
Explore journal overview, editorial leadership, indexing, articles in press, latest published work, and highlights from previous issues.
Commentary :
This article introduces the characteristics of postprandial Very Low Density Lipoprotein (VLDL) remnants (remnant lipoproteins; RLP) in plasma which significantly increased after fat load as a major component of increased Triglycerides (TG) and involved in obesity and insulin resistance. It has been long believed that postprandial RLP, mainly Chylomicron (CM) remnants, increases as the result of disturbed lipoprotein lipase (LPL) activity caused by insulin resistance, etc. However, based on this report, we recently proposed that elevated postprandial VLDL remnants produced by food intake, such as excessive fat and fructose, cause obesity and insulin resistance when exposed continuously [1]. VLDL remnants, but not CM remnants, is the key word of this article and VLDL remnants play a definitive role as a bridge between food intake and its metabolism. Here, we have explained the bridging role of VLDL remnants between the habit of food intake and its metabolism in body. Following 6 aspects between fat-rich meal intake and the increase of plasma postprandial TG and RLP are explained. (1) Why TG and RLP increase after food intake? (2) Which lipoproteins increase most after food intake? (3) What percentage of increased TG after food is comprised of RLP-TG? (4) How the increased TG is metabolized by LPL? (5) The increase of postprandial RLP is the result of obesity and insulin resistance or cause of obesity and insulin resistance? (6)Why postprandial TG is a risk of cardiovascular diseases?
Dietary fat provides as much as 30% to 40% of total daily caloric intake in the western diet and TG constitutes the majority of that fat. Dietary Long-Chain Triglycerides (LCTs), the most common dietary lipid structures, are mainly digested into two fatty acids and an SN-2 monoglyceride molecule [2]. Re-esterification of fatty acids occurs in the enterocytes of the small intestine. Subsequently, the resulting LCTs are incorporated into CM particles and released into the blood through the lymphatic system after food intake. Therefore, plasma TG concentration increases significantly after food intake, especially after fat-rich meal with LCTs [3]. But certain fatty acids such as Medium Chain Triglycerides (MCT) and Diacylglycerol (DG) do not increase plasma TG, because MCT and DG are absorbed directly into the portal circulation to liver rather than being incorporated into chylomicron particles at intestine [4,5]. Therefore, fat intake (LCTs) affects the increase of CM formation and secrete into blood circulation with increased amount of TG mostly as VLDL remnants in 3-6 hours after fat intake.
Zilversmit first proposed the postprandial increase of TG to be the most common form of hyperlipidemia which associated with increased RLP as a risk for Cardiovascular Disease (CVD). Therefore, the postprandial TG increase has been long believed as the increase of TG in CM, CM remnants in plasma [3]. Because, increase of RLP and its ratio in the postprandial TG has not been clearly shown by ultracentrifugation separation (IDL) or other separation methods [6]. Using RLP immuno-separation method, the differences between increased TG and RLP in the fasting and postprandial plasma have been clarified [7-11]. Although the cholesterol content in RLP (RLP-C) is commonly found to be less than 10% even in the postprandial plasma TC, TG content in RLP (RLP-TG) is found to be more than 20% in the fasting plasma TG and as much as 50% in the postprandial plasma TG under various physiological conditions [12]. The postprandial RLP contained both apoB-48 and apoB-100 carrying particles. The increase of RLP apoB-100 particles (VLDL remnants) in fact was much greater (more than 80%) than that of apoB-48 containing lipoproteins (CM remnants) in the postprandial state [13-15]. Because the particle sizes of postprandial RLP-apoB48 and RLP-apoB100 are very similar (postprandial apoB48 particles in plasma are not large as being believed) [10], we found that major component of postprandial TG increased is VLDL remnants, but not CM remnants. Possibly, most of CM and CM remnants increased in plasma after food intake are incorporated into liver within a very short time [16,17] and re-constituted to VLDL and secreted as VLDL remnants in plasma.
Significantly higher RLP-TG is contained in the postprandial plasma than in the fasting plasma when the TG level is adjusted as the RLP-TG/TG ratio [18]. These results show that the amount and ratio of RLP in the postprandial TG increased significantly compared with the fasting plasma TG. In particular, the increase in the postprandial delta RLP-TG (postprandial RLP-TG minus fasting RLP-TG) levels contributed to approximately 50-60% of the increase in the postprandial delta TG (postprandial TG minus fasting TG) after regular meal intake. However, more than 80% of the increased delta TG was comprised of delta RLP-TG after a fat load or fat rich meal [18]. These results clearly show that the kind of food as contained in a fat rich meal greatly enhance the formation of RLP in the postprandial plasma compared with a regular meal. Marcoux et al. [19], Ooi et al. [20] and Nakajima et al. [21] previously reported similar results in small number of Caucasian and Japanese volunteers, in whom approximately 60-80% of the delta RLP-TG in delta TGs were found in 3-6 h after a fat rich meal. The rest of the increased TG consisted of increased non-RLP-VLDL-TG, LDL-TG and HDL-TG in the postprandial plasma, but do not comprise of TG a much as RLP-TG.
We have found that majority of LPL in plasma is bound to RLP and released into circulation as RLP-LPL complex both in pre-heparin and post-heparin plasma [22]. LPL bound to RLP showed no activity in non-heparin plasma and didnt increase after fat load in spite of the increase of RLP [23]. However, LPL levels in non-heparin plasma reflect the LPL activity for hydrolysis of CM and VLDL at endothelium. RLP-TG concentration and particle size increased in plasma after food intake is mainly regulated by LPL activity at endothelium together with other factors such as GPIHBP1 [24] and apoA5 [25]. A significant increase in the RLP-TG/RLP-C ratio was always higher in the postprandial plasma and ratio of LPL/RLP-TG was significantly lower. When LPL activity is not sufficient to hydrolyze overloaded CM or VLDL on the endothelial cells, less efficient hydrolysis occur and enhance the formation of less metabolized, large RLP particles along with the higher RLP-TG/RLP-C ratio. Those RLP particles carry a significantly lower LPL compared to small RLP particles, as shown by the low LPL/RLP-TG ratio [23, 26, 27]. Therefore, when the LPL activity and concentration is low, overloaded CM and VLDL are not hydrolyzed enough. Also when CM and VLDL are over loaded, LPL cant hydrolyze the excessive amount of TG-rich lipoproteins, resulting large size RLP particles are secreted into the postprandial plasma. As large RLP particles carry small amount of LPL, the function as ligand for the receptor incorporation of remnants [28], may become less effective for the clearance of remnants and accumulate more in plasma [22]. These results suggest that the large RLP with reduced ratio of bound LPL (LPL/RLP-TG) found in the postprandial plasma is a higher risk factor for obesity, insulin resistance and cardiovascular disease, as shown previous reported [29-31].
We have long thought that postprandial remnant lipoproteins (RLP) in plasma are significantly increased as the results of disturbed lipoprotein metabolism followed by the obesity and insulin resistance. Thereby, we believed that the insulin resistance as the result of obesity caused the enhancement of postprandial RLP formation. However on the contrary, we have proposed that RLP cause to induce the insulin resistance as the results of obesity which is induced by the excessive supply of RLP to visceral fat. Since the increase of VLDL remnants in plasma is the first step of lipid metabolism right after fat-rich meal intake as blood sugar after carbohydrate intake, we proposed that postprandial VLDL remnants are the other factor which enables to play the role for the storage of TG in adiposetissue from the circulation. The consumption of fat-rich meal and fructose are known to increase postprandial TG, fasting and postprandial RLP-C and RLP-TG, whereas consumption of glucose did not in healthy volunteers without obesity and insulin resistance [32,34]. Therefore, the kind of food intake significantly affects the formation of VLDL remnants and enhances the visceral fat obesity in normal volunteers. Therefore, we have recognized that the role for the formation of VLDL remnants after food intake is to provide TG as energy supply to organs and tissues, in particular to adipose tissue to prepare against starvation [34]. Further excessive and continuous supply of RLP from the blood stream enhances the accumulation of TG in visceral fat. The enlarged adipocytes by accumulated TG increase the secretion of TNF-α and other adipocytokines [35] and induce insulin resistance.
Takahashi et al. [36] reported that VLDL receptor, which is actually the most important VLDL remnant receptor, played the key role to induce the obesity and insulin resistance when fed with high-fat refined-sugar (HFS) in mice. Although there are many experimental animal studies that HFS can induce obesity and insulin resistance [37-42], those literatures reported simultaneous increase of TG or postprandial hyperlipidemia with insulin resistance and obesity or TG increase after insulin resistance and obesity. Goudriaan et al [43] reported that mice increased TG (VLDL remnants) in plasma by HFS, but mice could not proceed to store TG in adipose tissue without VLDL receptor and could not induce insulin resistance. Therefore, the formation of VLDL remnants as the first step after food intake should be positioned before the obesity and insulin resistance at the metabolic domino as the same position with blood sugar (Figure 1).
RLP is known to be cleared by LRP-1 and VLDL receptors in the liver, muscle, endothelium and adipose tissue in humans [36,41,42]. We recently found that LPL and apo (a) (a part of Lp(a)) were bound to RLP and formed RLP-LPL and RLP-apo(a) complex in plasma [1, 22]. Therefore, LPL and apo (a) as well as apoE could be the ligands for RLP to bind remnant receptors, especially VLDL receptor in adipose tissue. Elevated plasma RLP-C and RLP-TG concentration have been reported with the presence of insulin resistance [44-46]. Also, Yatsuzuka et al. [47] reported that RLP-C and RLP-TG were strongly associated with visceral fat volume. This suggests the VLDL receptor in adipose tissue reacts with ligands of RLP [37,41,42] and RLP supply FFA or incorporate into adipocytes. However, the mechanism of incorporation of RLP into adipocytes is not yet clarified enough. Thus, enlarged adipocytes by the increase of TG storage leads to the induction of insulin resistance. Therefore, we suggest that after the intake of high fat and refined sugar diet and/or with the lack of exercise, excessively increased RLP in plasma cause to initiate obesity and insulin resistance through VLDL receptor.
Nordestgaard et al. [48] and Bansal et al. [49] as well as Iso et al. [50] reported that the TG measured in non-fasting samples were more sensitive than the conventional measurements of the fasting TG concentrations in predicting the risk of cardiovascular events (the Copenhagen Heart Study, Womens Health Study and in a Japanese population study). Also, the Framingham Offspring Study previously cardiovascular risk factor, while RLP-C was an independent risk factor in the fasting plasma in women[51].
Postprandial TG and RLP are known to attain their highest levels 3-6 h after food intake [7-9]. Therefore, we analyzed the RLP-TG/TG ratio (concentration) and RLP-TG/RLP-C ratio (particle size) [10] associated with the lipoprotein lipase (LPL) in both the fasting and postprandial plasma. We have shown that significantly higher RLP-TG is contained in the postprandial plasma than fasting plasma when the TG level is adjusted as the RLP-TG/TG ratio [18]. These results show that the amount and ratio of RLP in the postprandial TG increased significantly compared with the fasting plasma TG. Karpe et al. [29,30] reported that endogenous TG-rich lipoproteins (TRL) accumulate in the human plasma after fat intake and the mechanism behind this phenomenon is the delayed lipolysis of the apoB-100 TRL particles due to a competition with CM for the LPL active sites. The large apoB100 TRL is more atherogenic than small TRL. Postprandial RLP with large particle size and low LPL are atherogenic lipoproteins and cause higher risk of cardiovascular diseases.
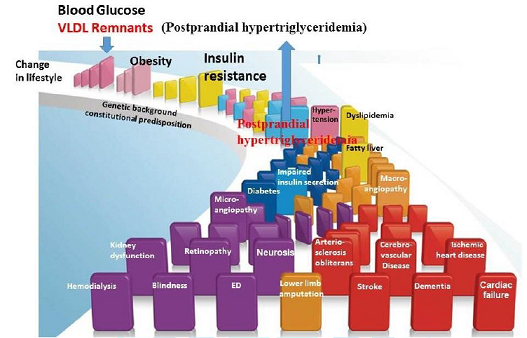
We have shown that postprandial VLDL remnants are increased significantly after fat rich meal or consuming fructose. Elevated VLDL remnants are significantly large sized RLP particles along with a smaller ratio of LPL in RLP in the postprandial plasma. Therefore, we have proposed that the increase of postprandial VLDL remnants comes first associated with unhealthy life style habit and cause to induce the insulin resistance as the results of obesity when the excessive RLP is supplied continuously to visceral adipocytes. Therefore, metabolic domino starts from the increased postprandial VLDL remnants in plasma as shown in Figure 1 and cause various cardiovascular diseases followed by the obesity and insulin resistance. The Japanese cuisine is an ideal food which increase low amount of VLDL remnants.
1. Nakajima K, Tokita Y and Tanaka A. Hypothesis: Postprandial remnant lipoproteins are the causal factors that induce the insulin resistance associated with obesity (2018) Clin Chim Acta 485: 162-132. https://doi.org/10.1016/j.cca.2018.06.029
2. Woods SC. Signals that influence food intake and body weight (2005) Physiol Behav 86: 709 –716. https://doi.org/10.1016/j.physbeh.2005.08.060
3. Zilversmit DB. Atherogenesis: a postprandial phenomenon (1979) Circulation 60: 473–485.
4. Nagao T, Watanabe H, Goto N, Onizawa K and Taguchi H. Dietary diacylglycerol suppresses accumulation of body fat compared to triacylglycerol in men in a double-blind controlled trial (2000) J Nutr. 130: 792-797. https://doi.org/10.1093/jn/130.4.792
5. St-Onge MT and Jones PJ. Physiological effects of medium-chain triglycerides: potential agents in the prevention of obesity (2002) J Nutr. 132: 329-332. https://doi.org/10.1093/jn/132.3.329
6. Cohn JS, Marcoux C and Davignon J. Detection, quantification, and characterization of potentially atherogenic triglyceride-rich remnant lipoproteins (1999) Arterioscler Thromb Vasc Biol 19: 2474-2486.
7. Nakajima K, Saito AT, Tamura M. Suzuki T, Nakano M, et al. Cholesterol in remnant-like lipoproteins in human serum using monoclonal anti apo B-100 and antiapo A-I immunoaffinity mixed gel (1993) Clin Chim Acta 223: 53-71. https://doi.org/10.1016/0009-8981(93)90062-9
8. Nakajima K, Okazaki M, Tanaka A, Pullinger CR, Wang T, et al. Separation and determination of remnant-like particles in serum from diabetes patients using monoclonal antibodies to apo B-100 and apo A-I (1996) J Clin Ligand Assay 19: 177-183.
9. Nakano T, Tokita Y, Nagamine T, Tanaka A, Okazaki M, et al. Measurement of serum remnant-like lipoprotein particle-triglyceride (RLP-TG) and RLP-TG/ total TG ratio using highly sensitive triglyceride assay reagent (2011) Clin Chim Acta 412: 71-78. https://doi.org/10.1016/j.cca.2010.09.040
10. Nakano T, Tanaka A, Okazaki M, Tokita Y, Nagamine T, et al. Particle size of apoB-48 carrying lipoproteins in remnant lipoproteins isolated from postprandial plasma (2011) Ann Clin Biochem 48: 57–64. https://doi.org/10.1258%2Facb.2010.010193
11. Nakajima K, Nakano T, Tokita Y, Nagamine T, Yatsuzuka S, et al. The characteristics of remnant lipoproteins in the fasting and postprandial plasma (2012) Clin Chim Acta 413 1077-1086. https://doi.org/10.1016/j.cca.2012.02.026
12. Nakajima K and Tanaka A. Atherogenic postprandial RLP;VLDL remnants as a causal factor in atherosclerosis (2018) Clin Chim Acta. 478: 200-215. https://doi.org/10.1016/j.cca.2017.12.039
13. Nakajima K and Tanaka A. Postprandial remnant lipoproteins as targets for the prevention of atherosclerosis (2018) Curr Opin Endocrinol Diabetes Obes 25: 108-117
14. Nakajima K, Nagamine T, Fujita MQ, Tanaka A and Schaefer E. Apolipoprotein B-48: A Unique Marker of Chylomicron Metabolism (2014) Advances in Clinical Chemistry, Burlington:Academic Press 64:117-177. https://doi.org/10.1016/B978-0-12-800263-6.00003-3
15. Schneeman BO, Kotite L, Todd KM and Havel RJ. Relationships between the responses of triglyceride-rich lipoproteins in blood plasma containing apolipoprotein B-48 and B-100 to a fat-containing meal in normolipidemic humans (1993) Proc Natl Acad Sci 90: 2069-2073.
16. Nestel PJ. Relationship between plasma triglycerides and removal of chylomicrons (1964) J Clin Invest 43: 943–949. https://doi.org/10.1172/JCI104980
17. Grundy SM and Mok HY, Chylomicron clearance in normal and hyperlipidemic man (1976) Metabolism 25: 1225–1239.
18. Nakajima K, Tokita Y, Sakamaki K, Shimomura Y, Kobayashi J et al. Triglyceride content in remnant lipoproteins is significantly increased after food intake and is associated with plasma lipoprotein lipase (2017) Clin Chim Acta 465: 45-52. https://doi.org/10.1016/j.cca.2016.12.011
19. Marcoux C, Hopkins PN, Wang T, Elizabeth TL, Nakajima K et al. Remnant-like particle cholesterol and triglyceride levels of hypertriglyceridemic patients in the fed and fasted state (2000) J Lipid Res 41: 1428-1436.
20. Ooi TC, Cousins M, Ooi DS, Steiner G, Nakajima K, et al. Postprandial remnant- like lipoproteins in hypertriglyceridemia (2001) J Clin Endocrinol Metab 86: 3134-3142. https://doi.org/10.1210/jcem.86.7.7627
21. Nakajima K, Nakano T, Moon HD, Nagamine T, Stanhope KL et al. The correlation between TG vs remnant lipoproteins in the fasting and postprandial plasma of 23 volunteers (2009) Clin Chim Acta 404: 124-137. https://doi.org/10.1016/j.cca.2009.03.051
22. Sato K, Okajima F, Miyashita K, Kobayashi J, Stanhope KL, et al. Majority of lipoprotein lipase is bound to remnant lipoproteins in plasma: A new definition for remnant lipoproteins (2016) Clin Chim Acta 46: 114-125 https://doi.org/10.1016/j.cca.2016.06.020
23. Ishiyama N, Nakajima K, Sakamaki K, Tokita Y, Tanaka A et al. Lipoprotein lipase does not increase in postprandial plasma (2017) Clin Chim Acta 464 204-210. https://doi.org/10.1016/j.cca.2016.11.035
24. Beigneux AP, Davies BS, Gin P, Weinstein MM, Farber E, et al. Glycosylphosphatidylinositol-anchored high-density lipoprotein-binding protein 1 plays a critical role in the lipolytic processing of chylomicrons (2007) Cell Metab 5: 279 –291. https://doi.org/10.1016/j.cmet.2007.02.002
25. Khovidhunkit W, Charoen S, Kiateprungvej A, Chartyingcharoen P, Muanpetch S, et al. Rare and common variants in LPL and APOA5 in Thai subjects with severe hypertriglyceridemia: A resequencing approach (2016) J Clin Lipidol 10: 505-511. https://doi.org/10.1016/j.jacl.2015.11.007
26. Shirakawa, T, Nakajima K, Shimomura Y, Kobayashi J, Stanhope K, et al. Comparison of the effect of post-heparin and pre-heparin lipoprotein lipase and hepatic triglyceride lipase on remnant lipoprotein metabolism. Clinica Chimica Acta 440 (2014) 193-200. https://doi.org/10.1016/j.cca.2014.07.020
27. Shirakawa T, Nakajima K, Yatsuzuka S, Shimomura Y, Kobayashi J, et al. The role of circulating lipoprotein lipase and adiponectin on the particle size of remnant lipoproteins in patients with diabetes mellitus and metabolic syndrome (2014) Clinica Chimica Acta 440: 123-132. https://doi.org/10.1016/j.cca.2014.10.029
28. Beisiegel U, W Weber and G Bengtsson-Olivecrona. Lipoprotein lipase enhances the binding of chylomicrons to low density lipoprotein receptor-related protein (1991) Proc Natl Acad Sci 88: 8342-8346. https://doi.org/10.1073/pnas.88.19.8342
29. Karpe F. Postprandial lipoprotein metabolism and atherosclerosis (1999) J Internal Med 246: 341-355. https://doi.org/10.1046/j.1365-2796.1999.00548.x
30. Karpe F, Steiner G, Olivecrona T, Carison LA and Hamsten A. Metabolism of triglyceride-rich lipoproteins during alimentary lipemia (1993) J Clin Invest 91: 748-758.
31. Rip J, Nierman MC, Wareham NJ, Luben R, Bingham SA, et al. Serum lipoprotein lipase concentration and risk for future coronary artery disease: the EPIC-Norfolk prospective population study (2006) Arterioscler Thromb Vasc Biol 26: 637-642. https://doi.org/10.1161/01.ATV.0000201038.47949.56
32. Stanhope KL, Schwarz JM, Keim NL, Griffen SC, Bremer AA, Graham JL, et al . Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J Clin Invest. 119 (2009) 1322-1334
33. Stanhope KL, Bremer AA, Medici V, Nakajima K, Ito Y,et al. Consumption of fructose and high fructose corn syrup increase postprandial triglycerides, LDL-cholesterol, and apolipoprotein-B in young men and women (2011) J Clin Endocrinol Metab 96: 596-605. https://doi.org/10.1210/jc.2011-1251
34. Stanhope KL, Medici V, Bremer AA, Lee V, Lam HD, et al. A dose-response study of consuming high-fructose corn syrup-sweetened beverages on lipid/lipoprotein risk factors for cardiovascular disease in young adults (2015) Am J Clin Nutr 101: 1144-1154. https://doi.org/10.3945/ajcn.114.100461
35. Hotamisligil GS, Shargill NS and Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance (1993) Sci 259: 87-91.
36. Takahashi S. Triglyceride Rich Lipoprotein -LPL-VLDL Receptor and Lp(a)-VLDL Receptor Pathways for Macrophage Foam Cell Formation (2017) J Atheroscler Thromb 24: 552-559.
37. Oscai LB, Miller WC and Arnall DA. Effects of dietary sugar and of dietary fat on food intake and body fat content in rats (1987) Growth 51: 64–73.
38. Oscai LB, Brown MM and Miller WC. Effect of dietary fat on food intake, growth and body composition in rats (1984) Growth 48: 415–424.
39. Xiao-Bing Cui, Jun-Na Luan, Jianping Ye and Shi-You Chen. RGC32 deficiency protects against high-fat diet-induced obesity and insulin resistance in mice (2015) J Endocrinol 224: 127–137. https://doi.org/10.1530/JOE-14-0548
40. Barnard RJ, DJ Faria, JE Menges, and DA Martin. Effects of a high-fat, sucrose diet on serum insulin and related atherosclerotic risk factors in rats (1993) Atherosclerosis 100: 229 – 236. https://doi.org/10.1016/0021-9150(93)90209-D
41. Barnard RJ, DA Martin, EJ Ugianskis and SB Inkeles. Role of diet and exercise in the management of hyperinsulinemia and associated atherosclerotic risk factors (1992) Am J Cardiol 69: 440–444. https://doi.org/10.1016/0002-9149(92)90981-4
42. Takahashi S, Sakai J, Fujino T, Hattori H, Zenimaru Y, et al. The very low-density lipoprotein (VLDL) receptor: characterization and functions as a peripheral lipoprotein receptor (2004) J Atheroscler Thromb 11: 200-208.
43. Goudriaan RJI, Tacken PJ, Dahlmans VE, Gijbels MJ, van Dijk KW, et al. Protection from obesity in mice lacking the VLDL receptor (2001) Arterioscler Thromb Vasc Biol 21: 1488-1493.
44. Imagawa M, Takahashi S, Zenimaru Y, Kimura T, Suzuki J, et al. Comparative reactivity of remnant-like lipoprotein particles (RLP) and low-density lipoprotein (LDL) to LDL receptor and VLDL receptor: effect of a high-dose statin on VLDL receptor expression (2012) Clinica Chimica acta 413: 441- 447. https://doi.org/10.1016/j.cca.2011.10.033
45. Chan DC, Watts GF, Barrett PH, Mamo JC and Redgrave TG. Markers of triglyceride-rich lipoprotein remnant metabolism in visceral obesity (2002) Clinica Chemica Acta 48: 278-283.
46. Ohnishi H, Saitoh S, Takagi S, Ohata J, Isobe T, et al. Relationship between insulin-resistance and remnant-like particle cholesterol (2002) Atherosclerosis 164: 167-170. https://doi.org/10.1016/S0021-9150(02)00057-6
47. Yatsuzuka S, Shimomura Y, Akuzawa M, Ando Y, Kobayashi J, et al. Plasma adiponectin is a more specific marker of fatty liver than a marker of metabolic syndrome in Japanese men (2014) Ann Clin Biochem 51: 68–79. https://doi.org/10.1177%2F0004563213487892
48. Nordestrgaad BG, Benn M, Schnohr P and Tybjaerg-Hansen A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women (2007) JAMA 298: 299-308.
49. Bansal S, Buring JE, Rifai N, Mora S, Sacks FM, et al. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women (2007) JAMA 298: 309-316.
50. Iso H, Naito Y, Sato S, Kitamura A, Okamura T, et al. Serum triglycerides and risk of coronary heart disease among Japanese men and women (2001) Am J Epidemiol 153: 490-499. https://doi.org/10.1093/aje/153.5.490
McNamara JR, Shah PK, Nakajima K, Cupples LA, Wilson PW, et al. Remnant-like particle (RLP) cholesterol is an independent cardiovascular disease risk factor in women: results from the Framingham Heart Study (2001) Atherosclerosis 154: 229- 236.
*Corresponding author:Katsuyuki Nakajima, Graduate School of Health Sciences, Gunma University, 3-47-4, Minami-Cho, Maebashi, Gunma, Japan(371-0805), Tel: +81-27-223-7267, Fax No: +81-27-223-7267, E-mail: nakajimak05@ybb.ne.jp
Citation: Nakajima K, Tokita Y and Tanaka A. Triglyceride is significantly increased in remnant lipoproteins after food intake and its association with lipoprotein lipase in the plasma (2018) J Obesity and Diabetes 2: 6-10