
Explore journal overview, editorial leadership, indexing, articles in press, latest published work, and highlights from previous issues.
Research Article :
Anita Aminoshariae, Mohammed Bakkar, Tracey Bonfield, Santosh Ghosh, Thomas A Montagnese, Andre K Mickel Objective: The aim of this study was to investigate the levels of Human Beta Defensin (hBD) 2 and 3, chemokine and cytokine expressions between teeth endodontically diagnosed with symptomatic irreversible pulpitis (SIP), asymptomatic irreversible pulpitis (ASIP) and normal pulps. We hypothesized that there would be a correlation between hBD’s and the immunoregulatory response. Design: Pulpal samples were collected with paper points. Six samples were obtained from normal teeth, 21 from SIP, 18 from ASIP. Levels of cytokines and betadefensins were measured by Luminex technology and ELISA, respectively. Data were statistically analyzed using Kruskal-Wallis, Wilcoxon Mann-Whitney test and Spearman correlation test. Differences were considered significant at p<0.05. Results: hBD-2 levels correlated with samples obtained from patients in the ASIP group, but not in the samples obtained from patients with SIP or the control group. HBD-3 concentrations associated with all of the cytokines and chemokines in both SIP and ASIP groups. However, in the normal group, hBD-3 correlated with only TNFα, IL-8, MCP-1, IL-1β, MIP-1a, RANTES, IL-17 in normal group. When comparing control levels of hBD-2 and hBD-3 with patients samples from either the ASIP or the SIP groups, hBD2 and hBD-3 concentrations were highest in the ASIP group. Conclusions: The hBD-2 and-3 were highly associated with the levels of the chemokines and cytokines in ASIP group. HBD-3 concentrations correlate with the levels of the chemokines and the cytokines in the SIP and ASIP groups. Dental pulp tissue is capable of innate
and adaptive immune
responses caused by various mmunological conditions [1-3]. One host-defense
system, involving the innate immune response upon exposure to the external
environment, is the production of defensins [4]. Human beta-defensins (hBD) are
small cationic antimicrobial
peptides produced by epithelial cells and expressed by all human mucosa [5]
including oral mucosa [6], odontoblasts [7] and pulp cells [8]. The mechanisms of the host immune defense against infections
in human dental pulp (HDP) cells are not completely understood and the role
that hBD play in protection of these cells has yet to be thoroughly explored. Human
beta-defensins have demonstrated immunologic response against grampositive
and -negative bacteria, mycobacteria, fungi, and certain enveloped viruses at
low micromolar concentrations [9,10]. Human beta-defensins have antiretroviral
activity by inhibiting HIV-1 infectivity of immunocompetent cells [11].
Additionally, hBDs can enhance adaptive immunity by acting as adjuvants and
chemoattracting T cells, immature dendritic cells [5], neutrophils [12] and
macrophages [13]. Human beta-defensins-2 is mediated through nuclear factor
kappa-light-chain-enhancer of activated B cells (NF-KB) and
mitogen-activated-protein-kinases (MAPK) pathways [14,15], while hBD-3 is
dependent upon epidermal growth factor receptor (EGFR) activation [16,17]. There are many studies in the medical literature that have
linked hBD-2 and hBD-3 with cytokine and chemokine production [18-21]. Human beta-defensins-2 are highly expressed when the human dental pulp cells are
stimulated with IL-1β or TNFα [22]. Dommisch et al. 2007 [23] reported that
hBD-2 stimulation of odontoblasts
and dental pulp
stem cells led to up-regulation of the IL-6 and IL-8 mRNA. Most of the above mentioned studies were in-vitro
investigations. To date only one clinical dental study investigated the
association between inflamed pulp and hBD’s in HDPs [8]. Since bacteria from
carious lesions elicit inflammatory and immunological responses in the dental
pulp [24], the current authors reasoned that the relative concentrations of
hBDs and inflammatory cytokines might modulate the outcome of pulp pathosis. A
better understanding of pulpal immune response at
different stages of inflammation may allow development of an immune
system-based pulp therapy in the future. To begin testing this hypothesis, the
current authors examined hBDs and cytokine profiles of symptomatic and
asymptomatic irreversible pulpitis in human teeth. To the best of our knowledge, no previously published work
has examined hBDs or cytokine and chemokine profiles involved in endodontic
pulpal pathosis. Thus, the aim of this study was to investigate the levels of
hBD-2 and hBD-3, and chemokine and cytokine expression levels in pulps from
teeth endodontically
diagnosed with symptomatic irreversible pulpitis, asymptomatic irreversible
pulpitis or normal pulps. We hypothesized that there would be a correlation
between hBD’s and the immunoregulatory response in the pulp. This study was approved by the Institutional Review Board
(IRB), Case Western Reserve University, Cleveland, Ohio, and written informed
consent was granted from all patients. Patients undergoing non-surgical root
canal treatment from August 2013 to April 2014 were selected. The investigation
did not alter the treatment plan of any patient. Patients were provided with
information about the purpose of the study and written informed consent was
obtained. Participants had to have met the following criteria: American Society
of Anesthesiologists (ASA) physical status 1 or 2, no history of known
allergies, not pregnant, nonsmokers, age between eighteen and sixty five,
healthy periodontal status and restorable teeth. Exclusion criteria: younger
than eighteen years or older than sixty five, patient on antibiotics, patients
with any known allergies, pregnancy, diabetes, immunocompromised patients, any periodontal probing depth
greater than 5mm or teeth with a furcation or trifurcation involvement, teeth
diagnosed with a necrotic pulp, previously initiated endodontic treatment
and/or previously endodontically treated teeth. Each patient’s pulpal and periradicular status were
evaluated by cold test (HYGIENIC®; ENDO-ICE®; Coltène/Whaledent Inc., Cuyahoga
Falls, OH, USA) and electric pulp test (EPT) (Vitality Scanner; SybronEndo,
Orange, CA, USA) to assess pulp vitality. Percussion, palpation, and periodontal
examinations were performed. Digital periapical and bitewing radiographs of the
tooth in question (Planmeca® ProSensor™; PLANMECA USA, Inc., Roselle, IL, USA)
were obtained. The following type patients were subsequently included:
patients diagnosed with symptomatic irreversible pulpitis (SIP) or asymptomatic
irreversible (ASIP) pulpitis where excavation of caries resulted in pulpal
exposure. The definitions of the SIP and ASIP were described in a previous
study [25]. No traumatized teeth were included in this research. As a negative
control, samples were also taken from six teeth which had no clinical or
radiographic evidence of pulpal and periapical pathosis
but needed routine endodontic treatment for prosthodontic reasons. Briefly, the
clinical characteristics of the cases included subjective and objective
findings which are described SIP: included sharp pain upon thermal stimulus,
lingeringpain (often 30 seconds or longer after stimulus removal). The sampling
procedure is a modified procedure as described by Martinho et al. 2008, 2015
[27,28]. In brief, teeth were mechanically cleaned and disinfected by 0.12%
chlorhexidine (Peridex™; 3M™, USA). After local anesthesia and rubber dam
placement, an access opening was made using a sterile size #2 round carbide bur
(Dentsply Maillefer, Tulsa, OK, USA) in a high speed hand piece to expose the
pulp. An ENDO-Z bur (Dentsply Maillefer, Tulsa, OK) was used to deroof the pulp chamber. Paper
points size 35/0.02 taper (Lexicon ®; DENTSPLY Tulsa Dental Specialties, John
City, TN, USA) were introduced into the pulp chamber and left for 60 seconds.
The procedure was repeated with 4 paper points. The paper points were placed
into Eppendorf tube (Eppendorf Tubes®, Lakewood, OH, USA) containing 400 µL of
phosphate-buffered saline (PBS) (Gibco® PBS pH7.4,Grand Island, NY, USA)
centrifuged at 10000 g at 4°C for 15 minutes and stored at -70°C until use. Bicinchoninic
acid assay was first introduced by Smith et al. [29] and is a sensitive
methodology for protein quantification [30]. Total proteins in the samples were measured using the BCA
protein assay kit (Pierce, Rockford, IL, USA) following the manufacturer’s
instructions. Levels of hBD-2 and
hBD-3 were measured by sandwich enzyme-linked immunosorbent assay (ELISA).
Ninety-six-well immunoplates (R&D, Minneapolis, MN) were coated with 100 μL
goat anti–hBD-2 or rabbit anti–hBD-3 antibodies (Peprotech, NJ) diluted to 1
mg/L in 0.05 mol/L carbonate buffer, pH 9.6, 4°C, for 18 h. Subsequently, the
sample were blocked with 200 μL 1% bovine serum albumin in PBS at room
temperature about 20-25°C] for 10 minutes. After washing three times with 200
μL phosphate buffered saline (PBS), 0.01% Tween 20, 50 μL of test samples + 50
μL of PBS per well were added and incubated at room temperature for 60 min.
Plates were washed three times with PBS, 0.01% Tween 20 and wells incubated at
room temperature with 100 μL biotinylated goat anti-human BD-2 or biotinylated
rabbit anti-human BD-3 (Peprotech, NJ) diluted to 0.2 mg/L in PBS, 0.01% Tween
20 for 30 min. Plates were washed three times with PBS, 0.01% Tween 20 and 100
μL/well streptavidin HRP (R&D, Minneapolis, MN) was added. Plates were then
incubated at room temperature for an additional 30 min, washed three times as
described above, and incubated with 100 μL of Reagent (A+B) (R&D,
Minneapolis) in the dark at room temperature for about 15 min. Reactions were
stopped by adding 50µL of stop solution (R&D, Minneapolis). Absorbance was
measured at 450nm in a microplate reader (Bio-Rad Model 680). Human beta
defensins were quantified by simultaneous ELISA using recombinant hBDs as
calibrators. The Clinical Translational Science Collaborative Bioanalyte
Core utilizes software that allows for the standardization between samples and
between studies longitudinally; all values are evaluated using a standard curve
which has been validated as a comparison of multiple assays to assure
consistency between analyses (found at:
http://casemed.case.edu/ctsc/cores/bioanalyte.cfm). The standard curve for
sample cytokine and chemokine concentration determination was used on the basis
of the standard curve using Bio-Plex Manager 6.1 (Bio-Rad Laboratories,
Hercules, CA). Luminex
analysis (Luminex®100 platform; Austin, TX, USA) of the samples was performed
using the Cytokine and Chemokine Human 10-plex panel multiplex assay (Novex®,
Life technologies™, Grand Island, NY, USA). These included: tumor necrosis
factor- α (TNFα), interleukins (IL-1β, IL-6, IL-8, IL-10, IL-17, IL-17F),
monocyte chemotactic protein-1 (MCP-1; also known as chemokine (C-C motif)
ligand 2 CCL2), macrophage inflammatory protein (MIP-1a, also known as CCL3)
and regulated on activation normal T cell expressed and secreted (RANTES, also
known as CCL5). Levels of the cytokines were normalized with total protein as
measured by BCA assay and expressed as pg/mg of total proteins. To normalize the data the levels of the hBDs, cytokines and chemokines were
expressed as per mg of total proteins The total concentration of protein was
measured in each specimen using a bicinchoninic acid (BCA) assay as per
manufacturer’s protocol (Pierce, Rockford IL). This assay has a reported
dynamic range of 20–20,000 μg/ml and has a 14.7% mean coefficient of variance
for repeat testing across 14 different human and non-human purified protein
targets. Specimens were diluted 1:10 and 1:100 in PBS and run in duplicate.
Colorimetric detection of test specimens was normalized to background specimens
that contain extraction buffer only. Total protein concentration
was estimated using an 8-point standard curve and is expressed as μg/ml. The
ratio of immune marker concentration to total protein concentration was then
calculated and expressed as [pg of immune marker]/[mg of total protein]. sample size calculation was performed before the beginning
of the study using the SAS Power and Sample Size 3.1 of SAS (Statistical
Analysis System) software for Windows version 9.1.3 (SAS Institute Inc. Cary,
NC, USA). Expecting the minimum correlation of 0.60 with power of 0.80 and
alpha of 0.05 and one sided test, the minimum sample size for the experimental
cases was 15 root canals. Demographic characteristics were expressed as the means and
standard deviations and Chi-square
test. Results of hBDs, chemokines and cytokines were normalized by the
equation: (cytokine or chemokine or HBD expressed) / total protein (BCA assay).
Data were statistically analyzed using Kruskal-Wallis, Wilcoxon Mann-Whitney
test and Spearman correlation test. The level of statistical significance was
set at 95% confidence interval (p < 0.05), and the statistical analysis was
calculated using Prism 6.0 software (GraphPad Prism version 6.0 for Windows,
San Diego, CA, USA). Table 1 details the demographic distribution and the mean
age. To determine whether the variables were statistically independent we
performed Pearson Chi
Square test and the result found not to be significant (χ2 =1.534,
p=0.464). Table 1: Demographic characteristics and endodontic diagnosis of the healthy patients. Table 2 details the mean, median and standard deviation of
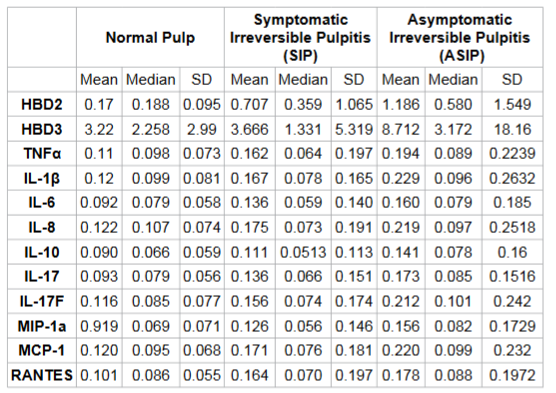
levels of hBD’s, cytokines and chemokines in normal, SIP and ASIP groups. Table 3 details the
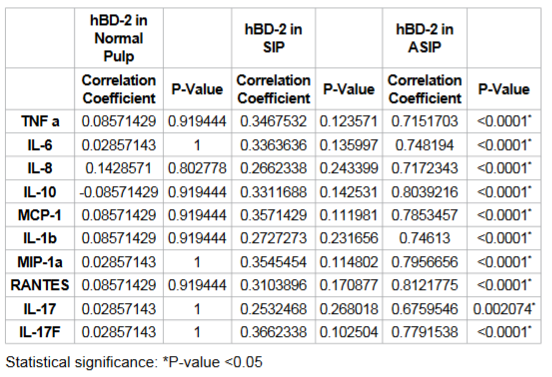
analysis between levels of normalized hBD-2 in comparison to chemokines,
cytokines with statistical analysis. To normalize the data the levels of the
hBDs, cytokines and chemokines were expressed as per mg of total proteins.
There was no correlation between the levels of hBD-2 in comparison to the
cytokines and chemokines in the normal and SIP groups; however, in the ASIP
group there was a correlation between the levels of hBD-2 and TNFα, IL-6, IL-8,
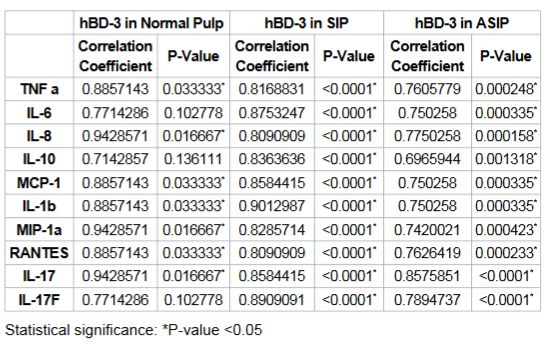
IL-10, MCP-1, IL-1β, MIP1a, RANTES, IL-17 and IL-17F. Table 4 details the levels of normalized hBD-3 in comparison
to chemokines, cytokines with statistical analysis. In the SIP and ASIP groups
there was a correlation with TNFα, IL-6, IL-8, IL10, MCP-1, IL-1β, MIP-1a,
RANTES, IL-17 and IL-17F (all the cytokines, chemokines studied). For all
groups, there was a correlation with the levels of hBD-3 to TNFα, IL-8, MCP-1,
IL-1β, MIP-1a, RANTES and IL-17. To the best of our
knowledge, this is the first endodontic clinical study that investigated the
role of hBDs in relation to pulpal cytokines and chemokines. Since there have
been many medical studies which have linked hBD-2 and hBD-3 with cytokine and
chemokine production suggesting these hBDs link innate and adaptive immunity
[5,31,32], we focused on correlating hBD levels with the cytokine/chemokine
panel used in this current investigation. Results supported the hypothesis that
there is a correlation between hBD’s and the immunoregulatory response. At this stage, it is not clear why the levels of hBD-2
correlated only in asymptomatic, but not in symptomatic, irreversible pulpitis
compared to the normal pulp (Table 3). In contrast, the levels of hBD-3
correlated in both ASIP and SIP (Table 4). These observations imply that there
are differences between SIP and ASIP and the involvement of hBD’s and
cytokines, chemokines. Studies are on-going in our laboratory to explore why
SIP and ASIP may be different and the potential contribution of alternative
inflammatory mediators such as neuropeptides and microbial differences between
the two cases (SIP and ASIP groups). The results of this study agree with Dommisch et al. who
reported, in their in-vitro study, that hBD-2 stimulated the gene expression of
pro-inflammatory cytokines [23]. Our findings also agree with Kim et al. [22]
who reported that there was a correlation between TNF-α and hBD-2. The reason
why, in the SIP group there was a correlation between the hBD-3 and cytokines,
chemokines (Table 4) but not hBD-2 levels with the cytokines and the
chemokines, needs further investigation (Table 3). In both SIP and ASIP groups
(Table 4) there was a significant correlation between hBD-3 and all the
chemokine and cytokines. Since the levels of hBD-3 were correlated in SIP and
ASIP groups, this again might suggest that perhaps hBD-3 may play an even more
extensive role in immunoregulation than previously reported in the endodontic
literature. There are many speculations and possibilities as to why an
inflamed pulp might be symptomatic or asymptomatic. These possibilities include
the presence of endotoxins [33], the immunoregulary response [34-36], and
pathogens of various microbial progression [24,37-39]. The latter describes how
various microbes elicit different immunologic responses. In the periodontal
literature, hBD-3 was reported to bind to a strain of Porphyromonas gingivalis
and attenuated a proinflammatory response [40]. The correlation between hBD-3
and the microbe was significantly higher than hBD1 or hBD-2 resulting in
significant attenuation of the interleukin (IL)-6, IL-10, granulocyte
macrophage colony stimulating factor (GM-CSF) and tumornecrosis factor-a
(TNF-α). Other similar studies [14,40 41] suggest that different microbial
pathogens elicit different immunological responses. In future studies,
differences between microbial pathogens in symptomatic and asymptomatic
irreversible pulpitis should be better explored. We do agree that the sample size for our normal group was smaller
compared to the other two groups (SIP and ASIP). We would have preferred to
have had a larger sample size for normal subjects, but it is not easy to get
samples from teeth with normal pulps. However, the previous studies [8,42,43]
had a similar sample size as ours for their control group and based on these
previous studies we performed our power calculation of the study as detailed in
the Methodology Section. We used paper points
for our clinical sampling technique. Previous clinical studies (excluding
extraction of teeth) have used paper points [27,28,44-50], cotton pellets
[34,51], barbed broach [43] to measure cytokines, chemokines, neuropeptides and
exotoxins in the root canal system. Currently there are no “gold standard
techniques” for sampling pulpal tissue, and to date, the current
molecular-based methods are still under continuous improvement [52]. Future studies evaluating microbial differences in root
canals and the concomitant host responses could provide an interesting
contribution to the understanding of the host-pathogen relationship. Human beta defensin-2
and hBD-3 were associated with the cytokines and chemokines in ASIP group.
HBD-3 concentrations correlated with the levels of the chemokine and the
cytokines in the SIP and ASIP groups. Acknowledgement The authors deny any conflicts of interest. 1. Hahn CL, Liewehr FR. Update on the adaptive immune
responses of the dental pulp. (2007) J
Endod 33: 773-781. 2. Hahn CL, Liewehr FR. Innate immune responses of the dental
pulp to caries. (2007) J Endod 33:
643-651. 3. Hahn CL, Liewehr FR. Relationships between caries
bacteria, host responses, and clinical signs and symptoms of pulpitis.
(2007) J Endod 33: 213-219. 4. Ganz T1. Defensins: antimicrobial peptides of innate
immunity. (2003) Nat Rev Immunol 3:
710-720. 5. Yang D, Chertov O, Bykovskaia S, Chen Q, Buffo M, et al.
ß-Defensins: linking innate and adaptive immunity through dendritic and T cell
CCR6. (1999) Science 286:525-528. 6. Dunsche A, Açil Y, Dommisch H, Siebert R, Schröder JM, et
al.. The novel human beta-defensin-3 is widely expressed in oral tissues.
(2002) Eur J Oral Sci 110: 121-124. 7. Shiba H, Mouri Y, Komatsuzawa H, Ouhara K, Takeda K, et
al.. Macrophage inflammatory protein-3alpha and beta-defensin-2 stimulate
dentin sialophosphoprotein gene expression in human pulp cells. (2003) Biochem Biophys Res Commun 306: 867-871. 8. Paris S, Wolgin M, Kielbassa AM, Pries A, Zakrzewicz A.
Gene expression of human beta-defensins in healthy and inflamed human dental
pulps. (2009) J Endod 35: 520-523. 9. Ghosh SK, Gerken TA, Schneider KM, Feng Z, McCormick TS,
et al. Quantification of human ß-defensin-2 and-3 in body fluids: application
for studies of innate immunity. (2007) Clin Chem 53:757-765. 10. Yadava P, Zhang C, Sun J, Hughes JA. Antimicrobial
activities of human ß-defensins against Bacillus species. (2006) International
Journal of antimicrobial agents 28:132-137. 11. Feng Z, Dubyak GR, Lederman MM, Weinberg A. Cutting
edge: human beta defensin 3--a novel antagonist of the HIV-1 coreceptor CXCR4.
(2006) J Immunol 177: 782-786. 12. Ganz T, Selsted ME, Szklarek D, Harwig SS, Daher K, et
al.. Defensins. Natural peptide antibiotics of human neutrophils. (1985) J Clin Invest 76: 1427-1435. 13. Soruri A, Grigat J, Forssmann U, Riggert J, Zwirner J.
beta-Defensins chemoattract macrophages and mast cells but not lymphocytes and
dendritic cells: CCR6 is not involved. (2007)
Eur J Immunol 37: 2474-2486. 14. Kota S, Sabbah A, Harnack R, Xiang Y, Meng X, et al.
Role of human ß-defensin-2 during tumor necrosis factor-a/NF-?B-mediated innate
antiviral response against human respiratory syncytial virus. (2008) J Biol
Chem 283: 22417-22429. 15. Krisanaprakornkit S, Kimball JR, Dale BA. Regulation of
human ß-defensin-2 in gingival epithelial cells: the involvement of
mitogen-activated protein kinase pathways, but not the NF-?B transcription
factor family. (2002) The Journal of Immunology168: 316-324. 16. Boughan PK, Argent RH, Body-Malapel M, Park J-H, Ewings
KE, et al. Nucleotide-binding Oligomerization Domain-1 and Epidermal Growth
Factor Receptor critical regulators of ß-defensins during helicobacter pylori
infection.(2006) J Biol Chem 281: 11637-11648. 17. Sørensen OE, Thapa DR, Rosenthal A, Liu L, Roberts AA,
et al. Differential regulation of ß-defensin expression in human skin by
microbial stimuli. (2005) J Immunol. 174: 4870-4879. 18. Tiriveedhi V, Banan B, Deepti S, Nataraju A, Hachem R,
et al.. Role of defensins in the pathogenesis of chronic lung allograft
rejection. (2014) Hum Immunol 75:
370-377. 19. Guo B, Xie G, Duan Z, Xia L. The effects of recombinant
human betadefensin-3 on expression of interleukin-17A and interleukin-22 in
BEAS-2B cell. (2013) Chinese journal of experimental and clinical virology 27:
260-262. 20. Kanda N, Kamata M, Tada Y, Ishikawa T, Sato S, et al..
Human β-defensin-2 enhances IFN-γ and IL-10 production and suppresses IL-17
production in T cells. (2011) J Leukoc
Biol 89: 935-944. 21. Petrov V,
Funderburg N, Weinberg A, Sieg S. Human ß defensin-3 induces chemokines from
monocytes and macrophages: diminished activity in cells from HIV-infected
persons. (2013) Immunology 140: 413-420. 22. Kim YS, Min KS, Lee SI, Shin SJ, Shin KS, et al. Effect
of proinflammatory cytokines on the expression and regulation of human
beta-defensin 2 in human dental pulp cells. (2010) J Endod 36: 64-69. 23. Dommisch H, Winter J, Willebrand C, Eberhard J, Jepsen
S. Immune regulatory functions of human beta-defensin-2 in odontoblast-like
cells. (2007) Int Endod J 40: 300-307. 24. Hahn CL, Liewehr FR. Relationships between caries
bacteria, host responses, and clinical signs and symptoms of pulpitis.
(2007) J Endod 33: 213-219. 25. Levin LG, Law AS, Holland GR, Abbott PV, Roda RS.
Identify and define all diagnostic terms for pulpal health and disease states.
(2009) J Endod 35: 1645-1657. 26. AAE. Glossary of Endodontic Terms. (2011) [cited
071315]. 27. Martinho FC, Gomes BP. Quantification of endotoxins and
cultivable bacteria in root canal infection before and after chemomechanical
preparation with 2.5% sodium hypochlorite. (2008) J Endod 34: 268-272. 28. Martinho FC, Nascimento GG, Leite FR, Gomes AP, Freitas
LF, et al. Clinical Influence of Different Intracanal Medications on Th1-type
and Th2type Cytokine Responses in Apical Periodontitis. (2015) J Endod
41:169-175. 29. Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH,
et al. Measurement of protein using bicinchoninic acid. (1985) Anal Biochem 150: 76-85. 30. Walker JM. The bicinchoninic acid (BCA) assay for
protein quantitation. In: The Protein Protocols Handbook. Springer. (2009)
Pp:11-15. 31. Klüver E, Schulz-Maronde S, Scheid S, Meyer B, Forssmann
WG, et al. Structure-activity relation of human ß-defensin 3: influence of
disulfide bonds and cysteine substitution on antimicrobial activity and
cytotoxicity(2005) Biochemistry 44: 9804-9816. 32. Funderburg N, Lederman MM, Feng Z, Drage MG, Jadlowsky
J, et al. Human ß-defensin-3 activates professional antigen-presenting cells
via Toll-like receptors 1 and 2. (2007) Proc Natl Acad Sci U S A104:1863118635.
33. Khabbaz MG, Anastasiadis PL, Sykaras SN. Determination
of endotoxins in caries: association with pulpal pain. (2000) Int Endod J 33: 132-137. 34. Elsalhy M, Azizieh F, Raghupathy R. Cytokines as
diagnostic markers of pulpal inflammation. (2013) Int Endod J 46: 573-580. 35. Cohen JS, Reader A, Fertel R, Beck M, Meyers WJ. A
radioimmunoassay determination of the concentrations of prostaglandins E2 and
F2alpha in painful and asymptomatic human dental pulps. (1985) J Endod 11: 330-335. 36. Byers MR. Dynamic plasticity of dental sensory nerve
structure and cytochemistry. (1994) Arch
Oral Biol 39 Suppl: 13S-21S. 37. Hahn CL, Best AM, Tew JG. Comparison of type 1 and type
2 cytokine production by mononuclear cells cultured with streptococcus mutans
and selected other caries bacteria. (2004)
J Endod 30: 333-338. 38. Gomes BP, Drucker DB, Lilley JD. Associations of
specific bacteria with some endodontic signs and symptoms. (1994) Int Endod J 27: 291-298. 39. Jacinto R, Gomes B, Ferraz C, Zaia A, FJ Filho S.
Microbiological analysis of infected root canals from symptomatic and
asymptomatic teeth with periapical periodontitis and the antimicrobial
susceptibility of some isolated anaerobic bacteria. (2003) Oral Microbiol
Immunol 18:285-292. 40. Pingel LC, Kohlgraf KG, Hansen CJ, Eastman CG, Dietrich
DE, et al. Human ß-defensin 3 binds to hemagglutinin B (rHagB), a non-fimbrial
adhesin from Porphyromonas gingivalis, and attenuates a pro-inflammatory
cytokine response. (2008) Immunol Cell Biol.
86: 643-649. 41. Varoga D, Tohidnezhad M, Paulsen F, Wruck CJ,
Brandenburg L, et al.. The role of human beta-defensin-2 in bone. (2008) J Anat 213: 749-757. 42. Abd-Elmeguid A, Abdeldayem M, Kline LW, Moqbel R,
Vliagoftis H, et al.. Osteocalcin expression in pulp inflammation. (2013) J Endod 39: 865-872. 43. Kokkas A, Goulas A, Varsamidis K, Mirtsou V, Tziafas D.
Irreversible but not reversible pulpitis is associated with up-regulation of
tumour necrosis factoralpha gene expression in human pulp. (2007) Int Endod J
40:198-203. 44. Safavi KE, Rossomando EF. Tumor necrosis factor identified
in periapical tissue exudates of teeth with apical periodontitis. (1991) J Endod 17: 12-14. 45. Martinho FC,
Chiesa WM, Leite FR, Cirelli JA, Gomes BP. Correlation between
clinical/radiographic features and inflammatory cytokine networks produced by macrophages
stimulated with endodontic content. (2012) J Endod 38:740-745. 46. Martinho FC, Chiesa WMM, Leite FR, Cirelli JA, Gomes BP.
Antigenic Activity of Bacterial Endodontic Contents from Primary Root Canal
Infection with Periapical Lesions against Macrophage in the Release of
Interleukin-1ß and Tumor Necrosis Factor alpha. (2010) J Endod 36:1467-1474. 47. Martinho FC, Chiesa WMM, Leite FR, Cirelli JA, Gomes BP.
Antigenicity of Primary Endodontic Infection against Macrophages by the Levels
of PGE 2 Production. (2011) J Endod 37: 602-607. 48. Martinho FC, Chiesa WM, Zaia AA, Ferraz CC, Almeida JF,
et al.. Comparison of endotoxin levels in previous studies on primary
endodontic infections. (2011) J Endod
37: 163-167. 49. Sousa EL, Martinho FC, Leite FR, Nascimento GG, Gomes
BP. Macrophage cell activation with acute apical abscess contents determined by
interleukin-1 Beta and tumor necrosis factor alpha production. (2014) J Endod 40: 1752-1757. 50. Yoo YJ, Shon WJ, Baek SH, Kang MK, Kim HC, et al..
Effect of 1440-nanometer neodymium:yttrium-aluminum-garnet laser irradiation on
pain and neuropeptide reduction: a randomized prospective clinical trial.
(2014) J Endod 40: 28-32. 51. Nakanishi T, Matsuo T, Ebisu S. Quantitative analysis of
immunoglobulins and inflammatory factors in human pulpal blood from exposed
pulps. (1995) J Endod 21: 131-136. 52. Sathorn C, Parashos P, Messer HH. How useful is root
canal culturing in predicting treatment outcome? (2007) J Endod 33: 220-225.Associations between HBD3 and Pro-Inflammatory Cytokines in Asymptomatic Irreversible Pulpitis
Abstract
Full-Text
Introduction
Materials and Methods
Patient selection
ASIP: these cases had no clinical
symptoms and usually respond normally to thermal testing deep caries resulted
in exposure following removal [26].
Control: normal pulp, where the teeth were symptom-free, healthy and free of
caries.
Operative procedure & site selectionBicinchoninic acid assay (BCA assay)
Enzyme-link immunosorbent assay (ELISA)
Cytokine and Chemokine measurement by Luminex
Normalization Methods
Total protein
Sample calculation
Statistical analysis
Results

Discussion
Conclusions
References
Keywords