Explore journal overview, editorial leadership, indexing, articles in press, latest published work, and highlights from previous issues.
Review Article :
N.Venkatesan and M. Ramanathan As per WHO “Drug is
any substance or product that is used or is intended to be used to modify or
explore physiological systems or pathological states for the benefit of the recipient”.
Hence the prime objective of using any substance as a drug is that it must be beneficial
for the humans. A large number of compounds are synthesized every year but they
cannot be directly used in humans as drugs because no one knows or can predict the
possible harmful effects of these compounds in humans. That is why to explore
the complete pharmacological profile of these compounds and to ensure complete
human safety they are first tested on animals before clinical use. Preclinical
Studies thus can be defined as “Testing the newly discovered compound in
animals with the objective of gaining information regarding the various aspects
of the compound with respect to the biological systems so that the same can be
extrapolated for the use of that compound in humans”. As the evaluation
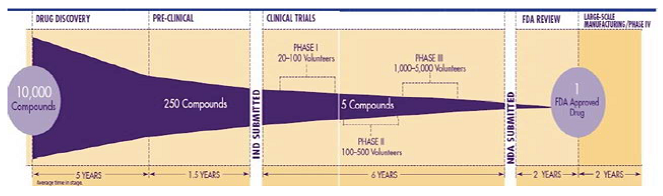
progresses undesirable compounds gets rejected at each step, so that only a few
out of thousands reach the stage when administration to the humans is
considered. (Figure 1) The selection of the type and the number of animals
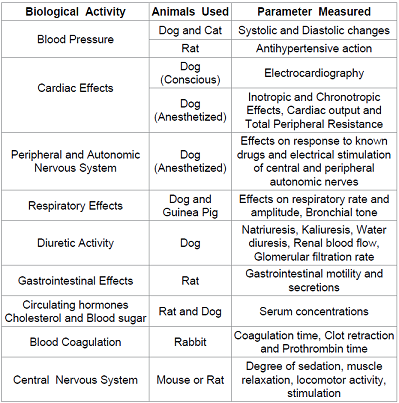
used for the preclinical studies is one
of the most important steps. The choice of the species is based on the fact
that which one will give the best correlation to the human trials. Generally,
experiments are first performed on the rodents
and then on the larger animals like canines. Differences in the gut, enzyme
activity, circulatory system, or other considerations make certain models more
appropriate based on the dosage form, site of activity, or noxious
metabolites. For example Canines may not be good models for solid oral
dosage forms because the characteristic carnivore intestine is underdeveloped
compared to the omnivores and gastric emptying rates are increased. Also,
rodents cannot act as models for antibiotic drugs because the resulting
alteration to their intestinal flora causes significant adverse effects. Depending on drugs functional groups, it may be
metabolized in similar or different ways between species, which will affect
both efficacy and toxicology [1]. Differences in drug response due to species
differences are taken into account while extrapolating the data to humans. For
Example: Amphetamine and Ephedrine are predominantly metabolized by oxidative
deamination in men and rabbits where as in rats aromatic oxidation is the major
route. Most studies are performed in larger species such as
dogs, pigs and sheep which allow for testing in a similar sized model as that
of a human. In addition, some species are used for similarity in specific
organs or organ system physiology. For Example: Swine for dermatological
and coronary stent studies, goats for mammary implant studies, dogs for gastric
studies, rabbits for dermatological studies etc. (Figure 2) (Table 1) Stages of
Preclinical Studies Preclinical clinical studies
require the time span of 3-6 years. The reason is that, the newly discovered compound is tested at various
levels to ensure its complete safety in each and every aspect in animals so
that it can be further utilized for testing in humans. Thus the preclinical
studies are performed at different stages which are as follows: Figure 1: Reduction in number of compounds in each stage of the drug discovery. Figure 2: The pancreas of the rabbit is removed. General
Observational Tests It is not possible to devise a
scheme of testing which will reveal all
the different types of potentially useful pharmacological activity which may be
possessed by a particular substance [2]. The person in charge of a screening
program must decide on the precise procedure which is to be adopted in
individual cases. Much can be learnt by making simple observations on the
effect of the compound on the behavior of conscious animals. One of the most
useful and best known methods of obtaining the maximum amount of information
from these simple tests is that designed by Irwin commonly called as the
“Irwin’s Primary Tests for Pharmacological Activity.” In this mice are given
intraperitoneal injections of the substance under test and its effects are
assessed by systematic observation of the animal’s subsequent behavior and
responses. The observations enable an activity profile to be built up so that
the actions of the compound being tested can be compared with that of reference
drugs of known activity. Most of the observations listed in the Irwin’s tests
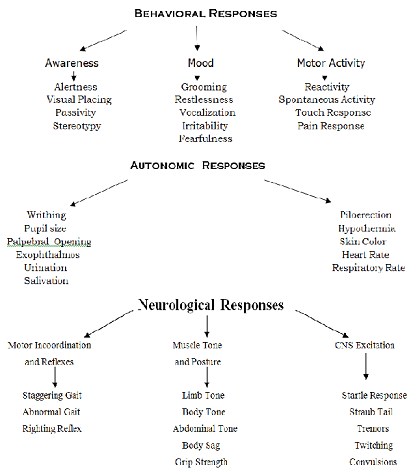
are simple and are as follows: (Figure 3) When a drug’s profile is being drawn
by the application of Irwin’s Method, small groups of mice are given the drug
at a number of dose levels, a useful range is 1, 3, 10, 30 and 100 mg/ kg of
body weight but with very active substances it may be given to establish
thresholds of activity by reducing the dose in progressive steps below the
level 1 mg/100 gms of body weight. Pharmacological activity detected by these
simple tests must of course be thoroughly investigated by the methods discussed
in the later part of the section. Screening Tests: The
word “screen” implies that the substances under test are exposed to a process which
will hold back substances of potential value and let through the rest. In order
to achieve this satisfactorily, the mesh of the screen should be small enough
to retain all compounds of interest, even if some have to be rejected when they
are subjected to the more rigorous procedures which constitute the secondary screening
tests [3]. The screening is thus defined as “The process of filtering among
the large number of compounds, the one with required biological activity”. The screening
program is basically of three types: Figure 3: Irwins Primary Tests for Pharmacological Activity. Simple or Single
Screening:
This employs a single test or perhaps two similar tests to find substances that
are active in a single way. A hypoglycemic
test, which measures the ability of a compound to diminish the
concentration of sugar in the blood, is an example of this type. This is
usually done when the new compound have been produced by a simple modification
of the structure of a substance of proved therapeutic value. Programmed or
rational screening: This
employs sound physiological, biochemical, pathological knowledge and identification
of drug action. The compound is aimed at mitigating the derangement caused by
the disease. Use of levodopa in Parkinson
based on the finding that the condition resulted from the deficiency of
dopamine in the striatum. Blind or random
screening:
When a compound of entirely new type comes into the hands of the
pharmacologist, it has to be subjected to a series of simple tests designed to
reveal the nature of any pharmacological activity it possess and thus to
indicate the direction to be taken by more detailed investigations if these seems
to be justified. For obvious reasons, the application of this battery of simple
tests is described as the blind
screening. If the pharmacological activity of a particular kind is
discovered in the course of a blind screening program, it will be necessary to investigate
it in more detail by one of the methods employed for the study of that
particular activity In practice, the overall strategies which are adopted for
the pharmacological investigation of new compounds do not differ greatly from
one another. Thus, if a substance has been produced for obtaining the
particular type of activity, it will still be necessary, when the presence of
the primary activity has been confirmed, to apply the test for other
pharmacological effects. An animal model is a living,
non-human animal used during the
research for investigation of human disease or for determining the therapeutic
potential of a newly discovered compound for the purpose of better
understanding the disease and its cure without the added risk of causing harm
to an actual human being during the process. The animal chosen will usually meet
a determined taxonomic equivalency to humans, so as to react to disease or its treatment
in a way that resembles human
physiology as needed. Many drugs, treatments and cures for human diseases
have been developed with the use of animal models. The animal models used can
be classified as follows: Chemical models:
This
involves the use of some chemical that selectively destroys some cells or
tissues or induce certain pathophysiological condition very similar to what
actually occurs in some disease to study the therapeutic effect of a compound. For
example: MPTP model for Parkinsonism, where 1-methyl- 4-phenyl-1,2,3,6-tetrahydropyridine
(MPTP) is used for the induction of parkinsonism. Physical models: This involves
the use of various physical factors to induce the symptoms similar to that
occurring in a disease or disorder. For example: Use of Electric shock to
induce convulsions in rats for studying the anticonvulsant activity of a compound. Surgical models: This involves
the surgical removal of certain tissue or organ of the body to study the
particular activity of a compound. For example: The pancreas of the rabbit is
removed to induce diabetes and to study the antidiabetic effect of a compound. Genetic models: Genetic
manipulation either by addition of genes
(Transgenic Technology) or by modification of the existing genes (Gene Knockout
Technology) is a very powerful tool with tremendous applications in the drug
discovery [4]. Transgenic mouse is becoming very popular animal for studying
the disease processes and testing of the newer drugs. It is relatively easy to manipulate
the mouse gene. Addition of genes or the loss of gene activity often causes
changes in a mouse’s phenotype, which includes appearance, behavior and other
observable physical and biochemical characteristics. Screening of bioactive
molecules for drug research is one of the important areas where transgenic and knockout
mice have tremendous potential. This technology is also being used to create
models of human diseases. Some of the newer variations of knockout and
transgenic technologies such as knockins (to replace a gene in the genome with
a modified version of itself) and the floxP system (to disrupt a gene at a
specific time of development) allow investigators to manipulate the gene functions
in particular tissues or at certain times. For example: ob/ ob knockout mice
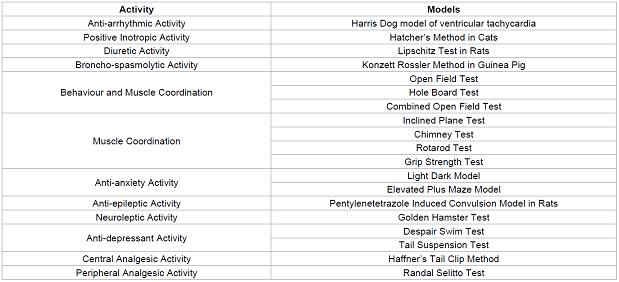
model to study obesity and db/db knockout mice model to study diabetes. Table 2 Bioassays
are the procedures by which the potency or the nature of the substance is
estimated by studying its effects on living matter. They are usually designed
to measure the relative potency of the two preparations, usually a standard and
an unknown. Use of standard substance for comparison also helps in solving
problems arising from biological variations. The observed response or effect of
the unknown would be always relative to the effect that is produced by a
standard substance. The standard substance is a pure substance and, in official
bioassays it refers to pharmacopoeial standards. In case of hormones,
biological products and vaccines
it is often necessary to establish the standard response of the standard substances
against which unknown samples can be calibrated. Bioassays are also essential
in the development of new drugs. In the preclinical assessment of a new
compound, the biological activity is compared with that of known (standard)
compound using appropriate test systems. In such studies, the tests must be
simple reproducible and economical. Biological assessments of a new compound
generally consists of carrying out a battery of such assays and based on these
tests, constructing a profile of activity. Clinical testing of drugs is guided
by such profile of activity generated in animals [5]. Bioassays are usually
employed when: 1. A chemical assay for the
substance is not available or the substance gets inactivated by interacting
with chemicals as in case with hormones. 2. When the quantity of sample is
too small. 3. To estimate the concentration
of the active principles present in the tissue extracts, the endogenous
mediators like acetylcholine,
serotonin, prostaglandins etc. 4. To measure the pharmacological
activity of new or chemically unidentified substances. 5. To measure the compound’s
toxicity. 6. When the bioassay is more
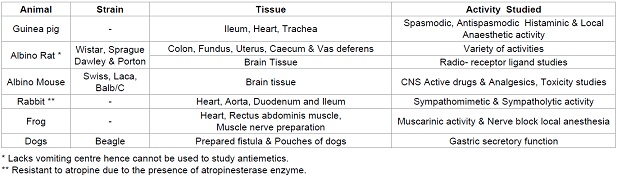
sensitive than the chemical assay. Isolated tissues
or organs: The
tissues or organs separated from some
animals can be used to study some specific activity of the compound. The type
of animal and tissue to be selected depends upon the type of activity to be
tested. Table 3 Cell cultures: In these
assays, the pure cells of specific lines are grown on a suitable culture media
and then these pure cell lines are used to measure the specific type of
activity. This type of study is commonly employed for measuring the antimicrobial
activity of a particular compound against specific microbe, where the pure
cultures of microbe are grown on specific culture media and then the
antimicrobial drug’s effect is evaluated by measuring the diameter of the zone
of inhibition. For example: i) Pure cultures of Mycobacterium megmatis is used
for the assay of the antibiotic Bleomycin sulphate. ii) Primary cell culture of
mouse spinal cord neurons are used in neuropharmacology for studying
neurotransmitter receptor functions. Intact animals:
In these types of assays, the whole animal is used to measure the potency of
the compound. These types of assays are less common and are employed for very
few substances. For example: Bioassay of insulin using mouse or of digitalis in
guinea pigs. Compounds that are found active in the above tests are then taken
up for the more detailed and elaborative study for confirming the activity in
the more precise manner. In this stage the compounds undergo testing by a large
number of more detailed tests for the particular activity and simultaneously
for the other analogous activities it possess. Table 2: Some of the most commonly used animal models for studying various activity. Table 3: Some of the animal tissues used and the activity. In most of the cases where the
new compound has been obtained via rational approach, combinatorial chemistry, molecular
modeling or sometimes even in case of chemically synthesized compounds, the
mechanism of action of compound is predetermined as the compound is synthesized
with the predetermined target in mind hence the mechanism of action is already
known. However in case of compounds obtained from natural sources like plant
extracts, microbes etc. the determination of mechanism of action and the
targets of their action is required which is a troublesome task. Radio ligand
Binding Assays are the most used techniques for this purpose which involves the
use of radioactive atom, bound to the new compound and then its fate inside the
biological system is studied using radio-detection techniques. Recently, newer
approaches with more precise measurements are employed for this purpose and
some of them are enumerated as follows: 1. Fluorescence Resonance Energy
Transfer (FRET) 2. Fluorescence Correlation Spectroscopy
(FCS) 3. Homogeneous Time Resolved
Fluorescence Technology (HTRFT) 4. Confocal Microscopy 5. Scintillation Proximity Assay
(SPA) and many more. Irrespective of the primary
mechanism of compound’s action, its effect on other vital organs of the body
like brain, heart, kidney, liver, respiratory organs, gastrointestinal
tract etc are simultaneously determined by measuring related parameters and
the complete pharmacological profile of the compound is explored. Quantitative
Tests: Quantification
of the compound’s response or activity
is very essential as it is the dose that makes a compound a drug or a poison.
There are basically two types of measurement whereby the effect of a compound
on an animal may be evaluated: i) Graded
Effect: These
are based on the principle that there is a proportionate increase in the
observed response with a subsequent increase in the concentration or the dose.
For example: The contraction of the smooth muscle preparation for assaying
histaminic activity of a compound. ii) Quantal
Effect: These
are based on “All or None” phenomenon. This type of response of a group of
animals is measured in order to determine the percentage responding. In these
cases the end point is an all or none response. For example: Whether a compound
causes cardiac arrest or not. One of the most important parameter that is
quantified by these studies is the ED50 value. ED50 is defined as the dose
effective for producing a certain sign in 50% of the animals of a group. The units
are those of the dose (mg/kg) and the value is, of course, different for each
route of administration. The ED50 is calculated, since it would be fortuitous
that one of the doses of a series should produce the effect in exactly half of
the animals. When the all or none response, also called the quantal response is
death, the ED50 becomes the LD50 or the lethal dose for 50% of the animals. Sometimes
the ED75, the ED10 and the ED99 etc, for example, are desired in order to know
a dose affecting most of the animals, a nearly minimally effective dose etc. There
are two types of methods for calculating ED50: i) Graphical Method: Miller and
Tainter Method, Litchfield and Wilcoxon Method. ii) Arithmetical Method: Reed and
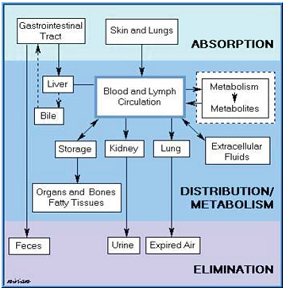
Muench Method, Karber Method. Pharmacokinetics
is the quantitative study of the drug movement in, through and out of the body
with respect to time. This is the one of the most important stage as it is
found that maximum percentage of compounds are rejected in preclinical studies
during this stage only due to undesirable pharmacokinetic profile. The
following parameters are determined during pharmacokinetic studies: Figure 4: Pharmacokinetic profile. 1. Absorption
Parameters: a) Absorption of compound via
various routes. b) Mechanism of absorption. c) Various factors affecting
absorption. d) Rate and extent of absorption
(Bioavailability). 2. Distribution
Parameters: a) Tissue permeability of
compound. b) Volume of distribution. c) Protein and tissue binding of
compound. d) Factors influencing
distribution. 3. Metabolic
Parameters: a) Pathways of metabolism. b) First Pass Metabolism. c) Factors influencing
metabolism. d) Enzyme induction / Enzyme
inhibition. e) Bioactivation and other
parameters. 4. Excretion
Parameters: a) Routes of excretion. b) Clearance. c) Dose Adjustments. d) Factors influencing excretion. 5. Interaction: a) Pharmacokinetic interactions. b) Pharmacodynamic interaction. Other
Parameters: a) Plasma Concentration – Time
profile. b) Bioavailability studies. c) Therapeutic
Concentration Range studies. d) Design of dosage regimen. e) Concentration response
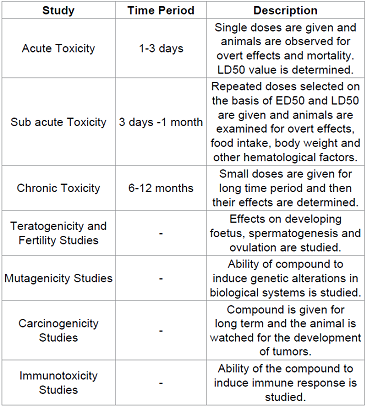
studies. It involves the study of
injurious effects of the compound on the animals along with the mechanisms of
toxicity. The main aim of these studies is to determine the safety of the
compound in at least 2 animal species, mostly mouse/rat and dog by oral and
parenteral routes. Table 4 It is during the toxicity studies when the multiple
dose levels are selected and assayed in order to determine NOAEL (No Observable
Adverse Effect Level) value, which will help to determine a safe starting dose
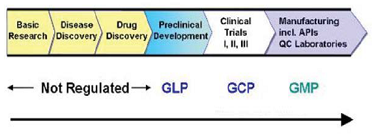
level and dose escalation scheme for the phase I clinical trials. (Figure 5) The regulation of the Drug
Discovery process begins from the preclinical stage where the large number
of regulatory authorities has established various well defined protocols that
are to be complied during the entire process. GLP (Good Laboratory Practices)
is one of the essential requirements of all modern testing facilities. It embodies
a set of principles that provide a framework within which laboratory studies
are planned, performed, monitored, reported and archived. In order to promote
international cooperation and harmonization, the Organization for Economic
Cooperation and Development (OECD) has developed certain test guidelines and principles
of GLP. GLP provides an assurance to regulatory authorities that the data
submitted are a true reflection of the results obtained during the study and
can, therefore, be relied upon when making risk/safety assessment. GLP are a
useful set of standards for any research laboratory wishing to standardize
procedures and verify the reliability of data produced. This is an issue of
increasing importance to the research sponsors. In 1980, OECD Council recommended
that all the member countries should apply these guidelines in the testing of
compounds and the data generated in accordance with the OECD test guidelines
and OECD principles of GLP shall be accepted by all the member countries. Some
other regulatory bodies involved are:
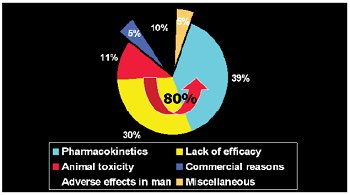
Table 5 Table 4: Various types of toxicity studies that are done during preclinical studies. Table 5: Some other regulatory bodies involved. Figure 5: Preclinical Studies are conducted as per GLP requirements as detailed in 21 CFR Part 58. Figure 6: Statistics regarding Preclinical Trials. • Approximately 10% of total cost
involved (15-30 Billion Dollars) is spent for preclinical studies during drug discovery process [6]. • Approximately 80% of the
compounds taken for preclinical study fail during the process. • Approximately 39% of the compounds
are rejected in the preclinical studies due to poor pharmacokinetic profiles. • Approximately 11% are rejected
due to toxicological issues. Figure 6 Preclinical Studies though
complicated and time consuming, acts as the bridge between the synthesis or
discovery of a newly discovered compound and its testing is human. The data
generated during preclinical study helps a lot in approximately picking up a
right compound, a right formulation and a right drug delivery system that
reduces the risk of injurious effects on humans. However there are certain
shortcomings of preclinical studies like there is limited information on
mechanism of action, biologically relevant animal species or models of disease
are not always available and sometimes limited information is available to
support the validity of extrapolation from animal to humans. Still preclinical studies
play a very important role in the drug discovery process. 1.
Ariens EJ, Simonis AM, Offermeier J. Introduction to General Toxicology (1976)
Academic Press Inc, USA. N Venkatesan, S.B College of Pharmacy, Anaikuttam Road, Anaikuttam, Sivakasi, India E-mail: nvenkatpharmabhu@gmail.com N.Venkatesan, M. Ramanathan (2017) Preclinical Toxicity Studies-Tool of Drug Discovery. PVPE 1: 1-7 Drug Discovery, Therapeutic Concentration, Pharmacokinetics, Spectroscopy, antimicrobial activityPreclinical Toxicity Studies-Tool of Drug Discovery
Abstract
Full-Text
Selection of Animals for Preclinical
Studies

Tests on Animal
Models of Human Disease
Bioassays and
Confirmatory Tests
Bioassays are
usually done using
Determination of
mechanism of action and systemic pharmacology
Determination of
Pharmacokinetic Parameters
Toxicity Studies
Regulatory
Bodies involved in Preclinical Trials

Statistics
regarding Preclinical Trials
Conclusion
References
2.
Crossland J. Lewis’s Pharmacology 4th edn (1971) Williams and Wilkins Co., USA.
3.
Turner RA, Peter Hebborn. Screening Methods in Pharmacology (1965) Elsevier,
USA.
4.
Vogel HG. Drug Discovery and Evaluation 2nd edn (2002) Springer, USA.
5.
Kulkarni S.K, Practical Pharmacology and Clinical Pharmacy 1st edn (2008)
Vallabh Publications, India.
6.
Barile FA. Principles of Toxicological Testing (2008) CRC Press, USA.*Corresponding author
Citation
Keywords