Introduction
Corrosion
is a noticeable phenomenon regarding the metal manufacturing and applications
of such metals. The metallic corrosion is usually
modified by the corrosive causing properties of some natural compounds [1]. Mineral oil is a mixture of
hydrocarbons since it is composed with some vehement corrosive compounds such as
elemental sulfur, active sulfur compounds, organic acids and salts [2-6].
Usually the corrosion is a term which is defined as the destruction of a
material due to the chemical of electrochemical process as results of the
interaction between such material and the surrounded environment also its found
as a chemical oxidation process for the metallic corrosion since the metallic
corrosion can be further categorized into several corrosion types such as
general corrosion or rust, pitting corrosion, galvanic corrosion and stress
corrosion including some specific features for each sub category of corrosion
[1-5]. The surrounded environment plays a major role in the cause of any
corrosion on such metal through creating a corrosive friendly background. The
essential factor for the formation of the corrosion the relevant metal need to
expose to both water and oxygen containing environment or another oxidizing
agent which is stronger than Fe2+ while the improving the process by the acids
and salts presence in the surrounding environment [1-9]. The
impacts of such corrosive properties of mineral oils on the corrosion of some
specific metal and nature of metallic corrosion due to the effects of several
chemical compounds have been investigated and interpreted much important
results from most of previous chemical and petroleum engineering researches
regarding both whilom and recent studies.
In
the current research there were aspired to speculate the performances of stainless steels against the
mineral oils when comparing with other metals, the contribution of such
corrosive properties of mineral oils together in the causing of the metallic
corrosion at general environmental conditions, the visible appearances of the
formed corrosion compounds on the metal surfaces due to the interaction between
the mineral oils and such metals and the variations of the surfaces properties
of metal due to the formation of the corrosion such as the hardness.
Materials and
Methodology
The
selected ferrous metals and their applications in the mineral oil industry have
been shortlisted in the Table 1.
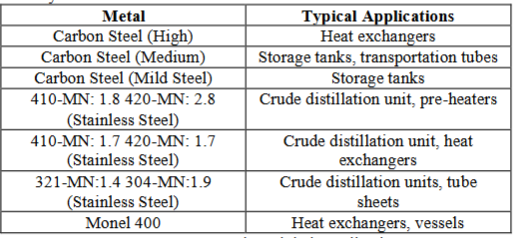
Table 1: Ferrous metals
and their applications.
The
chemical compositions of each type of metal was detected by the XRF detector
and according to the working principles of XRF detector able to detect the all
metallic elements composed in the crowing metal and some of nonmetals excluding
carbon as a percentage.
According
to the usage types of mineral oils there were selected two different types of
mineral oils namely as Murban and Das Blend. Das Blend is known as a “sour”
mineral oil or “sour” crude oil because of the relatively high sulfur content
of Das Blend and Murban is a mediocre crude oil or mineral oil which is used in
several refineries in the world. Those crude oils may slightly or extensively
different in their chemical compositions. The elemental sulfur contents,
mercaptans sulfur contents, acidity and salt contents of both crude oils were
investigated because of the corrosive tendency of such compounds. The standard
test methodologies for the testing of corrosive properties in both mineral oils
have been summarized in the Table 2.
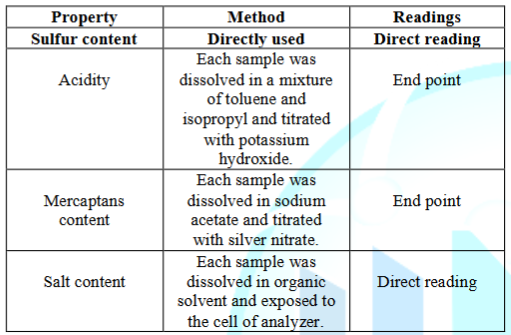
Table 2: Test
methodologies for the corrosive properties of mineral oils.
A
set of similar sized metal coupons was prepared from each type of metals as six
metal coupons from each type of metal and the dimensions and weights of such
metal coupons were measured according the requirements of mathematical
calculations. Those metal coupons were immersed
separately and homogeneously by considering both the mineral oil and the type
of metal as shown in the Figure 1.

Figure
1:
Metal coupons and apparatus setup.
After
15 days from the immersion one metal coupon was taken out from each mineral oil
container as representing both metal type and mineral oil and the corrosion
rates of such metal coupons were determined by the relative weight loss method
[10]. The same procedure was repeated for another two sets of metal coupons
after 30 and 45 days from the immersion. The corroded surfaces were observed by
the 400X of an optical microscope and the corroded metal surfaces were cleaned
by sand papers and isooctane to determine the weight losses of such metal
coupons due to the corrosion. The
mathematical expression and the terms of that expression have been given in the
below.
CR=W*k/(D*A*t)…………….(1)
Where
W = weight loss
in grams
k = constant
(22,300)
D = metal density
in g/cm3
A = area of metal
piece (inch2)
t = time (days)
CR= Corrosion
rate of metal piece
The
microscopic analysis was performed as an identification stage of the formation
of the corrosion and partial requirement of the weight loss methods to make
free the metallic surface from corroded particles. The surface of
each metal coupon was observed under the 400X lens of a laboratory optical
microscope before immersion in the mineral oils, after taking out from the
mineral oils and while determining the corrosion rate of such metal coupon as
necessity.
Based
on the observations of invisible weigh losses of some of metal coupons and as a
confirmation stage of the formations of the metallic corrosion the decayed
ferrous concentrations into crude oil samples which were exposed to the carbon
steels and stainless steels also the decayed copper concentrations into crude
oil samples which were interacted with Monel metal coupons were tested by the Atomic Absorption Spectroscopy (AAS). According
to the methodology of the sample preparation 1 ml of each crude oil sample was
diluted with 9 ml of 2-proponol and filtered.
Finally
the deductions of the initial hardness of metal coupons due to the corrosion
were tested by the Vickers hardness tester as a detection stage of the surface
changes of metal coupons due to the corrosion. The initial hardness and the
hardness after formation of the corrosion on the surface were tested in each
metal coupon. According to the test methodologies of the Vickers hardness
tester hardness of at least three points on the metal surface were tested at
once and the average value was interpreted the hardness of such metal at that
moment.
Results and
Discussion
According
to the results of the XRF detector the elemental compositions of the ferrous
metals are given in the Table 3.
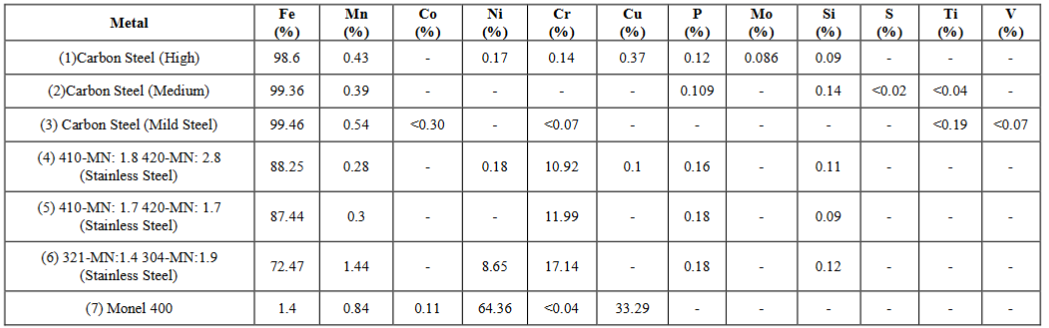
Table 3: Chemical
compositions of ferrous metals.
According
to the obtained results for the chemical compositions of the ferrous metals
higher ferrous concentrations from carbon steels, moderate ferrous
concentrations form stainless steels and a trace amount of ferrous were found.
In addition some other metals were found in trace amounts especially in
stainless steels such as nickel and copper. The major objectives of the doping
of some trace metals and non-metal with some ferrous metals are the
enhancements of the strength, hardness and the throttling of the corrosive tendency
of such ferrous metals [1,3]. The prominent and efficacy phenomenon regarding
the stainless steels is that the ability of the formation of self- corrosive
protection layer on the metal itself against any kind of corrosive environment
when having a chemical composition of at least 12% of chromium and sufficient
amount of nickel [1,3,5].
The
results for the analysis of the corrosive properties of both Murban and Das Blend mineral oils are summarized
in the Table 4.
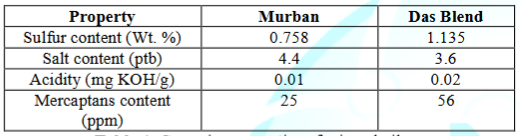
Table 4: Corrosive
properties of mineral oils.
Above
results showed relatively higher contents of both elemental sulfur and
Mercaptans in Das Blend crude oils than the Murban crude oil approximately in
twice as defined the Das Blend as a “sour” crude oil or more than that. Usually
the due to the reaction between elemental sulfur and water presence in crude
oils the metallic corrosion is happened and the process is known as in term
“localized corrosion” and the reaction between the active sulfur compounds
which are having fraction or some active functional group and metals is known
as the process of “sulfidation” [2,6]. Most of active sulfur compounds are
corrosive compounds foremost of Mercaptans which is having a formula of “RSH”.
The typical temperature for the consummate “localized” reaction is
approximately 80C and for the “sulfidation” process of corrosion it may be
approached at about 230C [8]. The general reactions regarding the formations of
the corrosion due to above processes have been given in the below.
S8(s) + 8 H2O (l) → 6 H2S (aq) + 2 H2SO4……… (2)
8Fe +S8→FeS…………….. (3)
According
to the acidity results of both mineral oils Das Blend was consisted with higher
amount of organic acids than the Murban. Organic acids are strong corrosive
causing agent that fond as organic forms in most of crude oils which are
defined under the general formula of “RCOOH” [2]. Acidity is an indicator
regarding a mineral
oil
that gives an idea about the presence of both organic acids and naphthenic
acids in such mineral oil [9]. The general reactions of the cause of the
metallic corrosion are given in the below.
Fe+2RCOOH→Fe(RCOO)2+H2…….……. (4)
FeS+2RCOOH→Fe(RCOO)2+H2S…………. (5)
Fe(COOR)2 + H2S →FeS + 2 RCOOH……… (6)
According
to the salt contents in both crude oils Murban crude oil was composed in
relatively higher amount of organic salts than Das Blend crude oil. The
summation of the CaCl2, MgCl2 and NaCl contents of some crude oil is known as
the total salt content in such crude oil. During increasing the temperature of
the mineral oils such salts tend to be broken into HCl molecules approximately
1000C although such HCl molecules do not behave as corrosive compounds at this
stage [7]. The general reaction for the initiation of such process is given in
the below.
CaCl2 + H2O →CaO + 2HCl………..……. (7)
While
reducing the temperature of the entire system some of HCl molecules tend to react
with the water or moisture presence in the medium and produce hydrochloric
acids and hydrogen sulfide. This is more prone process to the formation of the
metallic corrosion as given in the below.
HCl(g)+H2O →HCl(aq)……………….… (8)
HCl + Fe→ FeCl2 + H2 …………….…… (9)
FeCl2 + H2S FeS + 2HCl………………….. (10)
FeS
is the major corrosion compound that tend to be formed under this process and
there might have a severe effect of hydrogen sulfide on the metallic corrosion
although it is subtle to clarify because hydrogen sulfide is a gas and easy to
evolve with having too short retention time period in the relevant mineral oil
[2,4].
The
average corrosion rates of each metal with respect to both Murban and Das Blend
mineral oils have been interpreted in the Figure
2.
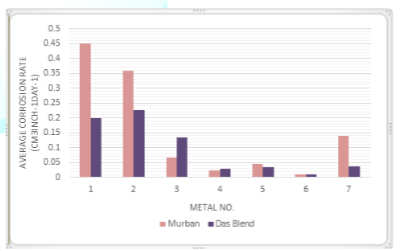
Figure 2: Average
corrosion rates of metal coupons in both mineral oils.
The
interpretation of the average corrosion rates of metal coupons is emphasized
the relatively higher corrosion rates from carbon steels with respect to both
Murban and Das Blend mineral oils, excessively lower corrosion rates form
stainless steels with respect to both mineral oils and intermediate corrosion
rates from Monel metal in both Murban and Das Blend mineral oils. The least
corrosion rates were found form 321-MN: 1.4 304 - MN: 1.9 (Stainless Steel) in
both Murban and Das Blend mineral oils. By referring the chemical compositions
of stainless steels which were used in this experiment 321-MN: 1.4 304-MN: 1.9
(Stainless Steel) was composed ~18% of chromium and ~8% of nickel which is
quite better chemical composition for the formation of the self-corrosive
protection film on the metal surface because the
minimum recommended chromium amount for that is ~12% and also sufficient amount
of nickel [1,4,5]. The 410-MN: 1.7 420-MN: 1.7 (Stainless Steel) was composed
~12% of chromium and lack of nickel since it showed the maximum corrosion rates
in both mineral oils among stainless steels. The 410-MN: 1.8 420-MN: 2.8
(Stainless Steel) was composed ~11% of chromium and ~0.2% of nickel also showed
higher corrosion rates in both mineral oils than 321-MN: 1.4 304-MN: 1.9
(Stainless Steel). Therefore it can be concluded the necessity of the minimum
recommended amount of chromium or more than that with sufficient amount of
nickel to obtain the maximum benefits from the self-corrosive protection film
against the corrosive environment.
When
comparing the corrosion rates of metal coupons with respect to both mineral
oils four types of metals showed higher corrosion rates in Murban since other
three metal types were showing higher corrosion rates in Das Blend and entirely
the distribution was an asymmetric distribution. In the explanation it can be
emphasize the improper progress of the “localization” and “sulfidation” regarding
the elemental sulfur and Mercaptans due to the requirements of higher
temperatures because the current research was conducted under the normal
environmental conditions. The effect of the salts on the metallic corrosion
might be stronger than the effects of organic acids on the metallic corrosion
according to the obtained results because Murban has higher salt content since
Das Blend has higher acidity [2,8].
The
variations of the corrosion rates of metal coupons with the exposure time
period against both Murban and Das Blend mineral oils have been shown in order
of Figure 3 and Figure 4.
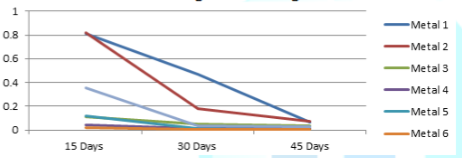
Figure 3: Variations of
the corrosion rates of metal coupons with respect to the exposure time period
in Murban.
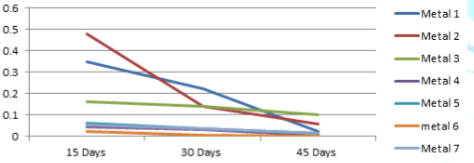
Figure
4: Variations of the corrosion rates of metal coupons with respect to the
exposure time period in Das Blend.
The
variations of the corrosion rates of metal coupons with the exposure time
period rudimentary there can be identified similar variations of the corrosion
rate regarding most of metal types also prove the inversely proportional
relationship between the corrosion rate and the exposure time period as
mentioned in the formula of the weight loss method [10]. According to the
microscopic analysis of the corroded surfaces of metal coupons some distinctive
features have been highlighted in the Figure
5.

Figure 5: Corroded
surfaces of (a) 410-MN: 1.8 420 - MN: 2.8 (stainless steel) in Murban and (b)
carbon steel (mild steel) in Das Blend.
The
analysis of such microscopic observations are based on the visible appearances
of such corrosion compounds foremost the color and surface properties [1,3].
The general features of the visible appearances have been summarized in the Table 5.
·
Ferrous
Sulfides/ Copper Sulfides
·
Ferrous
Oxides
·
Pitting
Corrosion
·
Corrosion
Cracks

Table 5: Visible
appearances of corrosion compounds.
Regarding
the most of observations there were identified the formations of FeS according
to the visible appearances of FeS. Especially regarding stainless steels some
corrosion cracks and pitting corrosion phenomenon were observed apart from the
formation of Fes. The formations of Fe2O3 were identified in seldom regarding
some of carbon steels. There were observed the corrosion compounds on the
surfaces of Monel metal which were much similar to the visible appearances of
FeS [1,3]. By referring only such visible appearances foremost the blackish
blue color it can be predicted as the CuS temporarily until an advanced
compositional analysis of such chemical compounds.
The
decayed ferrous concentrations of mineral oil samples which were interacted
with carbon steels, stainless steels and the decayed copper concentrations of
mineral oil samples which were interacted with Monel metal have been
interpreted in order of Figure 6 and Figure 7.
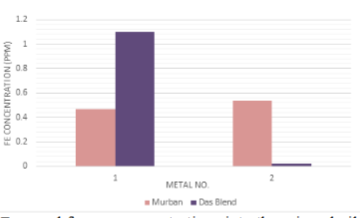
Figure 6: Decayed ferrous
concentrations into the mineral oil samples.

Figure 7: Decayed copper
concentrations into mineral oil samples.
There
were found higher decayed ferrous concentrations in crude oil samples with
respect to the carbon steels (high) and carbon steels (medium) since other
crude oil samples were free from decayed amounts of ferrous which were related
with stainless steels. Also some significant decayed copper concentrations were
found in crude oil samples which were related to the Monel metal. After
formation of the corrosion compounds on the metal surface such compounds tend
to be removed from the metal surface as soon as possible due to the attractive
and repulsive forces between successive protons and electrons (Figure 7) [1,3,4].
Therefore, such ferrous and copper tend to be decayed into the mineral oils as
corrosion compounds and the observed invisible weight loss during the
determination of the corrosion rates of metal coupons can be explained
easily with these observations and also these observations can be used as the
confirmation evidences for the formation of metallic corrosion forever.
The
deductions of the initial hardness of metal coupons due to the corrosion in
both Murban and Das Blend mineral oils are shown in order of Figure 8 and Figure 9.
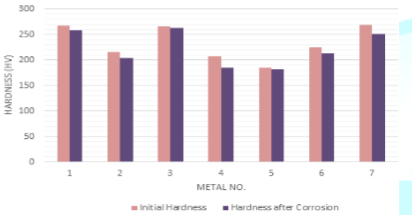
Figure 8: Deductions of
the initial hardness of metal coupons in Murban.
The
slight reductions of the initial hardness of metal coupons were observed by
referring the obtained results for the variations of the initial hardness of
metal coupons due to the corrosion. After the formation of corrosion compounds
on the metal surfaces such compounds tend to remove form such metal surfaces
due to the effects of the repulsive and attractive forces between successive
electrons and protons while creating an instability status on the initial
metallic surface [1,3]. Therefore, it can be emphasized the deductions of the
hardness of metal coupons were happened due to the formation of metallic
corrosion.
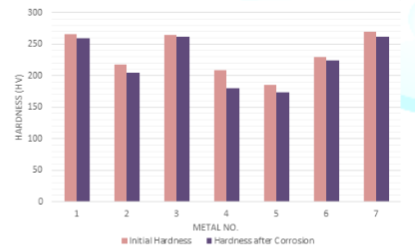
Figure 9: Deductions of
the initial hardness of metal coupons in Das Blend.
Conclusion
Basically
the obtained results interpreted the higher consistency in corrosive properties
of Das Blend than Murban crude oil although the proper progress of every
chemical process was not found at once especially the process of “sulfidation”,
higher corrosive protection strength of stainless steels due to the naturally
formed corrosive protection film, formations of the FeS as the corrosion
compound on the metal coupons with some corrosion cracks and pitting corrosion
in most of metals, decay of ferrous in high amount from carbon steels into both
crude oils since carbon
steels having higher corrosion rates, decay of copper in high amount from Monel
metal into both crude oils and slight reduction in the initial hardness of each
metal coupon due to the formation of the corrosion on the surfaces of metal
coupons. There can be recommended some compositional analysis of the corrosion
compounds that observed during the microscopic analysis by using some advanced
analytical method such as the X-ray diffraction (XRD) for the
future development of current research.
Acknowledgement
This
task was supported and accommodated by the Ceylon Petroleum Cooperation, Sri
Lanka, University of Moratuwa, Sri Lanka and Uva Wellassa University, Sri
Lanka.
References
1.
Khana
OP. Materials Science and Metallurgy (2009)
Dhanpet Rai and Sons, publication, India.
2.
Alsahhaf
TA, Elkilani A and Fahim MA. Fundamentals of Petroleum Refining (2010) Radarweg
Press, Netherland.
3.
Calister
WD. An Introduction of Materials Science and Engineering (2003) John Wiley
& Sons, USA.
4.
Davis
ME and Davis RJ. Fundamentals of Chemical Reaction Engineering (2003)
McGraw-Hill, USA.
5.
Singh
R. Introduction to Basic Manufacturing Process and Engineering Workshop (2006) New
Age International Publication, India.
6.
Bolton
W. Engineering Materials Technology (1994) B.H Newnes Limited, UK.
7.
Ajimotokan
HA, Badmos AY and Emmanuel EO. Corrosion in Petroleum (2009) Pipelines 36-40.
8.
Speight
JG. The Chemistry and Technology of Petroleum (1999) Marcel Dekker, USA.
9.
Afaf
GA. PhD. Thesis, University of Khartoum, 2007.
10.
Okpokwasili
GC and Oparaodu KO. Comparison of Percentage Weight Loss and Corrosion Rate
Trends in Different Metal Coupons from two Soil Environments (2014) Int j
Environ Bioremed Biodegrade 2: 243-249.
*Corresponding author:
Jagath
K Premachandra, Department of Chemical and Process Engineering, University of Moratuwa,
Katubedda, Sri Lanka, E-mail: bajagathp@gmail.com
Citation:
Aluvihara
S and Premachandra JK. Essential stuffs for the cause of
metallic corrosion in mineral oils (2018) Edelweiss Chem Sci J 1: 8-12

















