Introduction
The
modeling and simulation study is the worst need of ongoing technologies as per
the costly slandered and difficult pattern of experimental era. As by modeling
we can reduce the time and cost with precise calculations of thermodynamic
parameters gradually as calculated by Gibbs for thermodynamics and phase based
equilibria. In 1929 an Elman predicted the magnetic sense of alloys
Fe-Co system through high Curie
temperature and predicted its reportable permeability and saturation most
magnetizations [1,2].
The
thermodynamic parameters of the said iron-cobalt system have been studied well
by those electrical engines based on magnetic properties and materials has
results best prediction of this field as discussed by the software and flexible
tool thermo cal is used for various thermodynamic calculations and predictions
and also helpful by solving and interaction among various elements with many
ideal and non-ideal based behaviors .1n 1981 the first version of this package
was installed and in 2002 their modern and many database version is established
with various thermo dynamical calculation capacity with description in 1985 by Erwin
povoden, et al. [3-8].
Calphad Method
It
is the calculation of phase
diagrams method which is useful for finding thermodynamic parameters of
many materials with prescribed phase diagrams and phase equilibria, it has many
modules for calculations and simulations, recently it has been updated for many
thermodynamic properties of different materials in form of molar volume,
thermodynamic elastic moduli, and phase like properties, their range of
validity is also applicable for processing elements verses composition molar
and molar temperature verses Activity with thermodynamic and optical
properties, all that calculation is done by calculation of phase diagram method
[9-12].
Results and Discussion
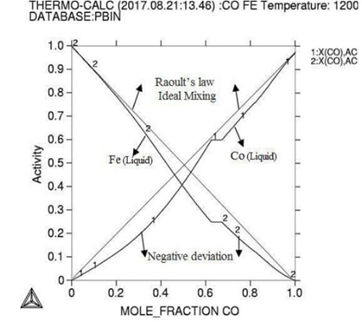
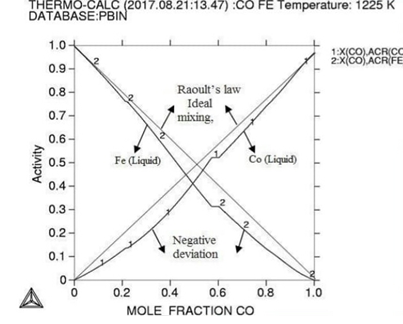
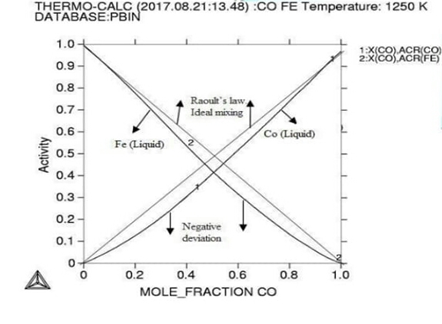
Figure
1:
Molar activity curves at respective 1200K, 1225K, and 1250K temperature.
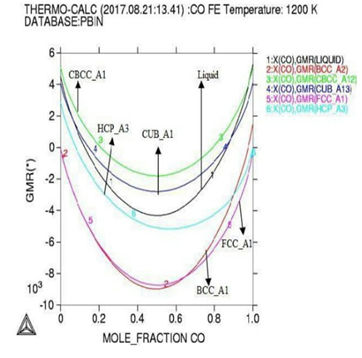
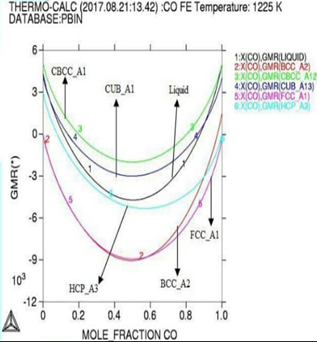
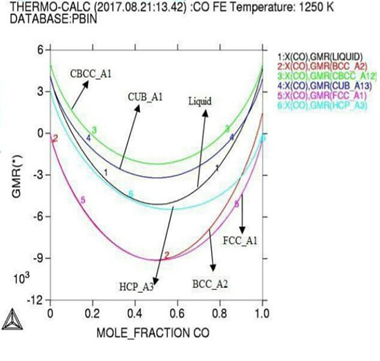
Figure 2: Gibbs energy curves at respective 1200K, 1225K, and 1250K temperature.
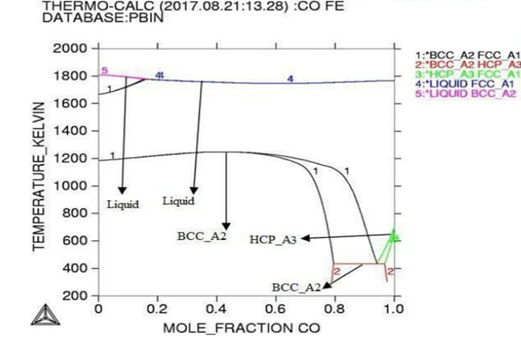
Figure 3: Phase diagram of the Co-Fe binary alloy system.
In
this research paper the thermodynamic calculations with valid predictions
through PBINE database is employed at various temperatures of 1200K, 1225K,
1250K, the activity behaviors is investigated with phase equilibrium and all
module used with lowering occurs in Gibbs
energy curve as shown by (Figures 1
and 2).
Here in calculation, the activity is found increasing as with temperature and all the deviation here is accordance with Roults law ideal curve [13]. As concern with calculated phase diagrams it is in accordance with literatures. Thermodynamic analysis of Co-Fe system is carried out by getting better description for five stable phases founded in Co-Fe binary system. The four among them hcp,fcc,bcc and liquid ate founded the stable phases in Co-Fe system by considering substitution solution phases, the molar Gibbs energies of the particular phases are being calculated by sub-regular solution and substitution model. As their Gibbs energy is analyzed as according to Inden, et al. and modified by k.oikawa, et al. [13-15].
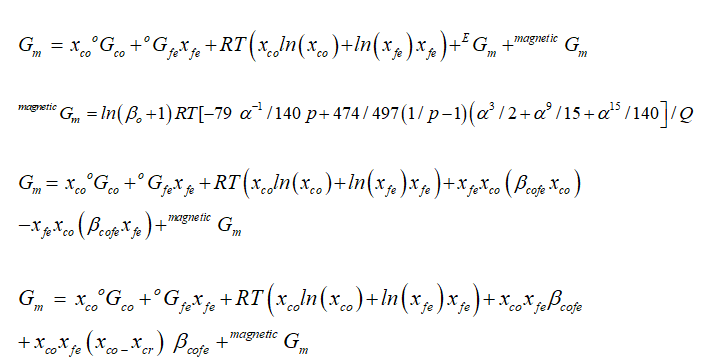
Where in Gibbs energy model, oGcr and oGfe are Fe, Co pure phases in solution, as xi denotes molar fraction of elements in solution, as Fe and Co mole fraction in solution, while the excess energy is shown by constant parameter β ij. Gm is the magnetic part of Gibbs energy. As by temperature dependent section, we have
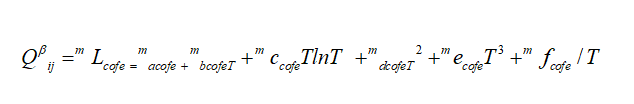
Where mL ij, are the excess binary energies of binary mixing system. While a, b, c, d, e, f are model parameters. The magnetic part of Gibbs energy is given as
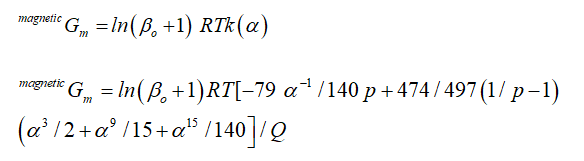
as Q ≤ 1, k(α) is normalized state polynomial of temperature, as by bohr magneton number.
k(α) = -[α-5/10+/497+α-15/315+α-25/1500]/Q
while
Q > 1,
Q = (11692-11692p/15975p)+518/1125
then
k(α) = -[α-5/10+/497+α-15/315+α-25/1500]/(11692-11692p/15975p)+518/1125
As
p is constant, whose value varies for different phases, for bcc is 0.4 and for
other phases is 0.28 [16].
phase are of activity based on highest strong atomic interaction based, FCC_A1=1 phase is found attached with highest temperature that indicated austenite solid solution for Cobalt-Iron system, all the phases calculated here are at different temperatures and molar compositions are in well accordance with data in literature, in between (400-450)K the magnetic properties occurs in alloy as ferromagnetic era at the concentration of (0.85-1.0%), that region is important for magnetic field industry.
As
our results indicating that at highest temperature range of 1250K, and
concentration of (0.4-0.6%) the Gibbs energy goes to its lowest value which
shows that the constituent particle is having strong negative interactions and
the alloy is reporting best stable behaviors at that temperature although the
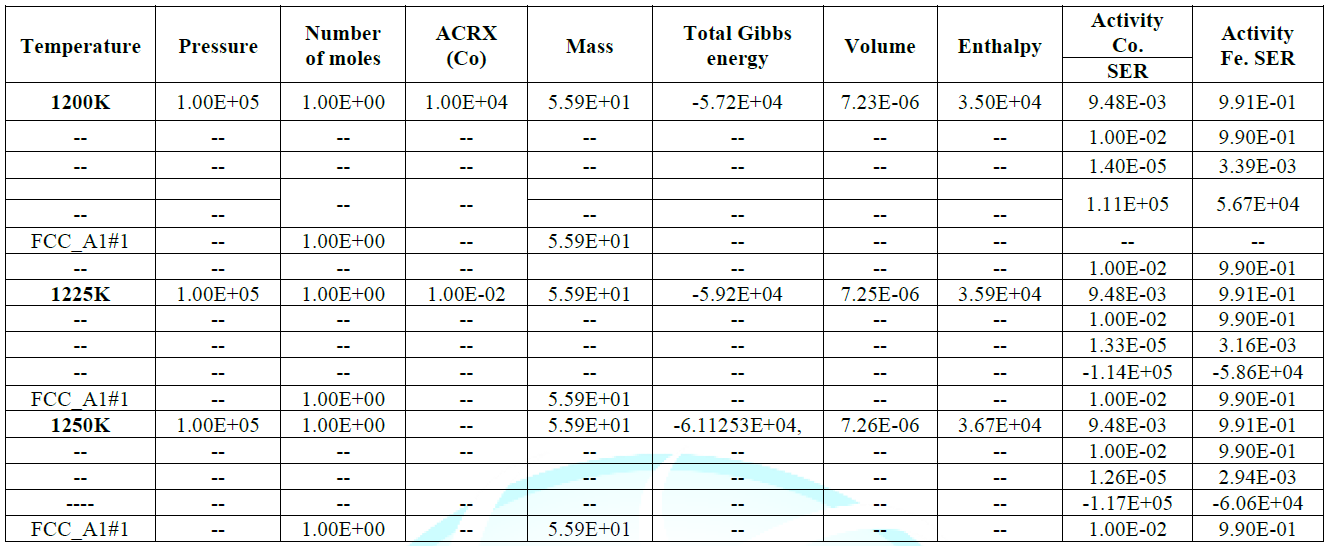
activity value is also at peak as confirmed from table during calculations,
different thermodynamic parameter like volume, enthalpy and entropy verses
temperature is varied here for confine result. At 1250k the smooth negative
deviation is seen from ideality at the range of 0.25-0.65 of molar composition
of alloying elements, the stage indicate here an ideal mixing and solid
solution and well good epitaxial growth of the alloying elements.Then
lattice elements and brillouin zones have no lack matching of alloy elements at
1250k. at range 0.8-1.0 of composition the well good magnetic properties in said
alloy
system are having importance for industrial sectors. The table indicates
frequent increase in the enthalpy
values for increasing heat contents of the alloying elements with
increasing temperature up to 1250k shown highest enthalpy of 3.67311E+04 with
corresponding Gibbs energy of -6.11253E+04, that indicate the vital stability
of the said alloy system at high temperature ranges.
Conclusion
Here
the thermodynamic calculations and predictions of binary alloy Co-Fe system has
been studied using Calphad approach. Thermodynamic analysis involves
calculation of phase diagram, Gibbs energy of mixing, excess Gibbs energies,
thermodynamic molar activities, and coefficient of activities, partial and
integral values of enthalpy for Ag-Cu alloy system at three elevated
temperatures 1200K, 1225K, and 1250K. The alloy reports positive deviation from
Vegards
law and corresponding good negative deviation from Rout’s law ideal Gibbs
curve.
References
- Hertz J. Josiah willard gibbs and teaching, thermodynamics of materials (1992) J Phase Equilibria 13: 450-458. https://doi.org/10.1007/bf02665759
- Dreval LA, Turchanin MA, Agraval PG and Yong Du. Cu-Fe-Co system verification of high temperature phase’s equilibrium and thermodynamic modeling of order phases (2018) Powder Metallurgy and Metal Ceramics 56: 546–555. https://doi.org/10.2320/matertrans.m2013174
- Maruno W, Morizono Y and Tsurekawa S. Differential scanning of ∞/ƒ transformation in Fe-Co alloys under magnetic field (2013) Material Transactions 54: 1823-1828. https://doi.org/10.2320/matertrans.m2013174
- lIkuo
Ohnumaa, Hirotoshi Enoki, Osamu Ikedaa, Ryosuke Kainumaa, Hiroshi Ohtani, et
al. Phase equilibria in co-fe system (2002) Acta Materialia 50: 379-393.
- Roberts
CS. Applications of high temperature magnetic materials (2000) IEEE Transaction
Magnetic 36: 3373-3375.
- Andersson JO, Helander T, Höglund L, Shi P and Sundman BO. Thermocalc and dictra computational tool for materials calculations (2002) Calphad J 26: 273-312. https://doi.org/10.1016/S0364-5916(02)00037-8
- Erwin
Povoden, Nicholas Grundy and Ludwig J. Gauckler. Thermodynamic reassessment of
the Cr-O system in the framework of solid oxide fuel cell research (2006) J
Phase Equilibria and Diffusion 27: 353-362.
- Thuvander
M. Improved thermodynamic modeling in fe-cr alloys system with key experiments
(2011) Calphad J 35: 355.
- Schille
J. New computer program for predicting materials properties (2001) J Phase
Equilibria 22: 463-469.
- Lukas
h, thermodynamic model for crystalline phase’s calculation, 2007, Calphad
journal volume 31, 28-37.
- Charred
P. Application of Calphad method for predicting thermal conductivity (2002)
Calphad J 39: 70-79.
- Ziv D, Ana K and Friedrich B. Thermo-calc in alloys Ti-Al, Al-Fe, Ti-Al (2008) J Mining and Metallurgy 44: 49-61. https://doi.org/10.2298/jmmb0801049k
- Shah
WU, Shah SM, Khan M, Faraz Khan D, Athanasios G, et al. Thermodynamic study of
binary alloys system Co-Cr using Calphad method (2018) J Surface Review and
Letters 25: 1850049. https://doi.org/10.1142/s0218625x1850049x
- Joysurya
Basu and Ranganathan S. Bulk metallic glasses new class of engineering
materials (2003) Springer 28: 783-798. https://doi.org/10.1007/bf02706459
- Iiu ZK. First principles calculations and Calphad modeling of thermodynamics (2009) J Phase Equilibria 30: 517-534.
- Bo Sundman, Qing Chen and Yong Du. A review of Calphad modeling of ordered phases (2018) J Calphad 11: 98.
Corresponding author
Waseem Ullah Shah, Department of physics, University of science and technology Bannu 28100, kpk, Pakistan, E mail: waseemullahshah303@gmail.com
Citation
Shah WU, Khan DF, Burki S, Khan MU and Yin H. Modeling, simulations, predictions, calculations and thermodynamic assessments of cobalt-ferric binary alloys system using calphad method and pbine database (2020) J of Heterocyclics 2: 3-6.
Keywords
(Co-Fe) system, Calphad, Thermo-calc, Thermodynamic
calculations.


 PDF
PDF