Introduction
In recent years,
new concepts have emerged that have led to changes in the way the sick
individual and the context of his disease are approached. This began with a
broader approach to the disease process, which in some way lost its status as
an inalienable state that can only partially and temporarily be remedied. The
disease has thus become a dynamic phenomenon
involved in a succession of pathological states more or less reversible
depending on their greater or lesser interactivity. In that sense, it has been
recently written by DG Limbaugh that one can conceive of cases where
simultaneous disorders prevent each other from being, in any traditional sense,
actually harmful [1]. On even broader
bases, the concept of One Health has developed in recent years, which requires
breaking down the interdisciplinary barriers that still separate human and veterinary medicine from ecological,
evolutionary, and environmental sciences [2]. On the same way appeared the
concepts of personalized medicine, precision medicine, integrative medicine or
P4 medicine, all interconnected by the concept of globality and interactivity
and using increasingly powerful tools. It should be noted at the outset that
all these concepts, which are relatively close to the others, share the same
concern, which is to take on as effectively as possible the major challenge
represented by the exponential development of chronic diseases throughout the
world. In 2011, we could find following definition of personalized medicine in
U.S. News: Personalized medicine is a young but rapidly advancing field
of healthcare that is informed by each persons unique clinical, genetic,
genomic, and environmental information. Because these factors are different for
every person, the nature of diseases—including their onset, their course, and
how they might respond to drugs or other interventions—is as individual as the
people who have them (Figure 1).
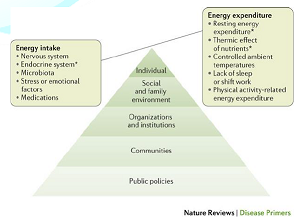
Figure 1: Key factors involved in the regulation of energy balance.
Personalized
medicine is about making the treatment as individualized as the disease. It involves identifying genetic, genomic, and clinical information that allows
accurate predictions to be made about a persons susceptibility of developing
disease, the course of disease, and its response to treatment [3-5] (Figure 2).
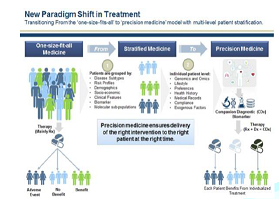
Figure 2: Frost and Sullivan: new paradigm shift in treatment.
In order for
personalized medicine to be used effectively by healthcare providers and their
patients, these findings must be translated into precise diagnostic tests and targeted therapies (Figure 3). Specific advantages that personalized medicine may
offer patients and clinicians include:
•
Ability
to make more informed medical decisions
•
Higher
probability of desired outcomes thanks to better-targeted therapies
•
Reduced
healthcare costs
•
Reduced
probability of negative side effects
•
Focus
on prevention and prediction of disease rather than reaction to it
•
Earlier
disease intervention than what has been possible in the past
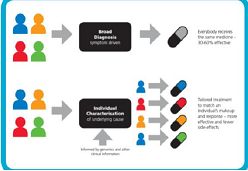
Figure 3: Personalized medicine and treatment, NHS explanation document.
The four Ps of
personalized medicine have thus given birth to P4 medicine, a term coined by
David Galas and Leroy Hood from the Institute for Systems Biology (ISB) in
Seattle, and based on four references: Predictive, Preventive, Personalized and
Participatory [6,7]. The conceptual matrix at the origin of all these
evolutions of medical thinking remains this of systems biology with its central
components that are genetically programmed networks (circuits) within cells and
networks of cells [8].
Prediction and prevention of
disease
Using genomic
technologies and other diagnostics, we will be able to identify people most at
risk of disease even before the onset of their symptoms. Earlier detection will
open up the prospect of new treatment options and support people to make
informed lifestyle choices.
More precise
diagnoses thanks to the knowledge of each individuals complex molecular and
cellular processes, informed by other clinical and diagnostic information
define the so-called Precision Medicine [9] (Figure 4).
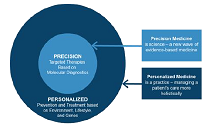
Figure 4: Personalized and precision medicine.
Targeted and personalized
interventions
We are already
beginning to see the development of simple point of care tests, based on
genomic knowledge, which enable clinicians in a wide variety of settings to
identify the best therapy. Variants in our genetic code can also be used to
predict the potential for adverse drug reactions [10]. The ability for a
clinician to discuss with their patients about information on individual genomic characteristics, lifestyle and
environmental factors, and interpret personal data from wearable technology
will drive a new type of conversation, that might lead patients to consider
preventive measures with the idea that patients should play a decisive role in
their own healthcare by actively controlling their health status and
participating in the decision-making process regarding their treatments [11].
Material and Methods
The Bio
Immune(G)ene Medicine, so-called BI(G)MED, is first and foremost a method
combining predictive diagnosis as well as biomimetic and personalized treatment
(Figure 5). To try to achieve this
objective, there is currently a very fruitful concept, that of systems biology,
aiming at a global approach of biological phenomena.
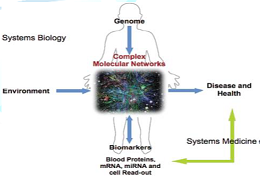
Figure 5: Understanding the complex molecular network is the center of systems biology and systems medicine [12].
Systems biology
employs a holistic approach to study all components and interactions in the
network of DNA (genes), RNA, proteins and biochemical reactions within a cell
or an organism. This concept of systems biology corresponds entirely to the
approach of human diseases that we have been using for many years in the
framework of BI(G)MED, and that we are
able to translate at both the diagnostic and therapeutic level. At the
diagnostic level, we have a very efficient overview of the cellular immune
system thanks to the lymphocyte phenotyping, which is not only an important
tool for the diagnosis of hematological and
immunological disorders, but also a means of fine-tuning cellular immune status
and drawing predictive conclusions [12-14] (Figure
6).
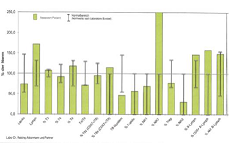
Figure 6: Example of a lymphocyte phenotyping.
To appreciate the humoral mediated immune system,
we use the protein profile, which allows us to measure several types of humoral
immune responses that are both complementary and interactive [15] (Figure 7).
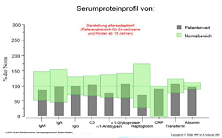
Figure 7: Example of a protein profile.
Thanks to these
two types of investigations, which are essential for us, we can direct the
biological diagnosis more specifically towards parameters related to
microbiology, oncology, allergy, autoimmunity or metabolism (Figure 8). The therapeutic goal of the
BI(G)MED is therefore to make the best use of all the molecular resources
available in the cell to restore its homeostasis at the different genomic,
epigenomic, transcriptomic, proteomic and metabolomic levels [16].
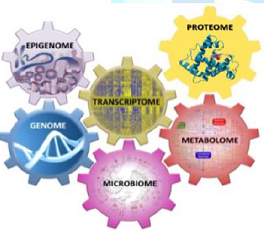
Figure 8: Systems biology of asthma and allergic diseases: a multiscale approach [16].
To achieve this
goal, it is necessary to use means borrowed to nanobiotechnologies,
to ensure a very efficient transport of molecules made available to the cells,
so as to facilitate their self-regulation through a truly biomimetic treatment.
Usually the BI(G)MED uses nanovectors with a xylitol base [17], carrying
molecules of all types at ultra-low concentrations (1g x10-4 until
1g x10-10 Mol). These small globules must be dissolved in the oral
cavity as in all sub-lingual
immunotherapies, from where the molecular information will be transmitted
to the target organ via the lymphatic network. These molecules can be fragments
of DNA and RNA (among these will be found mainly microRNAs as epigenetic
regulators), transcription factors, all kinds of molecules involved in the
various cell signalling pathways, but also cytokines and enzymes such as
kinases. All together are included in formulas in ultra-low doses [18]; all are
therefore at concentrations similar to those observed in the cell physiology,
making them unlikely to trigger unwanted side reactions. In this way we
can progressively regulate most of the disturbed molecular pathways in one or
more given pathologies according to the general rule of our kind of treatment:
from the gene to the whole cell [19,20].
Results
The results
obtained with this method will be presented through three case reports.
Primary Biliary Cirrhosis (PBC)
and Systemic Sclerosis (SSc)
This is a
67-year-old woman at the time of her first consultation in 2003, by which a Primary
Biliary Cirrhosis (PBC) and a Systemic Sclerosis (SSc) were recently discovered
on the occasion of pericarditis associated with pleural effusion. At this time,
biology shows the usual abnormalities for this type of pathology, namely
increased antinuclear and in particular anti-centromere antibodies; it is the
same with regard to anti-mitochondrial antibodies. It is also interesting to
note the viral reactivations concerning EBV, CMV and Parvovirus B19, which will
follow the patient in a very irregular way during all the coming years. The diagnosis is
confirmed by tissue biopsies, and the patient wants to stop the immediate
corticosteroid therapy, while agreeing to continue a treatment with ursodeoxycholic acid, which she never
interrupted. For this reason, we initiated a treatment with BI(G)MED
essentially oriented towards the regulation of the autoimmunity processes and
the neutralization of the various reactivated viruses mentioned above. The
clinical evolution has shown a remarkable stability even if it has been
complicated by various accidents such as:
· The
early development of a xerostomia, itself causing
recurrent problems of periodontal diseases and dental infections at the origin
of numerous local treatments
· The
accidental discovery in 2010 of a ductal adenocarcinoma of the left breast
requiring a lumpectomy followed by the establishment of an appropriate
BI(G)MED-immunogenetic regulation
· The
sudden appearance in 2014 of an autoimmune thrombocytopenia following osteosynthesis after traumatic wrist fracture and
needing the establishment of an additional regulatory treatment to stabilize
platelets between 30 and 40 thousand
· The
occurrence in 2017 of several episodes of bacterial pulmonary infections which
each time required the use of appropriate antibiotic therapy
Despite all these
health disorders, that adjustments at the level of the BI(G)MED nanotherapy
have each time contributed to rebalance, the patient has preserved until now a
very stable condition and a biological state hardly evolutive, where the
various parameters alternately normalize and then degrade again more or less.
PBC and SSc remained perfectly stabilized during all these years.
Waldenströms macroglobulinemia
The second
example is that of a 53-year-old woman at the time of her first consultation in
2012, by which a Waldenströms macroglobulinemia was discovered a few months ago
in the course of investigations carried out following multiple recurrent ENT
(Ear, Nose and Throat) infections.The first
biological parameters corroborate a triple monoclonal peak estimated at 13.42
g/l and including:
•
IgG
kappa
•
IgA
kappa
•
IgM
kappa
associated
with a positive Bence-Jones proteinuria and an increased value of thymidine
kinase by 10,2 U/L. After approximately eighteen months of a BI(G)MED
sublingual regulatory nanotherapy, the triple monoclonal peak had decreased to
3.3 g/L, Bence-Jones proteinuria had disappeared, and thymidine kinase was
normal to 5.0 U/L. In addition, the patient had not had the least ENT
infections during the previous winter. From that time on, no pejorative
developments have been reported.
Waldenström disease and sclerosing
cholangitis
A third and final
example concerns a 55-year-old man at his first consultation for Waldenström
disease and sclerosing cholangitis discovered the previous year. Otherwise an
ulcerative colitis has been known since the age of 22 and is regularly treated
by mesalazine. At this time, its
clinical condition is not bad, but the biological parameters show the typical
disturbances of the diagnosed diseases with:
· Increased
levels of liver enzymes
· IgM
greatly increased
· Positive
p-ANCA
There is also a
reactivation of Parvovirus B19. The patient followed for a year and a half a
regulatory treatment of BI(G)MED nanotherapy while continuing his regular
intake (but at a very reduced dosage) of mesalazine and ursodesoxycholic acid,
and results in biological parameters almost normalized among those initially
disturbed.
Discussion
This brief
description of three clinical examples drawn from a daily medical practice make
it possible to demonstrate the therapeutic interest and effectiveness of
resorting to the regulating potential of the cell itself, the only one able to
rebalance a disturbed mode of functioning, when the needed molecular
information is provided to it. It turns out that this regulatory capacity is as
greater as the molecular resources used by the cell for this purpose of global
regulation are made available by means of ultra-low doses of nanotherapeutic
compounds administered sublingually. This observation does not exclude that
other types of nanocarriers could be considered in the near future.
Conclusion
1.
Different
medical concepts have emerged in recent years all based on globality and
interactivity
2.
Personalized
medicine (also named precision medicine) individualizes the disease and
consequently the treatment that must regulate it
3.
The
best way to achieve this goal is probably to use the molecular resources of the
cell itself, and direct them towards a return to cellular homeostasis through a
regulatory treatment.
4.
The
BI(G)MED is suitable for this regulatory process in the form of a sublingual
nanotherapy, whose efficiency is confirmed in very variable clinical
situations.
References
1.
Limbaugh
DG. The harm of medical disorder as harm in the damage sense (2019) Theor Med
Bioeth 40: 1-19.
https://doi.org/10.1007/s11017-019-09483-y
2.
Garzón
DD, Jerome Boissier, Yann
Voituron, Patrick
Mavingui, Priscilla Duboz, et al. The one health concept: 10 years old and a
long road ahead (2018) Front Vet Sci 12: 5-14. https://doi.org/10.3389/fvets.2018.00014
3.
Di
Sanzo M, Marina Borro, Alessandro Santurro, Maurizio Simmaco, Vittorio
Fineschi, et al. Clinical applications of personalized medicine: a new paradigm
and challenge (2017) Curr Pharm Biotechnol 18: 194-203.
https://doi.org/10.2174/1389201018666170224105600
4.
Salari
P and Larijani B. Ethical issues surrounding personalized medicine: A
literature review (2017) Acta Med Iran 55:209-217.
5.
Anaya
JM, Duarte RC, Sarmiento C Juan, David Bardey, John Castiblanco, et al. personalized
medicine. Closing the gap between knowledge and clinical practice (2016)
Autoimmun Rev 15: 833-842.
https://doi.org/10.1016/j.autrev.2016.06.005
6.
Bengoechea
JA. Infection systems biology: from reactive to proactive (P4) medicine (2012)
Int Microbiol 15: 55-60.
7.
Sagner
M, Amy
McNeil, Pekka Puska, Nathan D Price, Elissa Epel, et al. The P4 health spectrum
- a predictive, preventive, personalized and participatory continuum for
promoting health span (2017) Prog Cardiovasc Dis 59: 506-521. https://doi.org/10.1016/j.pcad.2016.08.002
8.
Heath
JR, Phelps ME and Hood L. NanoSystems biology (2003) Mol Imaging Biol 5: 312-325.
9.
König
IR, Oliver Fuchs, Gesine Hansen, Matthias V Kopp and Matthias V Kopp. What is
precision medicine? (2017) Eur Respir J 50. https://doi.org/10.1183/13993003.00391-2017
10.
Ginsburg
GS and Phillips KA. Precision medicine: From science to value (2018) Health Aff
(Millwood) 37: 694-701. https://doi.org/10.1377/hlthaff.2017.1624
11.
Gameiro
GR, Sinkunas V, Liguori GR and Auler-Júnior JOC. Precision Medicine: Changing
the way we think about healthcare (2018) Clinics (Sao Paulo) 73: e723.
https://doi.org/10.6061/clinics/2017/e723
12.
Wang
K, Lee I, Galas D, Hood
L and Carlson G. Systems biology and the discovery of diagnostic biomarkers
(2010) Dis Markers 28: 199-207. https://doi.org/10.3233/DMA-2010-0697
13.
Caberlotto
L and Lauria M. Systems biology meets -omic technologies: novel approaches to
biomarker discovery and companion diagnostic development (2015) Expert Rev Mol
Diagn 15: 255-265.
https://doi.org/10.1586/14737159.2015.975214
14.
Szczepański
T, van der Velden VH and van Dongen JJ. Flow-cytometric immunophenotyping of
normal and malignant lymphocytes (2006) Clin Chem Lab Med 44: 775-796.
15.
Dupond
JL, Gil H, Giraudet P, Gibey R, Desmurs H, et al. Value of the protein profile
in unexplained inflammatory syndromes (1997) Rev Med Interne 18: 367-372.
16.
Bunyavanich
S and Schadt EE. Systems biology of asthma and allergic diseases: a multiscale approach
(2015) J Allergy Clin Immunol 135: 31-42.
https://doi.org/10.1016/j.jaci.2014.10.015
17.
Ur-Rehman
S, Anjum MM, Amir
Jamil, Zahoor T and Mushtaq Z. Xylitol: a review on bioproduction, application,
health benefits, and related safety issues (2015) Crit Rev Food Sci Nutr 55: 1514-1528.
https://doi.org/10.1080/10408398.2012.702288
18.
Muller
DA, Fernando GJP, Owens NS, Yeboah CA, Wei JCJ, et al. High-density
microprojection array delivery to rat skin of low doses of trivalent inactivated
poliovirus vaccine elicits potent neutralising antibody responses (2017) Sci
Rep 7: 12644. https://doi.org/10.1038/s41598-017-13011-0
19.
Marzotto
M, Debora Olioso, Maurizio Brizzi, Paola Tononi and Cristofoletti M, et al.
Extreme sensitivity of gene expression in human SH-SY5Y neurocytes to ultra-low
doses of Gelsemium sempervirens (2014) BMC Complement Altern Med 14: 104. https://doi.org/10.1186/1472-6882-14-104
20.
Olioso
D, Marzotto M, Moratti E, Brizzi M and Bellavite P. Effects of gelsemium
sempervirens L. on pathway-focused gene expression profiling in neuronal cells
(2014) J Ethnopharmacol 153: 535-539.
https://doi.org/10.1016/j.jep.2014.02.048
*Corresponding author:
Glady
Gilbert, President
of European Bio Immune(G)ene Medicine Association (EBMA), 29 Rue Charles
Sandherr 68000 Colmar, France, Tel: 0033 635 562 148, E-mail: info@ebma-europe.com
Citation:
Gilbert G. The bio immune(g)ene
medicine or how to use a maximum of molecular resources of the cell for
therapeutic purposes (2019) Edelweiss Appli Sci Tech 3: 26-29.










