Hemangiomas of the
small bowel are exceedingly rare benign tumors. When diagnosed they are found
in the setting of an acute gastrointestinal bleeding, but can also be involved
in intussusception, obstruction, and cause pain. There are few reports of this
case overall, and incidental or asymptomatic cases are rare for adult patients.
Although computed tomography with angiography or tagged red blood cell scans
are pivotal for acute active bleeding, endoscopy has become pivotal in
diagnosis of lesions that are quiescent. Management is also variable as
endoscopy, laparoscopy, and open surgery have all been utilized with success.
Introduction
Small bowel tumors are rare and
clinicians generally find them when patients become symptomatic. Metastatic
lesions such as melanoma have been found to outnumber primary tumors, although
47% of those primary tumors are benign [1, 2]. The most common benign histology
includes adenoma, lipoma, leiomyoma, and hemangioma. Hemangiomas are vascular
malformations involving the proliferation of blood vessels in the submucosa and
are commonly diagnosed in infants and children. Cavernous hemangiomas present
as the dominant type, but there is also multiple phlebectasia, capillary
hemangioma and angiomatosis with gastrointestinal lesion. The incidence of
hemangioma is approximately 10% of benign small bowel tumors and only 0.3% of
all gastrointestinal tumors [3].
The frequency of benign small
bowel tumors tends to increase from the duodenum to the ileum. In a similar
pattern, most reported cases of hemangiomas have occurred in the jejunum and
ileum, therefore making duodenal lesions exceedingly rare [4]. The most common
symptom is gastrointestinal hemorrhage and anemia, which accounted for 73.2% of
small bowel hemangioma cases. Obstructive symptoms are far less frequent,
occurring at only 12.8%. Interestingly, 30% of hemangioma cases have been found
to asymptomatic [5].
Much of the discussion in the
literature concerning hemangiomas of the duodenum occurs primarily in case
reports. Treatment strategies for patients with symptoms encompass medical
intervention, endoscopic therapy and surgical resection. No established
algorithm to the management of these lesions in adults exists. We will discuss
a patient who presented to us with an
incidental, asymptomatic hemangioma of the duodenum and review the various
treatment options available in the current literature.
Case
Report
An 18-year-old female was
involved in a motor vehicle accident and was noted to have an obvious seatbelt
sign, therefore abdominal trauma was suspected on initial assessment. The
initial Computed Tomography (CT) scan revealed a lobulated mass encompassing
most of the medial wall of the 1st and 2nd portion of the duodenum. The mass
measured 3.4 by 2.4 cm and was associated with some scattered calcifications. The
initial read favored duodenal hematoma, so she was transferred to a level one
trauma center. Magnetic Retrograde Cholangiopancreatography (MRCP) revealed a
sessile, polypoid mass extending from the first to third portion of the
duodenum, which measured 6 cm in length (Figure
1). Imaging modality did not reveal any evidence of metastatic lesions or
lymphadenopathy to suggest malignancy. The abdominal pain associated with the
trauma resolved after 2 days, and she was soon discharged from the hospital
with outpatient surgical oncology follow-up.
An endoscopic ultrasound was
recommended by surgical oncology to conclusively determine if this was a
traumatic hematoma or a potential malignancy. This was performed 33 days after
the accident and it demonstrated a large mass containing a cluster of vessels
extrinsic to the walls of the duodenal bulb. The liver, bile ducts, pancreas
and regional lymph nodes appeared normal and there was no concern for malignancy.
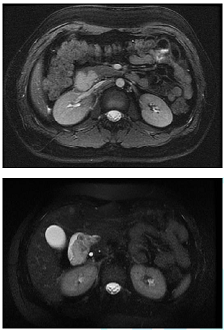
Figure 1: The images above show a sessile polypoid mass extending from the duodenal bulb into the second and third portions of the duodenum. This is isointense to the kidney on FIESTA images (top) and slightly hyper intense on T2 weighted sequences (bottom).
Biopsies were obtained and immune-histochemical
staining was consistent with a benign hemangioma, which exhibited strong
expression for CD 31 but was negative for D2-40. She denied a history of anemia
and her hemoglobin was normal at 12.7 g/dL. She remained asymptomatic so non-operative
management with yearly surveillance was recommended.
Discussion
Diagnosis of these rare tumors
can pose a challenge for clinicians, especially when the patient is
asymptomatic. Several diagnostic modalities are available, which include video
capsule endoscopy (VCE), double-balloon enteroscopy (DBE), CT scan, MRCP, CT
enterography and angiography. Angiography and technetium Tc 99m red blood cell
scanning is limited when there is no active bleeding [1]. CT enterography can
improve sensitivity to 85-97%, but endoscopy has emerged as the preferred
diagnostic modality [6]. VCE and DBE have shown the greatest impact on the
diagnosis and treatment of small bowel disease in the modern era of medicine
[7]. VCE can find significant lesions at any part of the small bowel and can be
pivotal in the work-up of gastrointestinal hemorrhage. Flexible endoscopy and
DBE also allow for tissue biopsy sampling, marking with ink tattoo and
therapeutic intervention.
Medical management has been
discussed in the pediatric literature available on this topic. Beta blockers
and other topical agents have an established role for cutaneous hemangiomas. When
beta blockers are not tolerated, prednisolone dosed at 3-10 mg/kg is provided
for 6-8 weeks [8]. Interferon alpha (IFN-α) administered as a subcutaneous
injection has found a role in treating steroid resistant, life threatening
cavernous hemangiomas. The indications for IFN-α include life threatening
lesions that are pulmonary, hepatic, and gastrointestinal and those causing
consumptive coagulopathy [9]. Interventional radiology can assist utilizing
embolization techniques, which rely on the rich collateralization of blood flow
to this region. It is unclear if medical management or percutaneous embolization
has a role in the treatment of this disease in adults.
Endoscopy has paved a new path in
both the diagnosis and treatment of gastrointestinal lesions. Endoscopic Mucosal
Resection (EMR) and ablative therapies have become a staple in the treatment of
dysplastic lesions of the gastrointestinal tract. EMR involves injecting saline
into the submucosal place under direct endoscopic visualization, followed by
snare resection of the lesion. Nishiyama et al. explored the role of EMR in the
treatment of duodenal hemangiomas. They developed their own criteria based on
accessibility, size of 2cm or less, and absence of large blood vessels within
the lesion. Esophagogastroduodenoscopy (EGD) followed by CT angiography was performed
to characterize a 2 cm lesion without large vessels along the superior duodenal
angle. This was successfully resected using EMR with excellent hemostasis [10].
Although technically demanding,
there has been successful laparoscopic resection of duodenal hemangioma. A few
reports of hand-assisted laparoscopic resections are documented but have been
faulted for poor visualization. Kanaji et al. reported one of the few totally
laparoscopic resections while employing endoscopy, termed as laparoscopic and
endoscopic cooperative surgery. The author documented the successful resection
of a 2 cm hemangioma found in the third portion of the duodenum and the mid
jejunum. The two key portions of the duodenal resection are the laparoscopic
Kocher maneuver and the EGD to visualize the lesion from the intraluminal side.
The laparo-endoscopic view enabled resection of the duodenal tumor with
suitable margins through visualization of both the mucosal and serosal sides of
the tumor [3].
Open surgery is a preferred
approach over laparoscopy when patients cannot tolerate pneumo-insufflation,
have significant adhesive disease or have acute bleeding with hemodynamic
instability. This approach can be augmented with intraoperative endoscopy,
generally performed for those with gastrointestinal bleeding and no
identifiable source as a last effort. The current shift towards minimally
invasive techniques mentioned previously has been explored to circumvent the
increased hospital stay, postoperative ileus and morbidity of open surgery
[10].
Conclusion
A hemangioma of the duodenum was
incidentally discovered on CT, which has remained asymptomatic to date. The
management plan consists of yearly surveillance with MRI imaging. Multiple
therapeutic options have been discussed in this article. If our patient becomes
clinically symptomatic, she may be a candidate for endoscopic or surgical
resection. A more aggressive approach such as pancreaticoduodenectomy may be
necessary if malignancy becomes a concern. Future management of these tumors
will include optimization of surveillance with continued improvements in
minimally invasive resection leading to decreased morbidity with improvement in
quality of life.
References
1.
Harris
Jennifer W and B Mark Evers. “Small Intestine.” Sabiston Textbook of Surgery,
edited by Courtney Townsend, 20th ed (2016) Saunders 1237-1295.
2.
Eckel
JH. Primary tumors of the jejunum and ileum (1948) Surgery 23: 467-475.
3.
Kanaji
S, Tetsu Nakamura, Masayasu Nishi, Masashi Yamamoto, Kiyonori Kanemitu, et al.
Laparoscopic partial resection for hemangioma in the third portion of the
duodenum (2014) World J Gastroenterol 20: 12341-12345. https://dx.doi.org/10.3748%2Fwjg.v20.i34.12341
4.
Wilson
JM, Melvin DB, Gray G and Thorbjarnarson B. Benign small bowel tumor (1975) Ann
Surg 181: 247-250.
5.
Nader
PR and Margolin F. Hemangioma causing gastrointestinal bleeding. Case report
and review of the literature (1966) Am J Dis Child 111: 215-222.
doi:10.1001/archpedi.1966.02090050147015
6.
Pilleul
F, Penigaud M, Milot L, Saurin JC, Chayvialle JA, et al. Possible Small-Bowel
Neoplasms: Contrast-Enhanced and Water-Enhanced Multidetector CT Enteroclysis
(2006) Radiology 241: 796. https://doi.org/10.1148/radiol.2413051429
7.
Takase
N, Fukui K, Tani T, Nishimura T, Tanaka T, et al. Preoperative detection and
localization of small bowel hemangioma: Two case reports (2017) World J
Gastroenterol 23: 3752-3757. https://doi.org/10.3748/wjg.v23.i20.3752
8.
A
Chattopadhyay, Kumar V, Maruliah M and Rao PL. Duodenojejunal obstruction by a
hemangioma (2002) Pediatr Surg Int 18: 501-502. https://doi.org/10.1007/s00383-002-0838-8
9.
R Alan B, Ezekowitz and Folkman J. Interferon
Alfa-2a Therapy for Life Threatening Hemangiomas of Infancy (1992) N Engl J Med
326: 1456-1463. https://doi.org/10.1056/NEJM199205283262203
10.
Nishiyama
N, Mori H, Kobara H, Fujihara S, Nomura T, et al. Bleeding duodenal hemangioma:
Morphological changes and endoscopic mucosal resection (2012) World J Gastroenterol
18: 2872-2876. https://doi.org/10.3748/wjg.v18.i22.2872



