Case presentation
A 71-year-old male with hyperlipidemia, hypertension, Paroxysmal Atrial Fibrillation (PAF), Ischemic Heart Disease (IHD), Ischemic Cerebrovascular Accident (CVA), with permanent ICD for primary prevention was admitted to the neurological department with a paresthesia and numbness of the face and extremities. On neurological examination extreme proximal muscle weakness with poor sensation of palms and soles, depressed reflexes and unsteady walking. The cranial CT showed old small infarcts. His background treatment included: P.O. Aspirin 100 mg q.d., P.O. Atorvastatin 40 mg q.d., P.O. Enaladex 5 mg q.d., P.O. Sotalol 40 mg b.i.d. He was started on treatment with plasmapheresis and 3 days later IVIG treatment was added. During admission bacteremia with Klebsiella Pneumonia on blood culture and treatment with Tazocin was started.
Three days later the patient began to have recurrent episodes of sustained VT (Figure 1). The ICD did not sense the events because the threshold rate for VT detection was higher than the actual rate of VT in this patient. The patient was hemodynamically stable during each episode of VT. Treatment was started with Amiodarone, beta-blockers and the patient received DCA cardio version on two occasions.
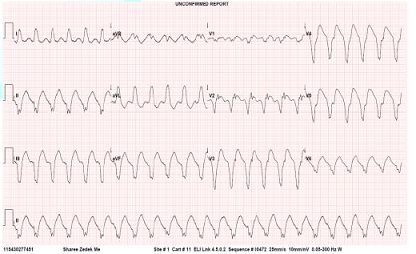
Figure 1: ECG showing Ventricular Tachycardia.
Intravenous Lidocaine was begun and patient was transferred to the ICCU. Laboratory examination: Electrolytes, creatinine was WNL. WBC was 17.2 K/uL (normal range 5-10 K/ul) with Neutrophil count predominance, CRP was 11.4 mg/dL (normal range 0-0.5 mg/dL). Cardiac enzymes normal. Prothrombin time 12.9 s (INR of 1.07), and a partial thromboplastin time normal. While treated with amiodarone and lidocaine no further tachyarrhythmias detected.
Discussion
The Guillain-Barre syndrome, which is characterized by acute areflexic paralysis with albuminocytologic dissociation (i.e., high levels of protein in the cerebrospinal fluid and normal cell counts), was described in 1916 [1]. Since poliomyelitis has been eliminated, the Guillain-Barre syndrome is currently the most frequent cause of acute flaccid paralysis worldwide and constitutes one of the serious emergencies in neurology [2].
The presentation of GBS may be acute or sub-acute in presentation [2]. It affects the peripheral nerves and is characterized by symmetrical progressive ascending weakness with areflexia and variable sensory complaints [3,4]. The GBS is presumed to be caused by an aberrant auto immune response against peripheral nerves by cross-reacting antibodies [5,6]. The incidence of the GBS is estimated at 1 to 2 per 100,000 per year with a preponderance in women over 50 years of age [7,8]. GBS is often preceded by an infection that is believed to evoke an immune response [9]. The classification is based on nerve-conduction studies and there is a notable difference in the geographic distribution of subtypes of the syndrome [10,11] The classic pathological findings in acute inflammatory demyelinating polyneuropathy are inflammatory infiltrates (consisting mainly of T cells and macrophages) and areas of segmental demyelination, often associated with signs of secondary axonal degeneration, which can be detected in the spinal roots, as well as in the large and small motor and sensory nerves (Figure 2) [12]. The immune response leads to a cross-reaction with peripheral nerve components because of shared epitopes resulting in acute polyneuropathy [13]. This is further supported by the identification of various antiganglioside antibodies noted in necropsy and animal models that cross-react with the ganglioside surface molecules of peripheral nerves [9,12]. This phenomenon may also explain the potential involvement of the heart, which possesses lactose-containing gangliosides.
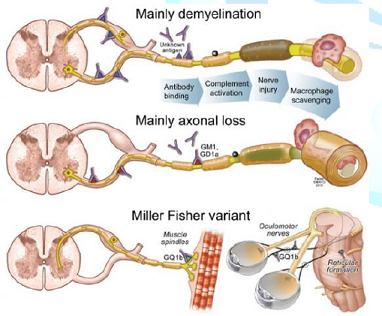
Figure 2: Guillain-Barré syndrome pathogenesis.
Different degrees of affliction of the autonomic nervous system can be seen in up to 70% of patients with the GBS [13]. Current data suggest sympathetic over activity rather than parasympathetic hypo activity in such patients [14]. It is postulated that a failure of catecholamine uptake in the “irritated” peripheral nerves may be responsible for this activity [15]. In addition, the denervated organs have been noted to be increasingly sensitive to catecholamines, resulting in denervation hypersensitivity [16].
Cardiovascular disturbances are believed to be secondary to a combination of this entity in addition to impairment of the carotid sinus reflex [17]. Cardiovascular abnormalities in the GBS have been attributed to autonomic neuropathy in the efferent fibers of the vagus nerves, and are seen in about of 70% of affected patients [18]. However, autopsy findings have not confirmed these changes [19,20]. Other cardiac complications have also been described in the patients with Guillian-Barre syndrome, mainly: heart rate variability, BP variability, cardiomyopathy, and electrocardiographic changes (Table 1) [12,21,22].
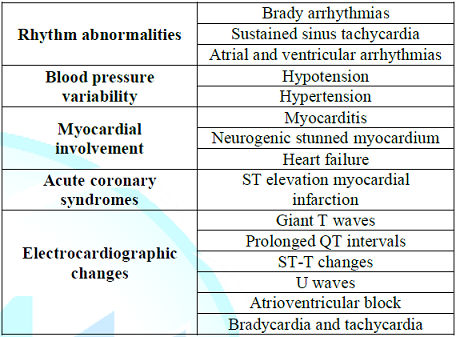
Table 1: Common cardiovascular complications of the Guillain-Barré syndrome.
Sustained sinus tachycardia is the most common abnormality. This was believed to be due to sympathetic hyperactivity [14]. Due to its transient nature, the treatment is usually supportive [23]. Other tachyarrhythmias, including atrial and ventricular arrhythmias, may also occur [22]. There have been two anecdotal reports of ventricular tachycardia and fibrillation after administration of muscle relaxants for tracheal intubation or tracheostomy [24]. There have been no other reports in the literature of spontaneous VT in patients with GBS. We know that there is a wide spectrum of main etiologies of VT like cardiomyopathies (with coronary disease), long QT and Brugada syndrome as well as adverse effects of different medications and electrolyte imbalances [25]. Our patient had known CAD with severe left ventricular dysfunction and this underlying condition may have responsible for the VT occurring during this acute illness.
Bradyarrhythmias including atrioventricular block and asystole, have been reported in 7% to 34% of patients and may occur in up to 50% of patients with GBS, and are potentially serious events necessitating the administration of atropine or pacemaker placement [14,26]. Vagal over activity caused by afferent baroreceptors reflex failure is believed to be a pathogenesis for bradycardia. Aggressive correction of associated factors such as hypoxia, medication side effects, and metabolic abnormalities may help in prevention [27]. Where severe bradycardia has been described no published consensus has been reached on whether to implant a temporary or permanent pacemaker-as recovery of these patients and infective complications are hard to predict [28].
Blood Pressure (BP) variability can be attributed to disturbances in the baroreceptor reflex pathway as well as to changes in the catecholamine levels. The dysregulation of the parasympathetic and sympathetic systems is responsible for alterations in venomotor tone and peripheral vascular resistance, most often causing transient, but in some cases, persistent hypotension. Fluctuations in BP are often considered as pathognomonic for the GBS and are likely to occur in critical illnesses or neuropathy [29].
Although these episodes of BP deviation were most often related to mechanical ventilation, analgesia, and sedation they may also occur without sedation [14]. There are no specific recommendations for target mean arterial pressure and the treatment is mainly symptomatic either with fluids and inotropes for hypotension and with IV antihypertensive therapy and/or vasodepressors for hypertension. The possible mechanisms are denervation hypersensitivity, but other contributing conditions, such as pulmonary thromboembolism, hypoxemia, sepsis, gastrointestinal bleeding, and metabolic abnormalities, need to be considered [13,30].
Myocardial involvement ranges from asymptomatic myocarditis to neurogenic stunned myocardium and heart failure. It can arise from the activation of the sympathetic nervous system, caused by catecholamine-associated myocardial injury but infectious, chemicals and hypersensitivity medications can also account for this damage [31,32]. It is possible that the extent of myocardial involvement has been underestimated as routine 2-dimensional echocardiography is not performed in this critically ill cohort of patients (including on mechanical ventilation) [33].
There are anecdotal reports of acute coronary syndromes, including ST-segment elevation myocardial infarction occurring during therapy for GBS with intravenous immunoglobulin [34]. In another report intracoronary Doppler flow measurements revealed an elevated baseline coronary flow velocity with a decreased coronary flow reserve, supposedly secondary to a catecholamine surge [33].
A wide spectrum of electrocardiographic changes have been demonstrated, including giant T waves, prolonged QT intervals, ST-T changes, U waves, and atrioventricular block, in addition to bradycardia and tachycardia as previously described [35]. These changes are also believed to be secondary to associated myocardial involvement. Along with 2-dimensional echocardiographic studies, other modalities to demonstrate cardiac involvement such as iodine-123 meta-iodobenzylguanidine myocardial scintigraphy and carbon-11 hydroxyephedrine positron emission tomography can also be used to study sympathetic innervation of myocardium [36].
Conclusions
GBS is the most common cause of acute flaccid paralysis in young adults and the elderly and an important cause of admission to intensive care units. Critically ill patients with paralysis and need for mechanical ventilation often have cardiac involvement. This ranges from variations in blood pressure to involvement of the myocardium and potentially fatal arrhythmias. A thorough review of the literature shows rare reports of a wide spectrum of cardiac abnormalities, with no reported spontaneous VT. We suggest that careful cardiac assessment of patients with GBS be performed including continuous ECG monitoring as well as measurement of cardiac enzymes and 2-D Echocardiography.
References
1. Guillain G, Barré JA and Strohl A. Sur un syndrome de radiculonévrite avec hyperalbuminose du liquide céphalo-rachidien sans réaction cellulaire: remarques sur les caractères cliniques et graphiques des reflexes tendineux (1916) Bulletins et mémoires de la Société des Médecins des Hôpitaux de Paris 40: 1462-1470.
2. Nobuhiro Y and Hartung H. Guillain–Barré Syndrome (2012) NEJM 366: 2294-2304. https://doi.org/10.1056/NEJMra1114525
3. Koeppen S, Kraywinkel K, Wessendorf TE, Ehrenfeld CE, Schürks M, et al. Long-term outcome of Guillain-Barré syndrome (2006) Neurocrit Care 5: 235-242. https://doi.org/10.1385/NCC:5:3:235
4. Teitelbaum JS and Borel CO. Respiratory dysfunction in Guillain-Barré syndrome (1994) Clin Chest Med 15: 705-714.
5. Flachenecker P. Autonomic dysfunction in Guillain-Barré syndrome and multiple sclerosis (2007) J Neurol 254: 96-101. https://doi.org/10.1007/s00415-007-2024-3
6. Hartung HP, Pollard JD, Harvey GK and Toyka KV. Immunopathogenesis and treatment of the Guillain-Barré syndrome-part I (1995) Muscle Nerve 18: 137-153. https://doi.org/10.1002/mus.880180202
7. McGrogan A, Madle GC, Seaman HE and de Vries CS. The epidemiology of Guillain-Barré syndrome worldwide. A systematic literature review (2008) Neuroepidemiology 32: 150-163. https://doi.org/10.1159/000184748
8. Jones HR Jr. Guillain-Barré syndrome: perspectives with infants and children (2000) Semin Pediatr Neurol 7: 91-102. https://doi.org/10.1053/pb.2000.6690
9. Hahn AF. Guillain-Barré syndrome (1998) Lancet 352: 635-641. https://doi.org/10.1016/S0140-6736(97)12308-X
10. Hadden RD, Cornblath DR, Hughes RA, Zielasek J, Swan AV, et al. Electrophysiological classification of Guillain-Barré syndrome: clinical associations and outcome (1998) Ann Neurol 44: 780-788. https://doi.org/10.1002/ana.410440512
11. Ho TW, Mishu B, Li CY, Gao CY, Blaser MJ, et al. Guillain-Barré syndrome in northern China: relationship to Campylobacter Jejuni infection and anti-glycolipid antibodies (1995) Brain 118: 597-605. https://doi.org/10.1093/brain/118.3.597
12. Finkelstein JS and Melek BH. Guillain-Barré syndrome as a cause of reversible cardiomyopathy (2006) Tex Heart Inst J 33: 57-59.
13. Zochodne DW. Autonomic involvement in Guillain-Barré syndrome: a review (1994) Muscle Nerve 17: 1145-1155. https://doi.org/10.1007/s10286-018-0542-y
14. Pfeiffer G, Schiller H, Kruse J and Netzer J. Indicators of dysautonomia in severe Guillain-Barré syndrome (1999) J Neurol 246: 1015-1022. https://doi.org/10.1007/s004150050506
15. Ahmad J, Kham AS and Siddiqui MA. Estimation of plasma and urinary catecholamines in Guillain-Barré syndrome (1985) Japanese J Med 24: 24-29. https://doi.org/10.2169/internalmedicine1962.24.24
16. Asahina M, Kuwabara S, Suzuki A and Hattori T. Autonomic function in demyelinating and axonal subtypes of Guillain-Barré syndrome (2002) Acta Neurol Scand 105: 44-50. https://doi.org/10.1034/j.1600-0404.2002.00099.x
17. Mitchell PL and Meilman E. The mechanism of hypertension in the Guillain-Barré syndrome (1967) Am J Med 42: 986-995. https://doi.org/10.1016/0002-9343(67)90079-4
18. Flachenecker P, Wermuth P, Hartung HP and Reiners K. Quantitative assessment of cardiovascular autonomic function in Guillain-Barré syndrome (1997) Ann Neurol 42: 171-179. https://doi.org/10.1002/ana.410420207
19. Tuck RR and McLeod JG. Autonomic dysfunction in Guillain-Barré syndrome (1981) J Neurol Neurosurg Psychiatry 44: 983-990. http://dx.doi.org/10.1136/jnnp.44.11.983
20. Bredin CP. Guillain-Barré syndrome: the unsolved cardiovascular problems (1977) Ir J Med Sci 146: 273-279. https://doi.org/10.1007/BF03030974
21. Annane D, Baudrie V, Blanc AS, Laude D, Raphaël JC, et al. Short-term variability of blood pressure and heart rate in Guillain-Barré syndrome without respiratory failure (1999) Clin Sci 96: 613-621. https://doi.org/10.1042/cs0960613
22. Mukerji S, Aloka F, Farooq M, Kassab M and Abela G. Cardiovascular Complications of the Guillain-Barré Syndrome (2009) Am J Cardiol 104: 1452-1455. https://doi.org/10.1016/j.amjcard.2009.06.069
23. Flachenecker P, Hartung HP and Reiners K. Power spectrum analysis of heart rate variability in Guillain-Barré syndrome. A longitudinal study (1997) Brain 120: 885-894. https://doi.org/10.1093/brain%2F120.10.1885
24. Graham IF. The heart in the Guillian-Barre syndrome (1984) British Medical J 288: 6411.
25. Tonelli A, Khasnis A and Abela GS. Peaked T-waves and sinus arrhythmia before prolonged sinus pauses and atrioventricular block in the Guillain-Barré Syndrome (2007) Indian Pacing Electrophysiol J 7: 249-252.
26. Kordouni M, Jibrini M and Siddiqui MA. Long-term transvenous temporary pacing with active fixation bipolar lead in the management of severe autonomic dysfunction in Miller-Fisher syndrome: a case report (2007) Int J Cardiol 117: 10-12. https://doi.org/10.1016/j.ijcard.2006.07.086
27. Hund EF, Borel CO, Cornblath DR, Hanley DF and McKhann GM. Intensive management and treatment of severe Guillain-Barré syndrome (1993) Crit Care Med 21: 433-436.
28. Wijdicks EF, Litchy WJ, Harrison BA and Gracey DR. The clinical spectrum of critical illness polyneuropathy (1994) Mayo Clin Proc 69: 955-959. http://dx.doi.org/10.1016/S0025-6196(12)61819-9
29. Ropper AH. Critical care of Guillain-Barré syndrome. In: (Ed) Ropper AH (2003) Neurological and neurosurgical intensive care. 4th ed. Philadelphia: Lippincott, Williams & Wilkins 278-298.
30. Aslam AF, Aslam AK, Vasavada BC and Khan IA. Cardiac effects of acute myelitis (2006) Int J Cardiol 111: 166 -168. https://doi.org/10.1016/j.ijcard.2005.06.018
31.Goldman MJ and Makaryus AN. Guillain-Barré syndrome complicated by myocarditis (2006) Mt Sinai J Med 73: 1126 -1128.
32. Barsheshet A, Marai I, Appel S and Zimlichman E. Acute ST elevation myocardial infarction during intravenous immunoglobulin infusion (2007) Ann N Y Acad Sci 1110: 315-318. https://doi.org/10.1196/annals.1423.033
33. Yoshii F, Kozuma R, Haida M, Shinohara Y, Yoshitake M, et al. Giant negative T waves in Guillain-Barré syndrome (2000) Acta Neurol Scand 101: 212-215. https://doi.org/10.1034/j.1600-0404.2000.101003212.x
34. Dagres N, Haude M, Baumgart D, Sack S and Erbel R. Assessment of coronary morphology and flow in a patient with Guillain-Barré syndrome and ST-segment elevation (2001) Clin Cardiol 24: 260-263. https://doi.org/10.1002/clc.4960240318
35. Münch G, Nguyen NT, Nekolla S, Ziegler S, Muzik O, et al. Evaluation of sympathetic nerve terminals with [(11)C] epinephrine and [(11)C] hydroxyephedrine and positron emission tomography (2000) Circulation 101: 516-523. https://doi.org/10.1161/01.CIR.101.5.516
36. Priori S. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death (2015) European Heart J 36: 2793-2867. https://doi.org/10.1093/eurheartj/ehv316
*Corresponding author:
Ilia Davarashvili, Jesselson Integrated Heart Center, Shaare Zedek Medical Center, the Hebrew University of Jerusalem, Jerusalem, Israel, Tel:972-2-6555320, E-mail: idavarashvili@yahoo.com
Citation:
Davarashvili I and Balkin J.Ventricular tachycardia in the guillain-barre syndrome. Cardiac complications in guillain-barre syndrome, review of the literature (2018) Clinical Cardiol Cardiovascular Med 2: 8-11.


 PDF
PDF