of parasite pathogenesis. By preventing malaria parasites from causing disease, these red cell variants could help discover clinically significant mechanism(s) of pathogenesis and investigating them as targets for future therapeutics.
Thalassemia
JBS Haldane was the first who speculated the Darwinian (Natural) selection that, depending on the genetic make-up, the people would have a different risk of dying when they are confronted by a parasitic organism; so much so that, even if, a gene offering protection against those parasites were, otherwise, harmful, its frequency would increase when a population was exposed to the parasites [4,13]. Later, Haldane hypothesized that one important example could be of thalassemia in the face of malaria for several reasons. First, one type of malaria, caused by Plasmodium falciparum, is highly lethal. Second, it is estimated to have been spread in many parts of the world for several thousands of years, i.e. for several hundreds of generations; thus, malaria as an agent of natural selection seemed to be a better candidate than an infectious disease, causing occasional epidemics, even if associated with high mortality (such as influenza or AIDS). Third, deaths from malaria take place mostly in children, i.e. before reproduction, a critical criterion for effective selection. Last but not the least, Plasmodia take on different forms in the course of their life cycle, but what causes a disease, are the intra-erythrocyte parasites. Therefore, in principle, it is not surprising that, if red cells are in any way abnormal (as they are, for instance, in thalassemia), they may affect the chance of success of the parasites [14].
Similarly, alpha (α+)-thalassemia, being very common in malaria endemic regions, it has been considered to confer protection against clinical disease caused by severe forms of Plasmodium falciparum malaria infection. In the same way, beta (β+)-thalassemia provides protection against the Plasmodium falciparum malaria with significantly lower growth of malaria parasite inside the infected erythrocytes and higher phagocytosis of β-thalassemic infected erythrocytes when compared to normal infected erythrocytes. Moreover, the resistance given to malaria parasite inside the infected erythrocytes is almost identical to that of sickle cell trait infected erythrocytes.
Sickle Cell Anemia
This brief review has been focused on the close and complex relationship of blood disease, e.g. the Sickle Cell Anemia (SCA) with infectious disease of malaria. Sickle cell anemia is a major hemolytic anemia and its epidemiology represents a remarkable signature of the past and present world distribution of Plasmodium falciparum malaria [14]. On one hand, heterozygotes (Hb AS) for the sickle gene are relatively protected against the danger of dying with malaria as now firmly established through a number of clinical field studies reviewed from different parts of Africa, South East Asia, Indian Sub-continentand the Middle East regions [11,15-20]. In addition, the experimental work is consistent with the heterozygote (Hb AS) red cells infected with Plasmodium falciparum are preferentially removed by a mechanism of macrophages [17,19]. On the other hand, patients homozygous for the sickle gene, suffer from sickle cell anemia, are highly susceptible to the lethal effects of malaria [14].
The simplest explanation of this fact is that malaria makes the anemia of homozygous (Hb SS) cells more severe; leading to often hyposplenism, which reduces the clearance of parasites. From public health point of view, it is important that in malaria-endemic countries, the patients with sickle cell anemiaand particularly the children, are being protected from malaria by appropriate prophylaxis [14,21,22]. Since the humans, like most animals, are diploid, therefore, have more options in this respect. Sickle cell anemia is a disease of homozygotes (Hb SS)-thats why it is called recessive disease-whereas, heterozygotes (Hb AS) are normal for most intents and purposes. The first test of Haldanes hypothesis was carried out by Allison when he showed not only that the S gene was frequent in areas of high malaria transmission, but also that AS (Hb) heterozygotes seemed to have less malaria [4,23].
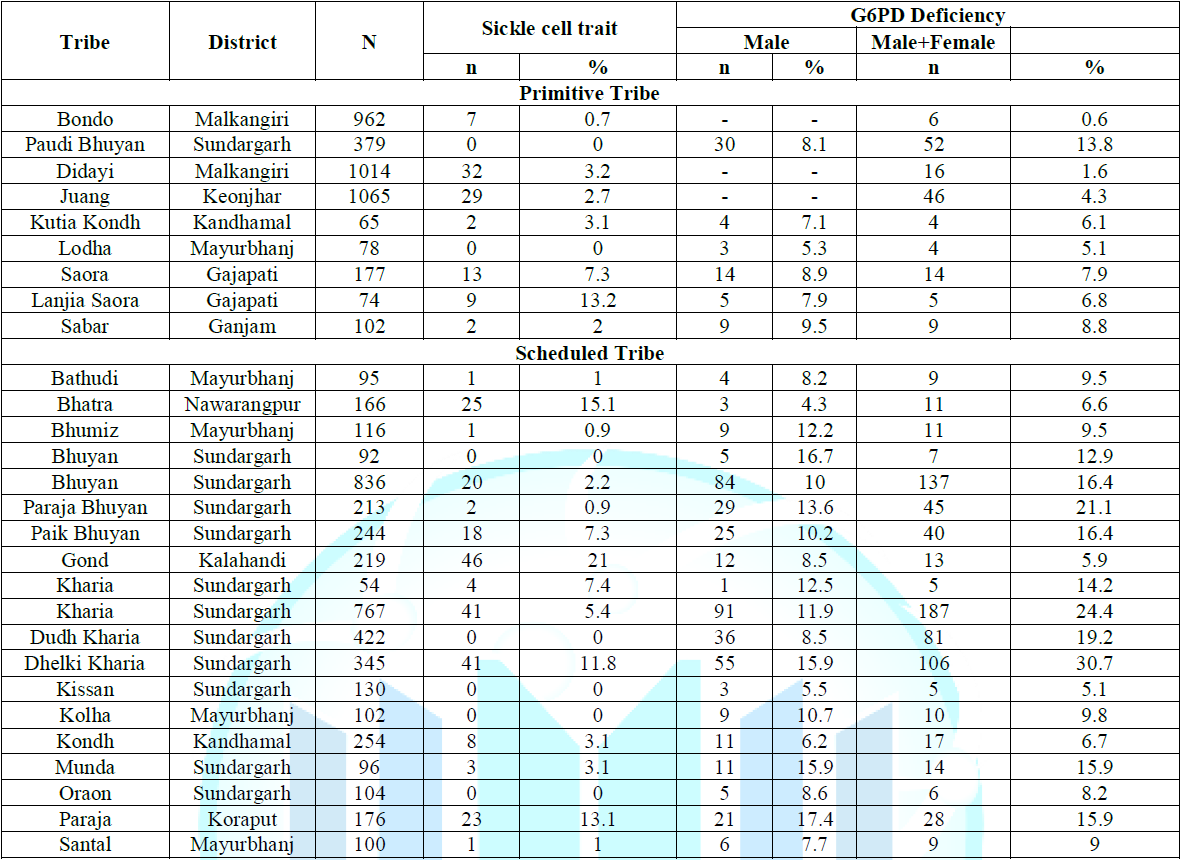
Note: *Data from Reference 2.
By the laws of population genetics it is to be expected that wherever the sickle (S) gene is common, there will be many patients suffering from sickle cell anemia, a severe burden in the population [21,24]. However, in the same population a much larger number of heterozygotes (Hb AS) will have the advantage of being malaria-resistant. The disadvantage of homozygotes (Hb SS) coexisting with the advantage of heterozygotes (Hb AS)-therefore called a balanced [2,16,17]. High fetal and childhood mortality have also recently been described [37,38]. Among the most relevant mechanisms, reduced erythrocyte invasion by the parasite, decreased intra-erythrocyte parasite growth enhanced phagocytosis of parasite-infected erythrocytes and increased immune response against parasite infected erythrocytes have been described [27,31,39,40]. Thus, malaria and sickle cell anemia are still major challenges, being the major public health problems. Patients with sickle cell anemia carry the genetic burden that has helped human populations to survive in malaria-endemic regions of the world. The protective effect of the Hb S gene against malaria is one of the best documented examples in the human species of balanced polymorphism, in which the severe disease of homozygotes (Hb SS or SCA) is balanced by the advantage of Hb AS heterozygotes (Tables 1-4).
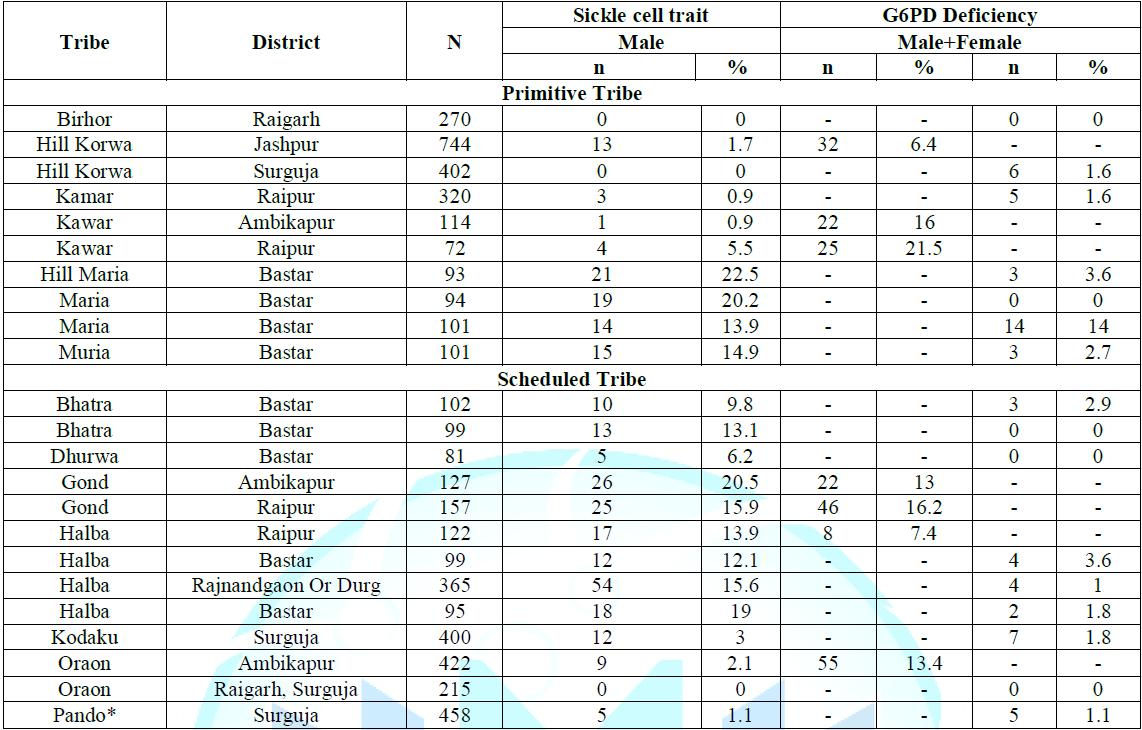
Note: *A case of hemoglobin AE was detected. **Data from Reference 2.
Hemoglobin C Disease
The hemoglobin variants/mutants, namely, Hb C and Hb S, are known to protect carriers from severe falciparum malaria. There is a malaria protection-inducing mechanism, that intra-erythrocyte parasite growth becomes reduced in individuals having Hb C erythrocytes in both mild and severe malarial infection. Individuals homozygous for Hb CC display a reduced risk of having severe or non-severe infection by Plasmodium falciparum malaria [17,41].
Hemoglobin E Disease
Heterozygous form of hemoglobin E confers protection against severe malarial episodes because there is reduced erythrocyte invasion by merozoites, lower intra-erythrocyte parasite growthand enhanced phagocytosis of infected erythrocytes [42-44].
Cytoskeletal Abnormalities
Hereditary spherocytosis (also known as Minkowski–Chauffard syndrome) is an abnormality of erythrocytes. The disorder is caused by mutations in genes relating to membrane proteins that allow for the erythrocytes to change shape. Both Hereditary Ovalocytosis/Ellyptocytosis (Band 3 Variant) and Hereditary Spherocytosis variants reduce the Plasmodium falciparum growth in vitro [45,46]. Ovalocytosis is an uncommon variant of hereditary ellyptocytosis belonging to the erythrocyte membrane inherited disorder. Only the heterozygotic form, which is asymptomatic and endemic in Southeast Asia, derives its name as Southeast Asian ovalocytosis [41]. It gives protection against cerebral malaria.
Glucose-6-Phosphate Dehydrogenase Enzyme Deficiency
The enzyme G6PD deficiency is a genetic disorder that occurs almost exclusively in males. This condition mainly affects red blood cells, which carry oxygen from lungs to various tissues throughout the body, resulting in reduced oxygen flow to the different organs. This can cause fatigue, yellowing of the skin and eyesand shortness of breath. Additional symptoms of G6PD deficiency include:
· rapid heart rate
· shortness of breath
· urine is dark or yellow-orange
· fever
· fatigue
· dizziness
· paleness
· jaundice, or yellowing of the skin and whites of the eyes
The G6PD enzyme deficiency results from mutations in the G6PD gene. The mutation reduces the amount of G6PD enzyme or alters its structure, so that enzyme can no longer play its protective role. As a result, reactive oxygen species can accumulate and damage red blood cells. Factors such as infections, certain drugs, or ingesting fava beans can increase the levels of reactive oxygen species, causing destruction of erythrocytes faster than the body can replace them. A reduction in number of red blood cells causes the signs and symptoms of hemolytic anemia. G6PD enzyme involves in the normal processing of carbohydrates metabolism. It also protects red blood cells from the effects of potentially harmful molecules called reactive oxygen species by products of normal cellular functions.
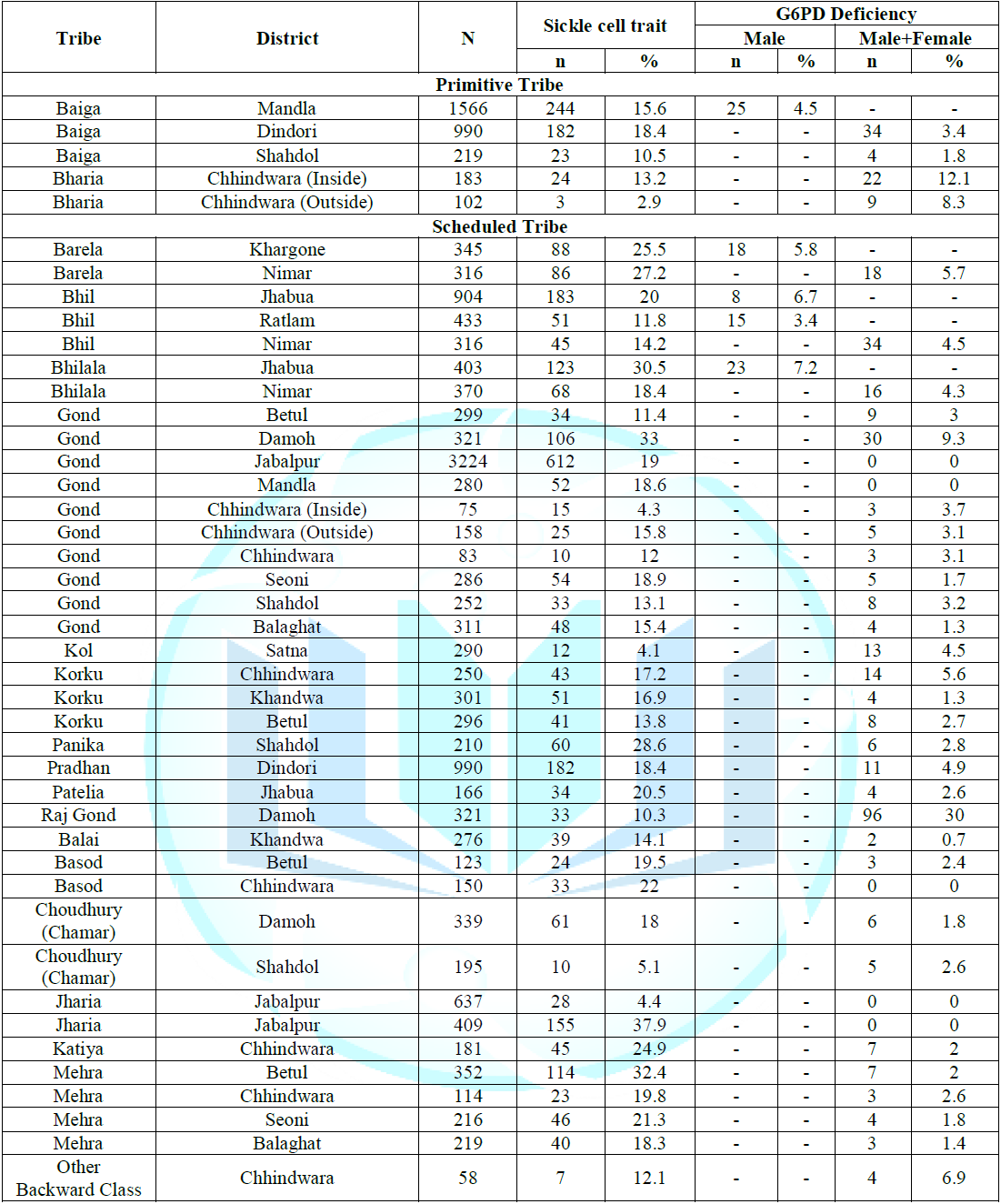
Note: *Data from Reference 2.
Chemical reactions, involving G6PD, produce compounds that prevent reactive oxygen species from building up to toxic levels within red blood cells. In affected individuals, a defect in G6PD enzyme causes red blood cells to break down, called hemolysis, prematurely faster than the body can replace them [30]. Once G6PD deficiency has progressed to hemolytic anemia, more aggressive treatment may be required. This usually includes oxygen therapy and blood transfusion to replenish oxygen and red blood cells. The affected person will need to stay in the hospital, while receiving these treatments as close monitoring required of severe hemolytic anemiaand is critical for ensuring a full recovery without complications. In people with G6PD deficiency, hemolytic anemia is most often triggered by bacterial or viral infections or by certain drugs (such as some antibiotics and medications used to treat malaria).
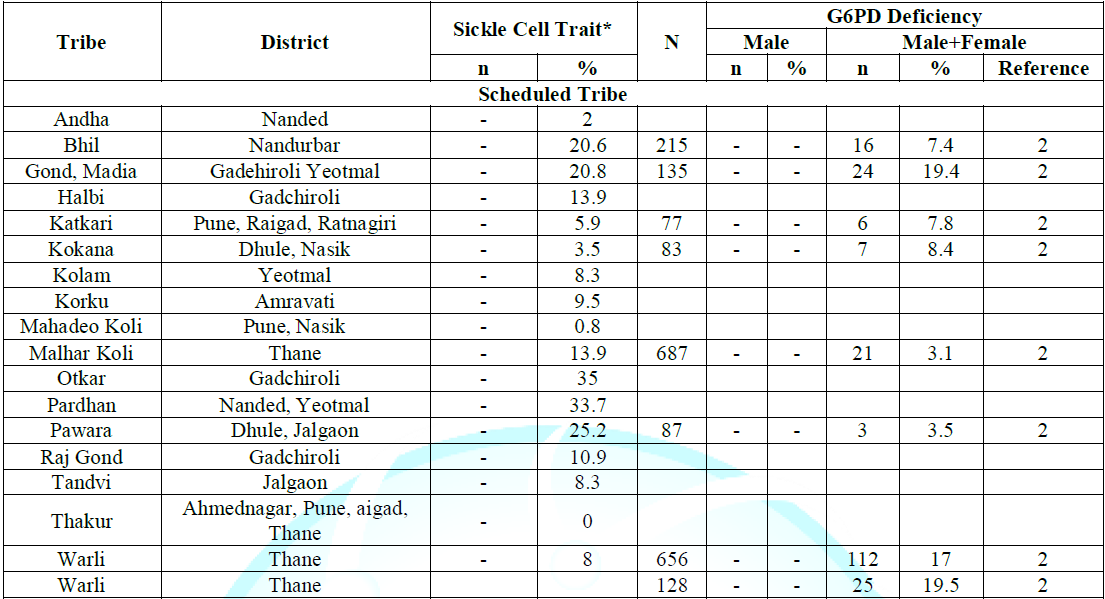
Table 4: Distribution of sickle cell trait and G6PD deficiency in scheduled caste and scheduled tribe communities of Maharashtra, India.
Hemolytic anemia can also occur after eating fava beans or inhaling pollen from fava plants (a reaction called favism) [2,9]. It may also be triggered by infections or by certain drugs such as:
· Antimalarials, a type of medication used to prevent and treat malaria
· Sulfonamides, a medication used for treating various infections
· Aspirin, a drug used for relieving fever, pain and swelling
· Some Non-steroidal Anti-Inflammatory Medications (NSAIDs).
Once the underlying cause is treated or resolved, symptoms of G6PD deficiency usually disappear within a few weeks. G6PD deficiency is also a significant cause of mild to severe jaundice in newborns. Many people with this disorder, however, never experience any signs or symptoms and are unaware that they have the condition.
On the other hand, age specific mortality is high, i.e., as the age advances, the number of G6PD deficiency individuals go on decreasing in a malaria endemic populations, has been reported by some investigators [32]. The G6PD enzyme deficiency is inherited as an X-linked recessive pattern. The gene associated with this enzyme deficiency is located on the X chromosome, which is one of the two sex-chromosomes. In males (who have only one X chromosome, in hemizygous condition), one altered copy of the gene in each cell is sufficient to cause the deficiency.
In females (who have two X chromosomes), a mutation would have to occur in both copies (alleles) of the gene to cause the disease. But, daughters always get their X chromosomes from parents, one each from father and mother. Therefore, they may be heterozygote, if father is affected and mother is normal. Similarly, they may also be heterozygote, if one of the X chromosomes is carrier or trait for G6PD deficiency. Further it all depends on the X chromosome, either inherited from affected father or carrier motherand also on the activation of one (normal or abnormal) out of the mothers two X chromosomes according to the Lyons Hypothesis [9,10]. Thus, G6PD deficiency would have to occur in both X chromosomes (counter parts) of females (from father as well as from mother) to fully express the defective gene (in homozygous state). Males are affected by X-linked recessive disorders much more frequently than the females (Tables 1-4). A characteristic of X-linked inheritance is that fathers cannot pass X-linked traits to their sons [47]. In contrast to the findings by Ruwende et al. a later study showed that a form of G6PD deficiency confers protection against severe malaria in its uniform state (hemizygous males and homozygous females) but not in its mosaic state, i.e. heterozygous females [48]. This finding is consistent with those protection mechanisms involving either enhanced phagocytosis or the effects on pathogenic consequences in the microcirculation of parasitized erythrocytes, since both are expected to operate preferentially on uniformly deficient erythrocytes [26].
Red Cell Genetic Variants and Natural Selection against Malaria
High frequency distribution of inherited hemoglobin disorders including thalassemiasand red cell G6PD enzyme deficiency, which have probably evolved simultaneously in relation to malaria in different vulnerable and malaria endemic populationsand high mortality caused by Plasmodium falciparum malaria in different tropical and subtropical parts of the world confirm that the natural selection is certainly operating against malaria in one way or another; and human population genetics play a major role in this process of co-evolution of human-beings and malaria. The people who have a G6PD deficiency mutation may be partially protected against Plasmodium falciparum malaria. The deficiency of G6PD enzyme or a reduction in the amount of functional G6PD appears to make it more difficult for the malaria parasite to invade red blood cells, inhibits its growth and phagocytises rapidly [49]. G6PD deficiency occurs most frequently in areas of the world where malaria is common. Moreover, disequilibrium of genetic markers such as various variants of hemoglobin and high occurrence of G6PD deficiency is reflected as the natural selection mechanism for protection against malaria [1,2,11,27,31].
Natural selection can maintain deleterious alleles in the population if there is a heterozygote advantage (positive selection) as in the case of sickle cell trait (Hb AS). When the frequency of sickle cell allele decreases in malaria endemic cross-section of the tribal population in India, the frequency of G6PD enzyme deficiency allele increases and vice versa [1,2]. This trend for an inverse relationship between sickle cell disorders and G6PD deficiency due to disequilibrium in major scheduled caste and tribal communities of Central-Eastern India, is fascinating one [1,2]. This medical aspect is important from an evolutionary biological background and could be an excellent starting point for molecular analyses to determine the signature of natural selection in the genomic regions of the β-globin and G6PD genes. This may further provide a mechanism for how natural selection operates against malaria when two mutations occur in the same geographical region [2].
Similarly, further work on the remarkable epistatic interactions between various malaria-protective polymorphisms could provide invaluable information about the mechanisms for the distribution of the different forms of inherited hemoglobin disorders particularly in high-frequency populations (Tables 1-4). Further natural selection had played a major role initially in favor of sickle cell, β -thalassemia and G6PD mutations so that they have probably evolved as a protective mechanism against the lethal effects of malaria. Since the selection favors the mutation with least cost to the population, as the clinical manifestations of G6PD deficiency are mild and do not result in a complete loss of enzyme activity against the sickle cell disease with high morbidity and mortality in the regionand predominant frequency of G6PD deficiency over the sickle cell disorders in some aboriginal communities in India. It seems that the replacement of the sickle cell allele for G6PD deficiency allele is occurring due to disequilibrium in the major scheduled castes/tribes of Chhattisgarh, Madhya Pradesh, Maharashtraand Odisha state in Central India [2]. This means that the decrease in sickle cell allele is compensated by the increase of G6PD deficiency alleles. It seems that different abnormal hemoglobin variants (C,D,E,F,S and thalassemias) and G6PD enzyme deficiency are the directed mutations against the malaria malady as the heterozygotes or carriers of these genetic traits do not suffer severely from the dreadful malaria (Tables 1-4).
Another important factor is the relatively high frequency of consanguineous marriages in many regions of India with high frequency of these red cell genetic variants; this mechanism has an important effect on increasing the gene frequency of the fore said recessively inherited disorders in vulnerable populations of central India [50,51]. Although accurate data on the frequency of consanguineous marriages are lacking, there is no doubt that this is an important factor in helping to maintain the global or regional or local health problem posed by the high frequency of red cell genetic variants and malaria conditionsand significantly contributing towards the high morbidity, maternal mortalityand fetal and childhood mortality [21,22,37,38].
The varying distribution of some of the hemoglobin disordersand G6PD enzyme deficiency reflects strong founder effects of their original inhabitants in different populations [6,8]. Another important factor is the epidemiological conditions, whereby as the public health and nutritional standards improve in the poorer countries, babies with these red cell hemolytic conditions who would, otherwise, have died in early life, are now living long enough to present for diagnosis and management [21]. An estimated 400 million people worldwide have G6PD deficiency. This condition occurs most frequently in certain parts of Africa, Southeast Asia including India, the Mediterraneanand the Middle East. It affects about 1/10 African American males in United States [17,41].
Concluding Comments
The frequency distribution of inherited hemoglobin disorders and Plasmodium falciparum malaria are posing increased burden on human health resources. Their high frequency is a reflection of natural selection combined with a high frequency of consanguineous marriages in many communities and regions, together with an epidemiological expansion due to public health improvement in the affected communities as more babies with these disorders survive to present for treatment in future too. The strongest evidence for Hb S and (α+)- and (β+)-thalassemias, without any doubt, that malaria is responsible for the current distributions of all the major hemoglobin disorders in the world. Malaria is one of the leading causes of death worldwide and has been suggested as the most potent type of selection in humans in recent millennia. As a result, genes involved in malaria resistance are excellent examples of strong selection in recent years. Perhaps best known is the sickle cell hemoglobin variant, which is often used as an example of heterozygote advantage.
In addition, G6PD deficiency illustrates strong selection at an X-linked locus, followed by β-globin variants C, D, E and S variants. In 1949, Haldane initially suggested that infectious disease; specifically the malaria could be a strong selective force in human populations. Evidence for the strong selective effect of malaria resistance includes the high frequency of a number of detrimental genetic diseases caused by the pleiotropic effects of these malaria resistance variants. In contrast, there are many changes that modify levels of expression and provide malaria resistance for G6PD deficiency, α-thalassemiaand β-thalassemia. Malaria parasites have co-evolved with the host and constitute an important deriving evolutionary force behind common erythrocyte variants such as thalassemia, sickle cell disease, Hb C, Hb D, Hb E and G6PD deficiency and other erythrocyte anomalies. Host-parasite interactions have led to a hosts relative resistance to the parasite. There could be two reasons for malaria mediated evolutionary selection:
· Strong selective pressure in case of higher frequency of Hb S allele found in malaria exposed populations; and
· Independent evolutionary responses developed by different populations both at global (e.g. Hb C, Hb D, Hb E and Hb S confer protection against malaria because mutations affect the hemoglobin functionality) and local level (four different HbS haplotypes found in Africa) and the Arab-Indian haplotype is different from the African haplotypes.
Different mechanisms conferring protection against malaria are widely found in different populations of the worldand that the populations have evolved and developed different genetic variants, which are related to resistance to the malaria disease. This could imply that the maintenance of these alleles in the population has been due to the effects of positive selection against the lethal malaria. There also seems to be disequilibrium and competition between two red cell variants, i. e. Sickle cell disease (Hb SS) and G6PD enzyme deficiency. When the frequency of sickle cell allele decreases in malaria endemic tribal population in India, the frequency of G6PD enzyme deficiency allele increases and vice versa.
This trend for an inverse relationship between sickle cell disorders and G6PD deficiency in major scheduled caste and tribal communities of Central-Eastern India, is fascinating one. Since the selection favors the mutation with least cost to the population, as the clinical manifestations of G6PD deficiency are mild and do not result in a complete loss of enzyme activity against the sickle cell disease with high morbidity and mortality in the region. Even though the above could explain, why mutations conferring malaria protection are highly variable and maintained in the population, the association between sickle cell disease and G6PD enzyme deficiency seems to be well-suited here. Thus, the protection is principally present for severe disease and largely absent for Plasmodium falciparum infection, suggesting that hemoglobin disorders specifically neutralize the parasites in vivo mechanisms of pathogenesis.
These genetic traits-including Hemoglobin C (Hb C), Hemoglobin D (Hb D), Hemoglobin E (Hb E), Hemoglobin S (Hb S) and α- and β-thalassemias-are the most common monogenic human disorders and can confer remarkable degrees of protection from severe, life-threatening falciparum malaria in African children: the risk is reduced 70% by homozygous Hb C and 90% by heterozygous Hb S (sickle-cell trait). These hemoglobin variants thus represent a natural experiment to identify the cellular and molecular mechanisms by which Plasmodium falciparum produces clinical morbidity, which remain partially obscured due to the complexity of interactions between this parasite and its human host.
Multiple lines of evidence support a restriction of parasite growth by various hemoglobinopathiesand recent data suggest this phenomenon may result from host micro RNA interference with parasite metabolism. Therefore, owing to the co-evolution of humans and Plasmodium falciparum parasites, the human genome is imprinted with polymorphisms that not only confer innate resistance to falciparum malaria, but also cause hemoglobinopathies to counter the adverse effects of severe malaria.
Acknowledgements
The author gratefully acknowledges all the researchers, investigators and authors whose works have been quoted directly or indirectly in this write up to explain or interpret the findings logically to make it understandable.
References
1. Balgir RS. Do tribal communities show inverse relationship between sickle Cell disorders and glucose-6-phosphate dehydrogenase deficiency in malaria endemic areas of central-eastern India? (2006) Homo J Compar Hum Biol 57: 163-176. https://doi.org/10.1016/j.jchb.2006.01.003
2. Balgir RS. Community expansion and gene geography of sickle cell trait and G6PD deficiencyand natural selection against malaria: experience from tribal land of India (2012) Cardiovasc Hematol Agents Med Chem 10: 3-13. https://doi.org/10.2174/187152512799201190
3. World Malaria Report 2017: WHO Key Points.
4. Haldane JBS. Disease and evolution (1949) Ricerca Sci 119: 68-76.
5. Balgir RS. Serogenetc studies in the Gypsy Sikligars of North western India (1986) Hum Biol 58: 171-187.
6. Balgir RS and Sharma JC. Genetic markers in the Hindu and Muslim Gujjars of North-western India (1988) Am J Phys Anthropol 75: 391-403. https://doi.org/10.1002/ajpa.1330750310
7. Balgir RS, Dash BP and Murmu B. Blood groups, hemoglobinopathy and G-6-PD deficiency investigations among fifteen major scheduled tribes of Orissa, India (2004) Anthropologist 6: 69-75. https://doi.org/10.1080/09720073.2004.11890830
8. Balgir RS. The spectrum of hemoglobin variants in two scheduled tribes of Sundargarh district in North-western Orissa, India (2005) Ann Hum Biol 32: 560-573. https://doi.org/10.1080/03014460500228741
9. Balgir RS. Genetic burden of red cell enzyme glucose-6-phosphate dehydrogenase deficiency in two major scheduled tribes of Sundargarh district in North-western Orissa (2007) Curr Sci 92: 768-774. https://doi.org/10.1080/03014460500228741
10. Balgir RS. Genetic diversity of hemoglobinopathies, G6PD deficiency and ABO and Rhesus blood groups in two isolates of primitive Kharia tribe in Sundargarh district of North-western Orissa, India (2010)J Comm Genet 1: 117-123. https://doi.org/10.1007/s12687-010-0016-y
11. Millimono TS, Loua KM, Rath SL, Relvas L, Bento C, et al. High prevalence of hemoglobin disorders and glucose-6-phosphate dehydrogenase (G6PD) deficiency in the Republic of Guinea (West Africa) (2012)Hemoglobin 36: 25-37. https://doi.org/10.3109/03630269.2011.600491
12. Balgir RS. Spectrum ofhemoglobinopathies andevaluation of prevalence of beta-thalassemia trait in the tribal land of middle India (2013) Intl Public Healt J 5: 165-177.
13. Haldane JBS. The causes of evolution (1932) Cambridge University Press, UK.
14. Luzzatto L. Sickle cell anaemia and malaria (2012) Mediterr J Hematol Infect Dis 4: e2012065. https://doi.org/10.4084/mjhid.2012.065
15. Kwiatkowski DP. How malaria has affected the human genome and what human genetics can teach us about malaria (2005) Am J Hum Genet 77: 171-192. https://doi.org/10.1086/432519
16. Lopez C, Saravia C, Gomez A, Hoebeke J and Patarroyo MA. Mechanisms of genetically- based resistance to malaria (2010) Gene 467: 1-12. https://doi.org/10.1016/j.gene.2010.07.008
17. Taylor SM, Cerami C and Fairhurst RM. Hemoglobinopathies: slicing the Gordian knot of Plasmodium falciparum malaria pathogenesis (2013) Plos Pathog 9: e1003327 https://doi.org/10.1371/journal.ppat.1003327.
18. Hedrick PW. Resistance to malaria in humans: the impact of strong, recent selection (2012) Malaria J 11: 349. https://doi.org/10.1186/1475-2875-11-349
19. Pathak V, Colah R and Ghosh K. Effect of inherited red cell defects on growth of Plasmodium falciparum: an in vitro study (2018) Indian J Med Res 147: 102-109. https://doi.org/10.1016/j.gene.2010.07.008
20. Goldsmith JC, Bonham VL, Joiner CH, Kato GJ, Noonan AS, et al. Framing the research agenda for sickle cell trait: building on the current understanding of clinical events and their potential implications (2012) Am J Hematol 87: 340-346. https://doi.org/10.1002/ajh.22271
21. Balgir RS. Public health challenges of hemoglobinopathies in tribal land of India: a necessity of introducing genetic services in the health care systems approach (2014) Br Biomed Bullet 2: 489-503.
22. Balgir RS. Hematological profile of hemoglobinopathies in maternal health and reproductive outcome in pregnant mothers at a tertiary hospital in central India (2018) J Hematol Multiple Myeloma 3: 1012.
23. Allison AC. Protection afforded by the sickle cell trait against subtertian malarial infection (1954) Br Med J 1: 290-294. http://dx.doi.org/10.1136/bmj.1.4857.290
24. Balgir RS. Prevalence of hemolytic anemia and hemoglobinopathies among the pregnant women attending a tertiary hospital in central India (2015) Thalassemia Reports 5: 16-20. https://doi.org/10.4081/thal.2015.4644
25. Olumese PE, Adeyemo AA, Ademowo OG, Gbadegesin RA, Sodeinde O, et al. The clinical manifestations of cerebral malaria among Nigerian children with the sickle cell trait (1997) Ann Trop Paediat 17: 141-145. https://doi.org/10.1080/02724936.1997.11747877
26. Luzzatto L and Pinching AJ. Commentary to R Nagel -Innate Resistance to Malaria: The Intra-erythrocytic Cycle (1990) Blood Cells 16: 340-347.
27. Ayi K, Turrini F, Piga A and Arese P. Enhanced phagocytosis of ring-parasitized mutant erythrocytes: a common mechanism that may explain protection against falciparum malaria in sickle trait and beta-thalassemia trait (2004) Blood 104: 3364-3371. http://dx.doi.org/10.1182/blood-2003-11-3820
28. Luzzatto L, Nwachuku-Jarrett ES and Reddy S. Increased sickling of parasitised erythrocytes as mechanism of resistance against malaria in the sickle-cell trait (1970) Lancet 1: 319-321. http://dx.doi.org/10.1016/S0140-6736(70)90700-2
29. Luzzatto L. Genetics of red cells and susceptibility to malaria (1979) Blood 54: 961-976.
30. Balgir RS. Ethnic and regional variations in the red cell glucose-6-phosphate dehydrogenase deficiency in India (1989) Indian J Hematol 7: 101-109.
31. Cappadoro M, Giribaldi G, OBrien E, Turrini F, Mannu F, et al. Early phagocytosis of glucose-6-phosphate dehydrogenase (G6PD)-deficient erythrocytes parasitized by Plasmodium falciparum may explain malaria protection in G6PD deficiency (1998) Blood 92: 2527-2534. https://doi.org/10.1182/blood.v92.7.2527.2527_2527_2534
32. Balgir RS, Mishra RK and Murmu B. Clinical and hematological profile of hemoglobinopathies in two tribal communities of Sundargarh district in Orissa, India (2003) Intl J Hum Genet 3: 209-216. https://doi.org/10.1080/09723757.2003.11885854
33. Cabrera G, Cot M, Migot- Nabias F, Kremsner PG, Deloron P, et al. The sickle cell trait is associated with enhanced immunoglobulin G antibody responses to Plasmodium falciparum variant surface antigens (2005) J Infect Dis 191: 1631-1638. https://doi.org/10.1086/429832
34. Cholera R, Brittain NJ, Gillrie MR, Lopera-Mesa TM, Diakite SA, et al. Impaired cytoadherence of Plasmodium falciparum-infected erythrocytes containing sickle hemoglobin (2008) Proc Natl Acad Sci 105: 991-996. http://dx.doi.org/10.1073/pnas.0711401105
35. Adeloye A, Luzzatto L and Edington GM. Severe malarial infection in a patient with sickle-cell anaemia (1971) Br Med J 2: 445-446. http://dx.doi.org/10.1136/bmj.2.5759.445
36. Weatherall DJ. Genetic variation and susceptibility to infection: the red cell and malaria (2008) Br J Haematol 141: 276-286. https://doi.org/10.1111/j.1365-2141.2008.07085.x
37. Balgir RS. Population and public health implications of child health and reproductive outcomes among carrier couples of sickle cell disorders in Madhya Pradesh, India (2014) Intl J MCH AIDS 2: 229-235. https://doi.org/10.21106/ijma.29
38. Balgir RS. Reproductive outcome in carrier couples of β–thalassemia disorders in a tertiary hospital in central India (2014) Thalassemia Reports 4: 10-15. https://doi.org/10.4081/thal.2014.1907
39. Pasvol G, Weatherall DJ and Wilson RJ. Cellular mechanism for the protective effect of haemoglobin S against P. falciparum malaria (1978) Nature 274: 701-703. https://doi.org/10.1038/274701a0
40. Duffy PE and Fried M. Red blood cells that do and red blood cells that dont: how to resist a persistent parasite (2006) Trends Parasitol 22: 99-101. https://doi.org/10.1016/j.pt.2006.01.009
41. Williams TN and Weatherall DJ. World distribution, population geneticsand health burden of the hemoglobinopathies (2012) Cold Spring Harb Perspect Med 2: a011692. https://doi.org/10.1101/cshperspect.a011692
42. Chotivanich K, Udomsangpetch R, Pattanapanyasat K, Chierakul W, Simpson J, et al. Hemoglobin E: A balanced polymorphism protective against high parasitemias and thus severe P falciparum malaria (2002) Blood 100: 1172-1176. https://doi.org/10.1182/blood.v100.4.1172.h81602001172_1172_1176
43. Balgir RS. Red cell genetic markers in malarial susceptibility and selective advantage hypothesis (2013) Online Healt Alli Sci 12: 6.
44. Balgir RS. Is hemoglobin E gene widely spread in the state of Madhya Pradesh in central India? Evidence from five typical families (2014) Mediterr J Hematol Infect Dis 6: e2014060. https://doi.org/10.4084/mjhid.2014.060
45. Genton B, Al-Yaman F, Mgone CS, Alexander N, Paniu MM, et al. Ovalocytosis and cerebral malaria (1995) Nature 378: 564-565. https://doi.org/10.1038/378564a0
46. Gallagher PG. Red cell membrane disorders (2005) Hematol Am Soc Hematol Edu Program 1: 13-18.
47. Ruwende C, Khoo SC, Snow RW, Yates SN, Kwiatkowski D, et al. Natural selection of hemi- and heterozygotes for G6PD deficiency in Africa by resistance to severe malaria (1995) Nature 376: 246-249. https://doi.org/10.1038/376246a0
48. Guindo A, Fairhurst RM, Doumbo OK, Wellems TE and Diallo DA. X-linked G6PD deficiency protects hemizygous males but not heterozygous females against severe malaria (2007) PLoS Med 4: 0516-0522. https://doi.org/10.1371/journal.pmed.0040066
49. Luzzatto L, Usanga EA and Reddy S. Glucose 6-phosphate dehydrogenase deficient red cells: resistance to infection by malarial parasites (1969) Science 164: 839-842. http://dx.doi.org/10.1126/science.164.3881.839
50. Balgir RS. Contribution of marital distance to community inbreeding, homozygosisand reproductive wastage for recessively inherited genetic disorders in Madhya Pradesh, India (2013) Mediterr J Hematol Infect Dis 5: e2013063. https://dx.doi.org/10.4084%2FMJHID.2013.063
51. Balgir RS. Impact of consanguinity and inbreeding on homozygosis of recessively inherited genetic disorders among tribes of central India: The most detrimental and widely practiced evil (2014) Trib Heal Bull 21: 18-24.
*Corresponding author
Balgir RS, Department of Biochemistry, National Institute for Research in Tribal Health (Indian Council of Medical Research), Jabalpur, Madhya Pradesh, India, E-mail: balgirrs@yahoo.co.in
Citation
Balgir RS.Protective resistance by human G6PD enzyme deficiency and hemoglobin variants against malaria and natural selection: further evidence from review of new studies (2019)Edelweiss Appli Sci Tech 3: 44-52.
Keywords
G6PD deficiency, Hemoglobin variants, Thalassemia, Protective resistance, Inverse relationship, Malaria, Natural selection.


 PDF
PDF