Left Ventricular Pseudo Aneurysm (LVPA) is a
rare complication occurs in population that carries the risk. Clinical
presentation of a large LVPA can be variable which make the diagnosis
challenging. Given the high risk of death in such cases, high clinical
suspicion in addition to the use of proper diagnostic modality may reduce the
mortality rate & guide the management decision. Echocardiogram is
considered the proper investigation tool to screen, diagnose the presence of
LVPA & identify the high-risk feature of rupture. In this review will
present catastrophic echocardiographic features in unusual clinical case
presentation of a patient with large LV pseudo aneurysm presented with ischemic
stroke & background of diabetes & hypertension.
Background
Left Ventricular Pseudo Aneurysm (LVPA) results from cardiac rupture contained by organizing thrombus and the pericardium, and are characterized by the nature of their walls, narrow necks, and high risk of rupture [1]. LVPA most frequently forms after trans-mural myocardial infarction but may result from cardiac surgery, trauma, infections, inflammation, or structural defects [2]. In a true aneurysm, LV out-pouching is a thinned out wall but with some degree of myocardium wall integrity intact. Such an entity calls for elective surgery. Pseudo aneurysms are considered to have poor prognosis. We have reviewed the literature on this challenging diagnosis and summarized the characteristic findings of this entity.
Discussion
Clinical presentation of LV pseudo aneurysm is unpredictable.
Heart failure,
chest pain, and
dyspnea are the most frequently reported symptoms, although pseudo aneurysm is frequently asymptomatic or present with nonspecific symptoms [2].
Myocardial infarction accounts for most LV pseudo aneurysms given higher chance of inferior wall MI rather than an anterior wall with finding is consistent with the location on the left ventricle posterior (infero-lateral), apical or inferior wall. Anterior wall pseudo aneurysm can be fatal.
Diagnostic modalities in predicting the high-risk population should be incorporated. The most common initial tools are resting 12-lead Electrocardiography with signs of infarction that include ST segment deviation, poor R-wave progression, or deep T-wave inversion in the pericardial leads (Figure 1) & chest X-ray may revealed increasing cardiac silhouette with or without evidence of interstitial pulmonary edema (Figure 2 A, 2B).
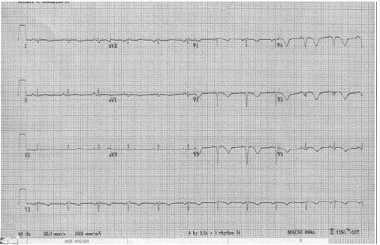
Figure 1: Twelve-lead electrocardiogram showing anterior T wave changes and poor R-wave progression in patient with history of anterior MI.
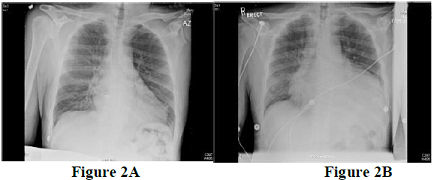
Figure 2: Chest X-ray showing significantly increased cardiothoracic ratio, without (A) and with (B) evidence of interstitial pulmonary edema.
Visualization of LVPA may be accomplished by a variety of methods. Echocardiography transthoracic or transesophageal remains the source of imaging, given its relatively low cost, reproducibility, ease of performance, lack of iodinated contrast, versatility, and its noninvasive nature [3].Assessing the location, size, involvement of valvular apparatus and presence of mural thrombus. Furthermore it defines the global /regional function of the ventricles, valvular regurgitation and diastolic dysfunction [4].
Left ventriculography will show a paraventricular chamber filling via a relatively narrow ostium. The diagnosis is confirmed by demonstrating an avascular wall on coronary arteriography. Cardiac MRI and cardiac CT allow visualization of any plane of the heart and can thus show segments that are difficult to see on echocardiography.
Left ventricular pseudo aneurysm (LVPA) is under high pressure, it is predisposed to rupture with subsequent hemorrhage and death; thus, surgical repair is the traditionally indicated. It is not currently possible to predict which patient will experience rupture and when rupture will occur. However, high-risk features such as peri-infarct discovery and LVPA identification within 3 months of a likely mechanism, rapid expansion, or large size (>3 cm) should prompt immediate surgical consultation. For patients without these high-risk features, frequent echocardiographic measurements of LVPA size may be an acceptable alternative [5].
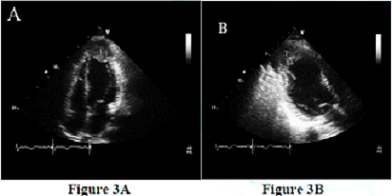
Figure 3: Apical four-chamber view (A) and apical long-axis view (B) are showing the apical thrombus (T) within the aneurysm.
An example of totally unpredictable clinical presentation of LVPA with fatal outcome prior to the surgical management was a case of 48 years old male patient presented with ischemic stroke. A transthoracic echocardiogram was performed to rule out cardiac source of emboli which demonstrate a large apical left ventricular aneurysm filled with large mobile thrombus measured 2.5 x 3.5 cm in size (Figure 3 A, 3B). There was moderate degree of LV systolic dysfunction associated with segmental wall motion abnormalities in form of dyskinesia at the left anterior descending artery territory. The aneurysm of LV is an area of thinned myocardium that is dyskinetic and involves the full thickness of the wall. Repeated transthoracic echocardiogram after 4 weeks revealed very large apical LV pseudo aneurysm measured 8.5 x 6 cm in diameter (Figure 4A, 4b) with bidirectional color flow. The neck originated at the apex and measured around 3 cm. There was a large laminated thrombus within the pseudo aneurysm with no mobile components. This large pseudo aneurysm of LV apax is a result of rupture of the ventricular free wall but contained by the overlying adherent pericardium or scar tissue, which is uncommon to occur [6-7]. Current literature reports that 30–45% of ventricular pseudo aneurysms will rupture. Consequent mortality reaches up to 48% after drug therapy and 23% after surgical treatment [6, 8].
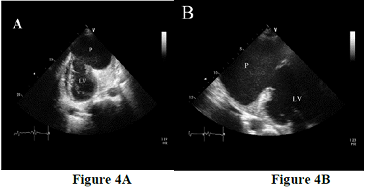
Figure 4: Repeat echocardiogram 4 weeks after admission from apical two-chamber (A) and apical long axis (zoomed view) (B) are showing a large left ventricular pseudo aneurysm (P) communicating with the left ventricular (LV) cavity.
Conclusions
LVPA is rare but deadly. As discussed above, the signs and symptoms in the population at risk are non-specific & can be unpredictable. The high clinical suspicion with early use of imaging modality is required. Glimpse into the natural history of this entity with the role of echocardiography as a diagnostic tool to identify the presence of this fatal pathology is necessary.
References
1. Dachman AH, Spindola-Franco H and Solomon N. Left ventricular pseudoaneurysm its recognition and significance (1981) JAMA 246: 1951-1953. doi:10.1001/jama.1981.03320170063036
2. Yeo TC, Malouf JF, Oh JK and Seward JB. Clinical profile and outcome in 52 patients with cardiac pseudoaneurysm (1998) Ann Intern Med 128: 299-305. https://doi.org/10.7326/0003-4819-128-4-199802150-00010
3. Catherwood E, Mintz GS, Kotler MN, Parry W R and Segal B L. Two-dimensional echocardiographic recognition of left ventricular pseudoaneurysm (1980) Circulation 62: 294-303.
4. Gatewood RP Jr and Nanda NC. Differentiation of left ventricular pseudoaneurysm from true aneurysm with two dimensional echocardiography (1980) Am J Cardiol 46: 869-878.
5. Natarajan MK, Salerno TA, Burke B, Chiu B and Armstrong PW. Chronic false aneurysms of the left ventricle: management revisited (1994) Can J Cardiol 10: 927-931.
6. Brown SL, Gropler RJ and Harris KM. Distinguishing left ventricular aneurysm from pseudoaneurysm A review of the literature (1997) Chest 111: 1403-1409. https://doi.org/10.1378/chest.111.5.1403
7. Bisoyi Samarjit, Anjan K Dash, Debashish Nayak, Satyajit Sahoo and Raghunath Mohapatra. Left Ventricular Pseudoaneurysm versus Aneurysm a Diagnosis Dilemma (2016) Annals of Cardiac Anaesthesia 19: 169-172. https://dx.doi.org/10.4103%2F0971-9784.173042
8. Si D, Shi K, Gao D and Yang P. Ruptured left ventricular pseudoaneurysm in the mediastinum following acute myocardial infarction: A case report (2013) Eur J Med Res 18: 2. https://dx.doi.org/10.1186%2F2047-783X-18-2
*Corresponding author:
Jamilah Al Rahimi, Division of Medicine/Cardiology, King Saud bin Abdulaziz Health Science University, King Faisal Cardiac Center, King Abdulaziz Medical City, Ministry of National Guard, Jeddah, Saudi Arabia, Fax: 920008668 ext25809, E-mail: dr_jameelah@hotmail.com
Citation:
Al Rahimi J. Is Expanding Heart Always Serious (2018) Clinical Cardiol Cardiovascular Med 1: 11-12.
Heart, hypertension and Left Ventricular Pseudo
Aneurysm.