Introduction
The consumption of global energy is increasing year by year. As the research progress show, in 1998, it was 12.7 TW, but in 2050, it is expected to be around 26.4 to 32.9 TW and in 2100, it will increase up to 46.3 to 58.7 TW [13]. The solar radiation from the sun is approximately 3×1024 J per year, which are ten times the current energy demands of the world [21,65].
As the storage of a fossil supply is ebbing every year the mankind must look for another source of energy [11,18,65]. The sun is a primary source of energy for most life forms in our planet. It is clear, abundant and renewable [14,65]. By fully grasping the power of the sun we can improve our way of life, reduce our dependence on fossil fuels or other types of energy sources and stimulate economy by bringing new jobs to all our planet industry.
Among sustainable and renewable energy resources, such as tidal power, solar thermal, hydropower and biomass, solar cell which is also known as photovoltaic cell is one of the promising options of renewable energy and the most efficient [22,23]. Among different categories of solar cell, the dye-sensitized solar cells (DSSC), which is invented by Professor M. Grätzel in 1991 (ORegan & Grätzel, 1991) [18,26], is a most promising inexpensive route toward sunlight harvesting. DSSCs are belong to the thin film group, emerged as a new class of low cost energy conversion devices with simple manufacturing Procedures [13]. The good light-harvesting efficiency of the best desensitized solar cells (DSSCs) is the product of a dye with moderate extinction and a photo anode of high surface area (∼1200 times the area of a flat electrode).This combination allows for ample absorbance over the majority of the visible spectrum with room for improvement in the red wavelengths [8,15,17].
The fundamental component of the DSC is a photo anode consisting of a monolayer of sensitizer (dye) adsorbed onto a mesoporous semiconductor oxide (typically TiO2). In contrast to conventional solar cell systems, where the semiconductor assumes both the task of light absorption and charge carrier, in dye-sensitized solar cells light is absorbed by the anchored dye and charge separation takes place at the interface via photo induced electron injection from the dye into the conduction band of the solid [7,61].
In general highly efficient photovoltaic conversions, combined with ease of manufacturing and low production costs [6], make the DSC technology an attractive approach for large-scale solar energy conversion comparing to other forms of solar cell.
In this review paper, the general DSSCs benefits and application, DSSCs materials, working principles, efficiency increment due to new materials investigation that suit for DSSC and research challenges will be discussed.
DSSCs Materials
The current DSSC construction involves a set of different layers of components, including glass substrate, transparent conducting layer,TiO2 nanoparticles, dyes, electrolyte (I-/ I-3 or Co II / Co III complexes), and counter electrode (Carbon or Pt) covered with sealing gasket. The typical construction of DSSC is shown in Figure 1.
The main in dye-sensitized solar cells components, including semiconductor films, dye sensitizers nonporous, redox electrolyte, conducting substrate and counter electrode [52].
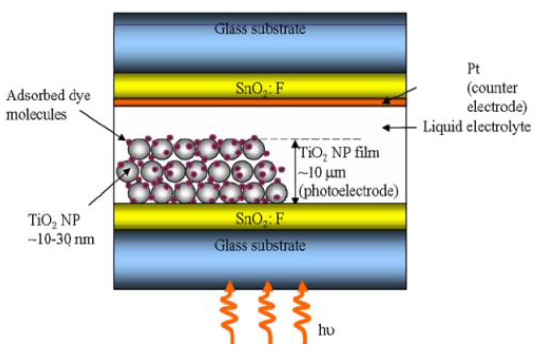
Figure 1: Typical design of a dye-sensitized solar cell
Transparent and Conductive Substrate
DSSCs are typically constructed with two sheets of conductive transparent materials, which help a substrate for the deposition of the semiconductor and catalyst, acting also as current collectors [66]. Substrates necessarily are high transparent (transparency > 80%) to permit the passage of optimum sunlight to the effective area of the cell. Its electrical conductivity should also be high for efficient charge transfer and to decrease energy loss. These two characteristics of substrate dictate the efficiency of DSSCs [21,66].
Typically, FTO (fluorine tin oxide, SnO2: F) and ITO (indium tin oxide, In2O3: Sn) are used as the conductive substrate. ITO and FTO and ITO substrates consist of soda lime glass coated with indium tin oxide layers and fluorine tin oxide, respectively. ITO films have a transmittance of above 80% and sheet resistance of 18 Ω/cm2, while FTO films show a transmittance of about 75% in the visible region and sheet resistance of 8.5/cm2 [21].
Nano Crystalline Semiconductor film Electrode
Semiconductor oxides used in dye-sensitized solar cell include SnO2, Nb2O5, TiO2, ZnO, and so forth, which serve as the carrier for the monolayers of the sensitizer using their high surface and the medium of electron transfer to the conducting substrate. Due to low-cost price, abundance in the market, nontoxicity, and biocompatibility, and as it is also used widely in health care products as well as in paints, TiO2 becomes the best choice in semiconductor till now [52]. Titanium dioxide (TiO2) films are covered on the conducting substrate such as metal foil, flexible polymer film and conducting glass.
Dye Sensitizer
Dye sensitizers serve as the solar energy absorber in DSC, whose proprieties will have much effect on the light harvesting efficiency and the overall photoelectric conversion efficiency. The ideal sensitizer for dye-sensitized solar cells should absorb all light just below a threshold wavelength of 920 nm and firmly grafted to the semiconductor oxide surface and inject electrons to the conduction band with a quantum yield of unity [11,52].Its redox potential should be sufficiently high that it can be regenerated rapidly via electron donation from the electrolyte or a hole conductor. Finally, it should be stable enough to sustain at least 108 redox turnovers under illumination corresponding to about 20 years of exposure to natural light [19].
The purpose of dye is to absorb light and exchange electrons to the conduction band of the semiconductor. It is chemically bonded to the porous surface of the semiconductor. An efficient photosensitizer should [21, 66]:
1. Show excellent absorption in the visible region (400nm to 700nm),
2. Adsorb strongly on the surface of the semiconductor,
3. Has a high extinction coefficient,
4. Be stable in its oxidized form allowing it to be reduced by an electrolyte,
5. Be stable enough to carry out ∼108 turnovers, which typically correspond to 20 years of cell operation,
6. Possess more negative LUMO than the CB of the semiconductor and more positive HOMO than the redox potential of the electrolyte.
In general there are three classes of photosensitizers: metal-free organic sensitizers, natural sensitizers and metal complex sensitizers [66].
Metal Complex Sensitizers
Metal complex sensitizers comprise of both Anchoring Ligands (ACLs) Ancillary Ligands (ALLs). The adhesion of photosensitizers to the semiconductor is highly dependent on the properties of ACLs. While ALLs can be used for the tuning of the overall nature of sensitizers, polypyridine complexes of d6 metal ions possess very high Metal To Ligand Charge Transfer (MLCT) bands in the visible region which is shown by polypyridine complexes of d6 metal ions [21].
Metal - Free Photo Sensitizers
Metal free organic sensitizers have been used both to replace the expensive ruthenium based sensitizers and to improve the electronic properties of devices. Even though, the efficacy of these sensitizers is still low when compared to devices based on ruthenium-based dyes, the efficacy and performance can be improved by the proper selection or tuning of the designing components.
Natural Sensitizers
Natural dyes have also been used in DSSCs due to their low cost, easy extraction, nontoxicity, and the environmentally benign nature [22] (Figure 2).
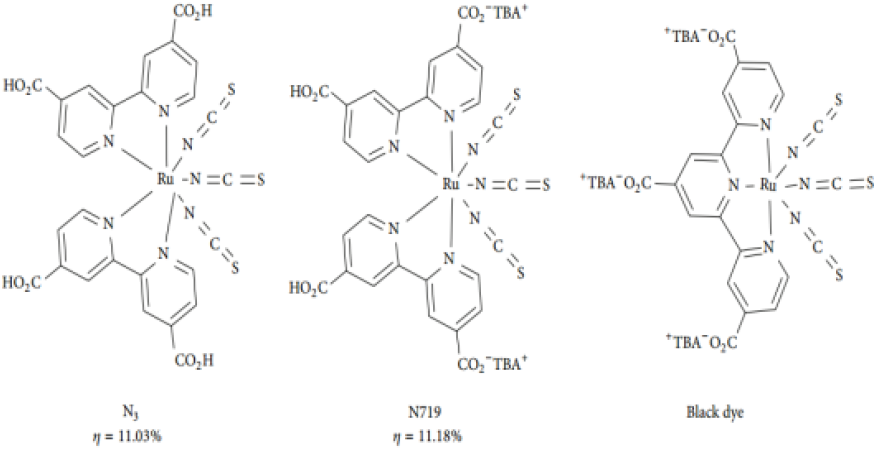
Figure 2: Structure of some efficient Ru-based photosensitizers adapted from
Electrolyte
The purpose of the electrolyte is to regenerate the dye after it injects electrons into the conduction band of the semiconductor. It also acts as a charge passage medium to transfer positive charges toward the counter electrodes. The long-functional life time stability of DSSCs strongly depends on the properties of electrolyte. Thus, the electrolyte must have the following characteristic [21,19].
1. Excellent electrical conductivity and low viscosity for faster diffusion of electrons.
2. Good interfacial contact with the nanocrystalline semiconductor and the counter electrode.
3. It should not be the cause of desorption of the dye from the oxidized surface and the degradation of the dye.
4. It should not absorb light in the visible region.
Electrolytes for DSSCs are classified into three types: solid state electrolytes, liquid electrolytes, and quasi solid state electrolytes.
Liquid Electrolytes
Liquid electrolytes are basically classified into two types: organic solvent based electrolytes and room temperature ionic liquid electrolytes (RTIL) based on the solvent used.
Organic Electrolytes: Each component of organic electrolytes such as the redox couple, solvent, and additives affects the performance of DSSCs. The major component of organic electrolyte is the redox couple. Many types of redox couples such as Br−/Br3, SCN−/ (SCN)2, SeCN−/(SeCN)2 [21,24], and substituted bipyridyl cobalt (III/II) [52] have been investigated. But I3−/I− is considered an ideal redox couple because of its excellent solubility, rapid dye regeneration, low absorbance of light in the visible region, suitable redox potential, and very slow recombination kinetics between injected electrons into the semiconductor and triiodide [13].
Ionic Electrolytes: RTIL have been employed successfully for reduction of a high evaporation rate due to high volatility of liquid electrolytes. They are a group of organic salts containing captions such as pyridinium, imidazolium, and anions from the halide or pseudohalide family [19]. They act simultaneously as an iodine source and as a solvent.
Solid-State Electrolyte
Leakage is the main problem in liquid-electrolyte based DSSCs, which drastically minimize the long-term stability of solar cells. In order to upgrade the performance and stability, solid state electrolytes have been developed. They replace the liquid electrolyte with a p-type semi-conductor [21].
Counter Electrode
The counter electrode is used for the regeneration of the electrolyte. The oxidized electrolyte diffuses towards the counter electrode where it receive electrons from the external circuit. A catalyst is needed to accelerate the reduction reaction and platinum (Pt) is considered a preferred catalyst due to its high exchange current density, good catalytic activity, and transparency. The performance of the CE depends on the method of Pt deposition on TCO substrate [19, 22].
Working Principles of DSSC
The basic operational principles of DSSC solar cells in comparison with conventional semiconductor solar cells are different. In semiconductor solar cells light absorption and charge carrier transport are not the separate task. In DSSC these two tasks are separate. Charge separation is done by photo-induced injection to the conduction band and such created carriers are transported to charge collector [11]. By using dyes the solar cell is capable to harvest large fraction of sunlight due to its high broad absorption band. Figure 3 shows the energy band structure of the DSSC device and principal carrier transfer channels [1]
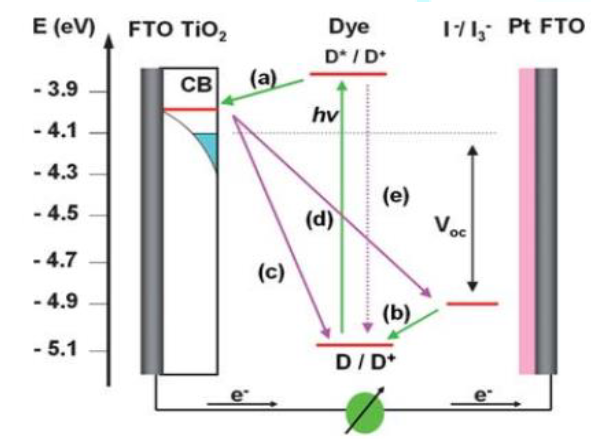
There are many other undesirable
carrier transfer channels including charge recombination of the injected
electrons from the TiO2 CB (defined as the injected electron) to
cations of the dyes (c) and to redox couples (d), and direct decay from the
LUMO to the HOMO of the dye (e).
The general chemical reactions, which take part in all the processes, described
as follow [11,13,18]:
Where D represents dye sensitizer.
In principle, the energy conversion efficiency of a DSSC is the product of the short-circuit photocurrent, Jsc, the open-circuit photovoltage Voc, as well as the fill factor [1, 11]. Referring to the energy band structure and the carrier transfer processes in Fig. 1, the Voc is calculated by the following equation.
Where n is the number of the
electrons in TiO2, NCB is the effective density of states
at conduction band, Eredox is the HOMO level of the redox couples,
and q is the unit charge in coulomb.
In general, working principles of DSSCs are distinct from other classes of
solar cells as the three key processes, i.e., light absorption and the
subsequent generation of electric charges, electron transport, and hole
transport are directed through three materials, thereby making them highly
interfacial devices [54].
Efficiency of DSSCs
Tremendous research efforts have
been invested to improve the efficiency of solar energy conversion which is
generally determined by the light harvesting efficiency, electron injection
efficiency and undesirable charge recombination degree. Pursuing high
efficiency is always the core task for photovoltaic devices. For DSSC, overall
energy conversion efficiency (η) of 11.0% has been achieved at AM 1.5 [7]
.In order to further enhance the energy conversion efficiency of DSSCs, it is
critical to improve the Voc by (1) reduce the charge recombination
between redox couple and the injected electrons in the TiO2 CB; (2)
reduce charge recombination between the oxidized sensitizer and the injected
electrons in the TiO2 CB; (3) increase the electron injection
efficiency; (4) increase the TiO2 ECB; (5) downshift the Eredox; (6)
tandem DSCs.[1]. To reduce charge recombination, the following factors about
sensitizers should be considered. First, it should form a compact blocking
layer on the TiO2 surface. Secondly, the undesirable complexation
between the sensitizer and iodide should be prevented. Thirdly, the electron
donor unit should be separated from the TiO2 surface to impede
charge recombination between the injected electrons and the oxidized
sensitizer. To improve electron injection efficiency, molecular aggregation
should be prevented and the LUMO of the sensitizer should overlap well with
that of TiO2. Finally, to broaden the absorption spectra of the
sensitizer, strong electron donor and acceptor groups might be a good choice.
Furthermore, multiple electron donor substituents are encouraged under the
condition that the oxidized sensitizer can be reduced effectively by the redox
couple. The fast-developing organic sensitizers are promising for reinforcing
the Voc and efficiency by exquisite molecular tailoring.
The DSSC performance also depends on the film morphology Nanoparticles are
essential to increase surface area, and hence, amount of dye, while large
particles are required to enhance absorption of red light through light
scattering. It is impossible to increase surface area and light scattering
simultaneously, because they oppose each other. Therefore, there must be a
balance between them. Such a balance was well controlled by tuning the layer
structure, and an energy conversion efficiency of 10.2% was obtained using a
multilayer structure. The multilayer structure is also suitable for other dyes
in terms of improving light harvesting efficiency, and hence, photocurrent. In
order to scatter the red light more efficiently, a more sophisticated
multilayer structure with gradually increased particle size from the most-inner
layer is desirable [53].
The performance of a DSSC is explained by its I-V characteristics which is
attained from parameters such as short-circuit current Isc and open
circuit voltage Voc and Fill factor (FF) . The ff can be given by
the equation:
Where Voc; open circuit voltage, Isc; short circuit current. Imax and Vmax are the maximum cell current and voltage respectively at the maximum power point, Pmax = Imax x Vmax .
The incident photon conversion efficiency (IPCE) of DSSC is an incident energy-dependent quality. It is a measure of the useful range of the cell. The IPCE is given by
Where λ is wavelength, PINincident optical power, e is the fundamental electron charge, h Plancks constant and c is the speed of light in vacuum. The global power conversion efficiency of energy to electricity conversion efficiency (η) of a cell with Pout electrical power under standard illumination conditions is given by
As the different researcher progress work on DSSC shows, it has currently have low conversion efficiency. Many researchers have attempted to resolve this problem, by increasing the surface area of TiO2 photo-electrodes used in the DSSC [62].
Low efficiency and low stability are the major challenges for the commercial deployment of DSSCs [21].The main causes of low efficiency in DSSCs are
1. Low red and near-IR absorption.
2. Low extinction coefficient requires high surface area.
3. Only redox couple has slow recombination kinetics, but it has unnecessarily large over potential.
4. Poor contact between the electrodes
5. Degradation of electrolyte properties due to UV absorption of light.
Improving the environmental stability of cells is the most important issue in studying these cells [60]. Stability refers to the performance of individual processes or the entire solar cell at any time relative to the initial time. Good stability leads to long lifetimes [59]. The critical issue regarding to stability and robustness of DSSCs are
1. Liquid electrolyte is undesirable, but solid state hole conductors give lower efficiency.
2. Achieving DSSC module lifetimes of more than 20 years requires 108 turnovers for dye molecules and high quality encapsulation to prevent leakage of the electrolyte and ingress of water [59].
3. Is corrosive.
Advantages of
DSSCs
Dye-sensitized solar cells have
the following main advantages:
Capable of production in a simple way: Dye-sensitized
solar cells require no vacuum system for manufacturing, and thus have an
essential advantage in terms of production cost [63]. It reduces manufacturing
cost by 1/5 to 1/10 as compared to silicon solar cells production cost [22, 23
and 55].
Colorable, transparent: The use of dye allows wide selection of colored cells and transparent cells. The transparency and varied color of DSSCs could be utilized for decorative purposes like window and sunroof [58].
Flexible and thin structure: By using aggregates of fine particles of photoelectric conversion materials, the solar cells can be formed as flexible thin films.
Generation
characteristics of insusceptible to the incident angle and intensity of the
sunlight: Even
though the light condition is very week generation characteristics can be maintained,
such as under faint light in the morning and evening and when indoors.
Lighter weight:
Plastic
substrates can be used to minimize the weight of solar cells and panels. Dou to
its light weight, dye-sensitized solar cells can be installed in locations
where appearance is important and other solar cells are not applicable, such as
the glass panes and outer and inner walls of a building, the sunroof and outer
panels of an automobile, and
the enclosure of a hand phone. This allows the creation of new markets with
high demand [55,56.58] (Figure 4).
Environmentally friend and recyclable
Dye-sensitized solar cells do not have harmful substance as a cell component material. The materials are comparatively easy to separate and get back, which is advantageous in view of a recycling and reuse framework for solar cell panels [63].
Conclusion
In conclusion, the worlds nonrenewable energy degrades time by time and the consumption rate increases inversely. To weaken these two controversies, new environmental friend green renewable energy resources are highly needed to our planet. Among different types of renewable green energy resources, solar energy is regarded as one of the perfect energy resources.
There has been a continuous effort in searching for affordable organic solar energies among which dye-sensitized solar cells (DSCs) thus far demonstrate the highest energy conversion efficiency, and have been regarded as the most prospective technology in the near future. Dye-sensitized solar cells have gained widespread attention in recent years because of their low production costs, easy of fabrication, its lighter weight property, environmentally friend and recyclable advantages and tunable optical properties, such as color and transparency regardless of its low efficiency output comparing to silicon solar cell.

Figure 4: Prototype Models of Dye-Sensitized Solar Cell Panels for decoration purpose
1. Zhijun Ning, Ying Fu and He Tian. Improvement of dye-sensitized solar cells: What we know and what we need to know (2010) Energy Environmental Science. DOI: 10.1039/C003841E
2. Jason B Baxter and Eray S Aydil. Nanowire-based dye-sensitized solar cells (2005) Applied Physics Letters 86: 053114. https://doi.org/10.1063/1.1861510
3. Renu Guliani, Amit Jain and Avinashi Kapoor. Exact Analytical Analysis of Dye-Sensitized Solar Cell: Improved Method and Comparative Study (2012) The Open Renewable Energy J 5: 49-60. http://dx.doi.org/10.2174/1876387101205010049
4. QB Meng, K Takahashi, XT Zhang, I Sutanto, TN Rao, et al. Fabrication of an Efficient Solid-State Dye-Sensitized Solar Cell (2003) Langmuir, 19: 3572-3574. DOI: 10.1021/la026832n
5. Jeong-Hyeok IM, Chang-Ryul Lee, Jin-Wook Lee, SangWon Park and Nam-Gyu Park. 6.5% efficient perovskite quantum-dot-sensitized solar cell (2011) Nanoscale 3: 4088. DOI: 10.1039/C1NR10867K
6. Seigo Ito, Takurou N. Murakami, Pascal Comte, Paul Liska, Carole Grätzel, et al. Fabrication of thin film dye sensitized solar cells with solar to electric power conversion efficiency over 10% (2008) Elsevier Thin Solid Films 516: 4613-4619. https://doi.org/10.1016/j.tsf.2007.05.090
7. Qing Wang, Seigo Ito, Michael Gra1tzel, Francisco Fabregat-Santiago, Iva´n Mora-Sero´, et al. Characteristics of High Efficiency Dye-Sensitized Solar Cells (2006) J Phys Chem B 110: 25210-25221. https://doi.org/10.1021/jp064256o
8. Alex BF Martinson, Jeffrey W Elam, Joseph T Hupp and Michael J Pellin. ZnO Nanotube Based Dye-Sensitized Solar Cells (2007) Nano letters 7: 2183-2187. DOI: 10.1021/nl070160+
9. Jason B Baxter. Dye Sensitized Solar Cells: R&D Issues (2010) NSF PV Workshop.
10. Simon Mathew, Aswani Yella, Peng Gao, Robin Humphry-Baker, Basile FE Curchod, et al. Dye-sensitized solar cells with 13% efficiency achieved through the molecular engineering of porphyrin sensitizers (1861) Nature chemistry. https://doi.org/10.1038/nchem.1861
11. Michal Sokolský and Július Cirák. Dye-sensitized solar cells: Materials and processes (2010) Acta Electro technica et Informatica 10: 78-81.
12. Rajaram S. Mane, Won Joo Lee, Habib M. Pathan and Sung-Hwan Han. Nanocrystalline TiO2/ZnO Thin Films: Fabrication and Application to Dye-Sensitized Solar Cells (2008) Physical chemistry 109: 24254–24259. DOI: 10.1021/jp0531560
13. Arini Nuran Binti Zulkifili, Terauchi Kento, Matsutake Daiki and Akira Fujiki. The Basic Research on the Dye-Sensitized Solar Cells (DSSC) (2015) J Clean Energy Technologies 3. DOI: 10.7763/JOCET.2015.V3.228
14. Andigoni Apostolopoulou, Dimitris Karageorgopoulos, Andreas Rapsomanikis and Elias Stathatos. Dye-Sensitized Solar Cells with Zinc Oxide Nanostructured Films Made with Amine Oligomers as Organic Templates and Gel Electrolytes (2016) J Clean Energy Technologies. DOI: 10.18178/JOCET.2016.4.5.303
15. Takeru Bessho, Shaik M Zakeeruddin, Chen-Yu Yeh, Eric Wei-Guang Diau and Michael Grtzel. Highly Efficient Mesoscopic Dye-Sensitized Solar Cells Based on Donor–Acceptor-Substituted Porphyrins (2010) Communications. https://doi.org/10.1002/anie.201002118
16. Yong-Bing Tang, Chun-Sing Lee, Jun Xu, Zeng-Tao Liu, Zhen-Hua Chen, et al. Incorporation of Graphenes in Nanostructured TiO2 Films via Molecular Grafting for Dye-Sensitized Solar Cell Application. DOI: 10.1021/nn100449w
17. Zhen Huang, Xizhe Liu, Kexin Li, Dongmei Li, Yanhong Luo, et al. Application of carbon materials as counter electrodes of dye-sensitized solar cells (2007) Elsevier, Electrochemistry Communications 9: 596-598. https://doi.org/10.1016/j.elecom.2006.10.028
18. Yang Jiao, Fan Zhang and Sheng Meng. Dye Sensitized Solar Cells Principles and New Design. Beijing China.
19. Khalil Ebrahim Jasim. Dye Sensitized Solar Cells - Working Principles, Challenges and Opportunities. Department of Physics, University of Bahrain Kingdom of Bahrain.
20. Geargg. A low cost, high efficiency solar cell based on dye synthesized colloidal Tio2 films (2015) Nature. https://doi.org/10.1038/353737a0
21. Umer Mehmood, Saleem-ur Rahman, Khalil Harrabi, Ibnelwaleed A. Hussein, BVS. Reddy. Recent Advances in Dye Sensitized Solar Cells (2014).
22. Suriati Suhaimi, Mukhzeer Mohamad Shahimin, ZA Alahmed, J Chyský and AH Reshak. Materials for Enhanced Dye-sensitized Solar Cell Performance: Electrochemical Application (2015) Int J Electrochem Sci 10: 2859-2871.
23. Sanghoon Yoon, Sehyun Tak, Jinsoo Kim, Yongseok Jun, Kisuk Kang, et al. Application of transparent dye-sensitized solar cells to building integrated photovoltaic systems (2011) Elseveir Building and Environment 46: 1899e1904. https://doi.org/10.1016/j.buildenv.2011.03.010
24. In Chung, Byunghong Lee, Jiaqing He, Robert PH Chang and Mercouri G Kanatzidis. All-solid-state dye-sensitized solar cells with high efficiency (2012) Nature 485: 486-489 https://doi.org/10.1038/nature11067
25. Kroon JM. ENK6-CT2001-00575 NANOMAX 8-02-2005.
26. S Karuppuchamy, K Nonomura, T Yoshida, T Sugiura and H Minoura. Cathodic electrodeposition of oxide semiconductor thin films and their application to dye-sensitized solar cells (2001) Solid State Ionics 151: 19-27 https://doi.org/10.1016/S0167-2738(02)00599-4
27. Anders Hagfeldt, Gerrit Boschloo, Licheng Sun, Lars Kloo and Henrik Pettersson. Dye-Sensitized Solar Cells (2010) Chem Rev 110: 6595-6663. DOI: 10.1021/cr900356p
28. Michael Grätzel. Review Dye-sensitized solar cells (2003) Elsevier J Photochemistry and Photobiology C: Photochemistry Reviews 4: 145-153. doi:10.1016/S1389-5567(03)00026-1
29. Md K Nazeeruddin, Etienne Baranoff and Michael Gratzel. Dye-sensitized solar cells: A brief overview (2011) Elsevier Solar Energy 85: 1172-1178. https://doi.org/10.1016/j.solener.2011.01.018
30. Monzir S Abdel-Latif, Mahmoud B Abuiriban, Taher M El-Agez, et al. Dye-Sensitized Solar Cells Using Dyes Extracted From Flowers, Leaves, Parks, and Roots of Three Trees (2015) Int J Renewable Energy Res.
31. Kohjiro Hara and Hironori Arakawa. Dye-sensitized Solar Cells. National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba, Japan.
32. Yasuo Chiba, Ashraful Islam, Yuki Watanabe, Ryoichi Komiya and Naoki Koide. Dye-Sensitized Solar Cells with Conversion Efficiency of 11.1% (2006) Japanese J Appl Physics 45: L638–L640. https://doi.org/10.1143/JJAP.45.L638
33. David Riehm. Improving Dye-Sensitized Solar Cell Efficiency by Modification of Electrode Surface Charge.
34. Michael Gra1tzel. Solar Energy Conversion by Dye-Sensitized Photovoltaic Cells (2005) Inorg Chem 44: 6841-6851. DOI: 10.1021/ic0508371
35. Michael Grätzel. Review Dye-sensitized solar cells (2003) Elsevier J Photochemistry and Photobiology C: Photochemistry Reviews 4: 145-153. doi:10.1016/S1389-5567(03)00026-1
36. Michael Grätzel. Conversion of sunlight to electric power by Nano crystalline dye-sensitized solar cells (2004) Elsevier Journal of Photochemistry and Photobiology A: Chemistry 164: 3-14. https://doi.org/10.1016/j.jphotochem.2004.02.023
37. Brian E Hardin, Henry J Snaith and Michael D McGehee. The renaissance of dye-sensitized solar cells (2012) Nature photonics 6: 162-169. https://doi.org/10.1038/nphoton.2012.22
38. H Chang, TL Chen, KD Huang, SH Chiena and KC Hung. Fabrication of highly efficient flexible dye-sensitized solar cells (2010) Elsevier 504S: S435-S438. http://dx.doi.org/10.1016/j.jallcom.2010.02.044
39. Tamotsu Horiuchi, Hidetoshi Miura, Kouichi Sumioka and Satoshi Uchida. High Efficiency of Dye-Sensitized Solar Cells Based on Metal-Free Indoline Dyes (2004) J Ame Chemical Society 126:12218-12219. https://doi.org/10.1021/ja0488277
40. Tzi-Yi Wu, Ming-Hsiu Tsao, Fu-Lin Chen, Shyh-Gang Su and Cheng-Wen Chang. Synthesis and Characterization of Organic Dyes Containing Various Donors and Acceptors (2010) Int J Mol Sci 11: 329-53. https://doi.org/10.3390/ijms11010329
41. Seigo Ito, Shaik M. Zakeeruddin, Robin Humphry-Baker, Paul Liska, Raphaël Charvet, et al. High-Efficiency Organic-Dye-Sensitized Solar Cells Controlled by Nanocrystalline-TiO2 Electrode Thickness (2005) Communications 18: 1202-1205 https://doi.org/10.1002/adma.200502540
42. Kohjiro Hara, Tadatake Sato, Ryuzi Katoh, Akihiro Furube, Yasuyo Ohga, et al. Molecular Design of Coumarin Dyes for Efficient Dye-Sensitized Solar Cells (2003) J Phys Chem B 107: 597-606. DOI: 10.1021/jp026963x
43. Qing Wang, Jacques E. Moser and Michael Graltzel. Electrochemical Impedance Spectroscopic Analysis of Dye-Sensitized Solar Cells (2005) J Phys Chem B 109: 14945-14953. http://dx.doi.org/10.1021/jp052768h
44. Matt Law, Lori E. Greene, Justin C. Johnson, Richard Saykally and Peidong Yang. Nanowire dye-sensitized solar cells (2005) Nature Materials 4: 455-459 https://doi.org/10.1038/nmat1387
45. Amaresh Mishra, Markus KR Fischer and Peter B_Uerle. Metal-Free Organic Dyes for Dye-Sensitized Solar Cells: From Structure, Property Relationships to Design Rules (2008) Angew Chem Int Ed Engl 48: 2474-2499 https://doi.org/10.1002/anie.200804709
46. Qifeng Zhang, Christopher S. Dandeneau, Xiaoyuan Zhou, and Guozhong Cao. ZnO Nanostructures for Dye-Sensitized Solar Cells (2008) Advanced materials 21: 4087-4108 https://doi.org/10.1002/adma.200803827
47. Diah Susantia, Maula Nafi, Hariyati Purwaningsiha, Rindang Fajarin and George Endri Kusuma. The Preparation of Dye Sensitized Solar Cell (DSSC) from TiO2 and Tamarillo Extract (2014) Elsevier Procedia Chemistry 9: 3-10. https://doi.org/10.1016/j.proche.2014.05.002
48. Jun-HoYum, Peter Chen, Michael Grtzel and Mohammad K Nazeeruddin. Recent Developments in Solid-State Dye-Sensitized Solar Cells (2008) ChemSusChem 1: 699-707 https://doi.org/10.1002/cssc.200800084
49. Tomohiro Nagata and Hirohiko Murakami. Development of Dye-sensitized Solar Cells (2009).
50. Kazuharu Suzuki, Makoto Yamaguchi, Mikio Kumagai and Shozo Yanagiday. Application of Carbon Nanotubes to Counter Electrodes of Dye-sensitized Solar Cells (2003) Chemistry Letters 32: 1. http://dx.doi.org/10.1246/cl.2003.28
51. Lukas Schmidt-Mende and Michael Gratzel. TiO2 pore-filling and its effect on the efficiency of solid-state dye-sensitized solar cells (2006) CH-1015 Lausanne, Switzerland.
52. Fan-Tai Kong, Song-Yuan Dai and Kong-Jia Wang. Review of Recent Progress in Dye-Sensitized Solar Cells (2007) Advances in optoelectronics http://dx.doi.org/10.1155/2007/75384
53. Zhong-Sheng Wang, Hiroshi Kawauchi, Takeo Kashima and Hironori Arakawa. Review significant influence of TiO2 photo electrode morphology on the energy conversion efficiency of N719 dye-sensitized solar cell (2004) Coordination Chemistry Reviews 248 1381-1389. https://doi.org/10.1016/j.ccr.2004.03.006
54. Qamar Wali, Azhar Fakharuddin and Rajan Jose. Tin oxide as a photo anode for dye-sensitized solar cells: Current progress and future challenges (2015) J Power Sources 293: 1039e1052. https://doi.org/10.1016/j.jpowsour.2015.06.037
55. M. Grätzel. Nature (2001).
56. http://www.sony.co.jp/Products/SCHP/cx_pal/vol80/pdf/sideview80.pdf
57. Norasikin A. Ludin, A.M. Al-Alwani Mahmoud, Abu Bakar Mohamad, Abd. Amir H. Kadhum, Kamaruzzaman Sopian, et al. Review on the development of natural dye photosensitizer for dye-sensitized solar cells (2014) Renewable and Sustainable Energy Reviews 31: 386-396. https://doi.org/10.1016/j.rser.2013.12.001
58. Jiawei Gong, Jing Liang and K Sumathy. Review on dye-sensitized solar cells (DSSCs): Fundamental concepts and novel materials (2012) Renewable and Sustainable Energy Reviews 16: 5848-5860. https://doi.org/10.1016/j.rser.2012.04.044
59. Jason B Baxter. Commercialization of dye sensitized solar cells: Present status and future research needs to improve efficiency, stability, and manufacturing (2012) J Vacuum Sci Tech A30: 020801. https://doi.org/10.1116/1.3676433
60. Nilofar Asim, Kamaruzzaman Sopian, Shideh Ahmadi, Kasra Saeedfar, M.A. Alghoul, et al. A review on the role of materials science in solar cells (2012) Elsevier 16: 5834–5847. https://doi.org/10.1016/j.rser.2012.06.004
61. Adedokun Oluwaseun, Titilope Kamil and Awodugba Ayodeji Oladiran. Review on Natural Dye-Sensitized Solar Cells. Int J eng tech.
62. Jeong-Hwa Kim, Dae-Hwan Kim, Kang-Pil Kim, Dong-Hwan Jeon and Dae-Kue Hwang. Enhancement of the light harvesting efficiency in a dye-sensitized solar cell by a patterned reflector (2013) Thin Solid Films 546: 326-330. https://doi.org/10.1016/j.tsf.2013.03.062
63. Hironori Arakawa. Recent Advances in Research and Development for Dye-Sensitized Solar Cells II, CMC Publishing, 2007.
64. Monishka Rita Narayan. Review: Dye sensitized solar cells based on natural photosensitizers (2012) Renewable and Sustainable Energy Reviews 16: 208-215. https://doi.org/10.1016/j.rser.2011.07.148
65. Michal Sokolský and Július Cirák. Dye-Sensitized solar cells: Materials and Processed (2010) Acta Electrotechnica et Informatica10: 78-81.
66. Umer Mehmood, Saleem-ur Rahman, Khalil Harrabi, Ibnelwaleed A Hussein and Reddy BVS. Recent Advances in Dye Sensitized Solar Cells (2014) Hindawi Publishing Corporation Advances in Materials Science and Engineering 18: 155-162. https://doi.org/10.1016/j.mattod.2014.09.001
*Corresponding author:
Andualem A, School of Materials Science and Engineering, Jimma University, Jimma, Ethiopia, E-mail: antenehanduale@ymail.com
Citation:
Andualem A, Demiss S. Review on Dye-Sensitized Solar Cells (DSSCs) (2018) J Heterocyclics 1: 29-34


 PDF
PDF