Introduction
Water mites are one of the polyphilic groups in the Acari subclass. They are known as Hydracarina, Hydrachnidia or Hydrachnellae. Over 6.000 species have been defined worldwide, representing 57 families, 81 subfamilies and more than 400 genera. Water mites have a complex life cycle. Their eggs are found on many different water plants in the water. They live in different animal species as ectoparasites in larval stages [1-3]. There is a special significance in the determination of living areas and communities in streams, lakes and ponds. The water mites which spread almost all over inland waters are used as biological indicator organisms in the determination of clean water resources [3-8].
Up to this time, it seems that the studies on the water mites are classical systematic studies. Recently, ecological, genetic, and other molecular studies have also been carried out in this group [9-16]. The determination of fatty acid compositions has been done in water mites (Hydrachnadia) group, for the first time in the present study. In this study, the fatty acid ratios of water mites species were determined and similarities between species have been discussed in terms of these ratios. Oils are one of the important organic compounds required for all living things, including humans. In addition to being a high energy source, they are very important in terms of combining with proteins to form lipoproteins and to contain fat soluble vitamins [17,18].
Fatty acids are monobasic organic acids with a straight chain and varying chain length, usually containing a double number of carbon atoms. All fatty acids have long hydrocarbon chains, a methyl group at one end of the chain and a carboxyl group at the other end. The presence of long or short chain fatty acids depends on the number of carbon atoms it contains and ranges from 4 to 26. Fatty acids predominantly present in fats usually contain 16-18 carbon atoms. The fatty acids to which all of the carbon atoms are bound by a single bond are called saturated fatty acids (e.g., palmitic acid). Fatty acids containing at least one double bond between carbon atoms are called unsaturated fatty acids (e.g., oleic acid). If there is more than one double bond between carbon atoms, it is called polyunsaturated fatty acid (e.g., linoleic acid) [19].
According to their physical properties, unsaturated fatty acids up to 10°C are present in liquid form at room temperature, while longer chain fatty acids are solid [20]. Studies on fatty acids have been done more in vertebrates, but much less in invertebrate species. In the Acari team, which is an invertebrate subgroup, these studies are seen only in terrestrial forms [21-24].
Some notable of these are as follows:
The fatty acids contained in some Acari species have been identified in the study entitled: “Variability in cuticular hydrocarbons and phenotypic discrimination of Ixodes ricinus populations (Acarina: Ixodidae) from Europe” by Estrada-Pena and coworkers and in the study “Fatty acids as cuticular surface components in oribatid mites (Acari: Oribatida)” by Raspotnig and Krisper [25] and in the study “Cuticular fatyy acid profile analysis of three Rhipicephalus tick species (Acari: Ixodidae)” by Shimshoni et al. [26].
No studies have been done about water mites on this subject. This study has been the first on this subject. In this study, it has been seen that the similarities in fatty acid compositions increase with the systematic proximity of water mites.
Materials and Method
Collection of samples
The present study was carried out on water mites Hydrachna processifera,
Eylais setosa and Hydrodroma despiciens species (Acari; Hydracnidia) collected
from Karamik Lake within the boundaries of Afyonkarahisar province. Species
determinations were made under the microscope in the laboratory environment.
Determination of fatty acids
Chloroform: Methanol mixture (2:1) was used to obtain oil samples [27]. To obtain crude oil, chloroform:methanol (2:1). 30 ml mixture was added to 0.1 g of the fractionated water mites samples. The samples were then crushed until they became slurry with 24000 rpm ultra-homogenizer. The mixture was filtered with filter paper (1st filtration). The residue on the filter paper was removed and chloroform: methanol mixture (20 ml) was added and homogenized for a second time. The resulting slurry mixture was again filtered. The first and second filtrates were combined. It was then taken up in a 250 ml separating funnel and 20 ml of reagent solution was added and shaken well and the phases were allowed to stand until separated. The organic phase (chloroform) was taken up in the evaporation flask and the solvent was completely evaporated in the Heidolph-2 brand vacuum rotary evaporator at 45ºC. Chloroform remaining in the oil was removed with dry nitrogen and crude oil was obtained.Esterification process: A 16-20 mg homogenized oil sample was taken in a capped cap and 4 ml of 2% methanolic NaOH solution was added. The tube was sealed with nitrogen gas and then boiled for 10 minutes until saponification occurred on the water bath. At the end of the saponification, 2 ml of 14% BF3-methanol complex was added to the cooled mixture and boiled for more 5 min.
Then the tube was cooled to 30-40ºC and shaken vigorously for 30 seconds by adding of isooctane. 4 mL saturated NaCl solution was added over it. After the mixture was thoroughly shaken, it was taken into the separation funnel and allowed to stand for 5 to 10 minutes to separate the phases. At the end of the extractions, the upper phase (organic phase) was taken and dried with Na2SO4.It was then passed through special filters of 0.45 mm in diameter and placed in vials and filled with nitrogen gas to close the caps tightly [28]. This extract was injected into gas chromatography [29].Injecting the samples into the gas chromatograph: Fatty acid methyl esters were analyzed by gas chromatography (HP Agilent 7890A) using an HP capillary column [(100-m length and 0.25-mm internal diameter and 0.20 μm of film thickness; HP 88)].
Gas chromatographic conditions were follows:
• Injector temperature: 250°C, detector temperature: 250°C, carrier gas: H2, 30 mL / min.
• Split ratio 50:1, split, flow rate 71.0 ml/min. temperature program, initial 80°C for 50 minutes, temperature 10°C/minute for 40 min at 210°C.
The methylated extract was taken with an automatic injector of gas chromatography and peaks were detected in the chromatograph. The peaks obtained from the samples were identified by comparing the fatty acids with the standard peaks and fatty acids were calculated as percentages.Fatty acid methyl ester standards: To determine the fatty acids, mix standards containing 37 fatty acids and mix standards containing 4 and 5 fatty acids were used. The fatty acids in the samples were determined by comparing the peaks of the samples with the peaks of the fatty acid standards. Peaks of fatty acids in the standard are shown in Figure 1.
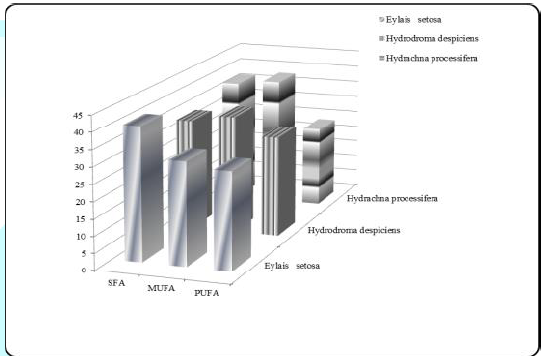
Figure 1: Average SFA, MUFA and PUFA ratios (%) for species living in Karamık Lake.
Statistical evaluation: Statistical analyses were performed with the SPSS 18.0 computer program. First, the normality test of the data was performed. Single and two-way analysis of variance was used because the data showed normal distribution and there were more than two groups. The Turkey test was used for multiple comparison tests of homogeneous groups and the Tamhane test was used for the non-homogenous groups by controlling the homogeneity of the variances. Also, the arithmetic average ± standard deviation values were given for each group.
Results and Discussion
This study was carried out with common species of water mites (Acari, Hydrachnidia) Eylais setosa, Hydrodroma despiciens, and Hydrachna processifera in lakes. These three species are included in a separate water mite family group. In the present study, fatty acid compositions were determined for the first time in water mites species.No studies have been found on the identification of fatty acid compositions in the water mites until now. However, few studies have been made on this issue with some terrestrial and parasitic species of Acari. Among these, some notable studies are as follows:In their study, Aboshi et al. have reported that Tyrophagus similis and Tyrophagus putrescentiae (Astigmata: Acaridae) species have the ability to biosynthesize linoleic acid [(9Z, 12Z) -9, 12-octadecadienoic acid]. Murungi and colleagues reported that fatty acids in essential oils (camphor, limonene, decanoic acid, hexadecanoic acid, dodecanoic acid) from the leaves and fruits of Solanum sarrachoides plant had negative effects on the laying of Tetranychus evansi (tomato spider mite) [30].
In another study, Maazouzi et al. investigated the effects of diarrhea and feeding on the hepatopancreatic fatty acid composition of the Eriocheir sinensis species at different periods and they reported that total saturated and monounsaturated fatty acids differed by nutritional status [31].In a similar study, Wen et al., showed that fatty acid analysis of phospholipids, neutral lipids and total lipids of the Unio elongatulus species showed the highest C16:0 (18.23% -24.86% 9 (10.23% -45.10%) and C18:2n-6 (3.50% -16.94%) fatty acids [32].
In the study of Wen et al., the ratio of total polyunsaturated n-3 and n-6 PUFA (43.86%) to phospholipid; the proportion of total monounsaturated fatty acids (61.39%) was found to be high in neutral lipid. Another important study on this subject was carried out by Hayashi and Takagi [33]. In this study, researchers found that saturated fatty acids such as myristic, palmitic and stearic acid taken via food and unsaturated fatty acids such as oleic, linoleic and linolenic acid were stored directly in fish oils and that the seasonal variation of these fatty acids was due to the phytoplankton, zooplankton .In the present study titled "Fatty acid and lipid composition of Water Mites (Acari, Hydracnidia) species by GC-MS", fatty acid ratios in the waters (Eylais setosa, Hydrodroma despiciens and Hydrachna processifera) are evaluated as saturated, monounsaturated and polyunsaturated in Tables 1-3.
Saturated and unsaturated fatty acid results
In this study, oil acid analysis was carried out by gas chromatography (GC-MS) in water mites (Acari, Hydracnidia) species Hydrachna processifera, Eylais setosa and Hydrodroma despiciens collected from Karamik Lake. The results obtained were evaluated as saturated monounsaturated and polyunsaturated fatty acids. The data were statistically evaluated and included with standard deviations in Table 1.
Results of saturated fatty acids: From Table 1, it is seen that palmitic (C16:0) and stearic acid (C18:0) of saturated fatty acids are found in high proportion in three of the water mites samples studied. According to these numerical values, there is a remarkable difference between species in terms of palmitic and stearic acid.
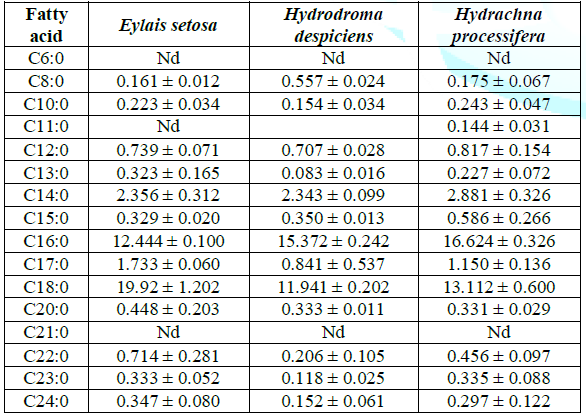
Table 1: Average saturated fatty acid compositions (%) of water mites species in Lake Karamık.
The ratio of total saturated fatty acid was found 40.072% for Eylais setosa, while Hydrodroma despiciens 33.299% and Hydrachna processifera 37.235% were found in other species. There was a significant difference in SFA ratios between species. The change in total SFA rates is shown in Table 1 and Figure 1.
The ratio of stearic acid was found to be 11.941% in the lowest Hydrodroma despiciens and 19.924% in the highest Eylais setosa. There was no significant difference found between Hydrodroma despiciens and Hydrachna processifera when compared to the other two species in Eylais setosa in terms of stearic acid ratio.The lowest percentage of palmitic acid was found in Eylais setosa (12.444%) and the highest in Hydrachna processifera (16.624%). There was a significant difference in palmitic acid ratio compared to the other two species of Eylais setosa, but no difference was observed between Hydrodroma despiciens and Hydrachna processifera.Monounsaturated fatty acid results: The changes in the monounsaturated fatty acid compounds in the samples are shown in Table 2 and Figure 1.
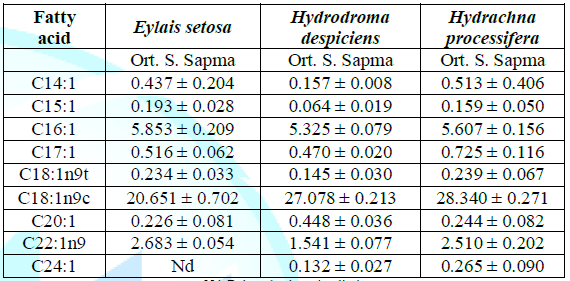
Table 2: Average monounsaturated fatty acid compositions (%) of water mites species in Lake Karamık.
Among the monounsaturated fatty acids, the most abundant fatty acids are palmitoleic acid (C16:1), oleic acid (C18:1) and erucic acid (C22:1). The total monounsaturated fatty acid content was found to be 38.638% in Hydrachna processifera, 30.792% in Eylais setosa and 35.400% in Hydrodroma despiciens. There was a significant difference in the MUFA ratios between the species.Palmitoleic acid ratios was 5.325% in Hydrodroma despiciens and 5.853% in Eylais setosa. In terms of palmitoleic acid ratios, there were no significant differences detected between the species.The lowest oleic acid content was found in Eylais setosa (20.651%) and the highest in Hydrachna processifera (28.340%). In terms of oleic acid ratio, there was a significant difference in comparison with the other two species of Eylais setosa, but no difference was observed between Hydrodroma despiciens and Hydrachna processifera.
Erucic acid ratio was found to be 1.541% in Hydrodroma despiciens and 2.683% in Eylais setosa. In terms of erucic acid ratios, Hydrodroma despiciens showed a significant difference when compared to the other two species, but no difference was observed between Eylais setosa and Hydrachna processifera.Polyunsaturated fatty acid results: The main polyunsaturated ω3 fatty acids are linolenic acid (C18:3ω3), Eicosatrienoic acid (C20:3ω3), Eicosapentaenoic acid (C20:5ω3) and Dokosahexaenoic acid (C22:6ω3). ω6 fatty acids are Linoleic acid (C18:2ω6), γ-Linolenic acid (C18:3ω6), Eicosadienoic acid (C20:2ω6) and Arachidonic acid (C20:4ω6).The total polyunsaturated fatty acid ratio was found to be 30.076% in Hydrodroma despiciens, 29.122% in Eylais setosa and 24.238% in Hydrachna processifera. There was a significant difference in PUFA ratios between species. The variation of the total PUFA ratios is shown in Table 3.
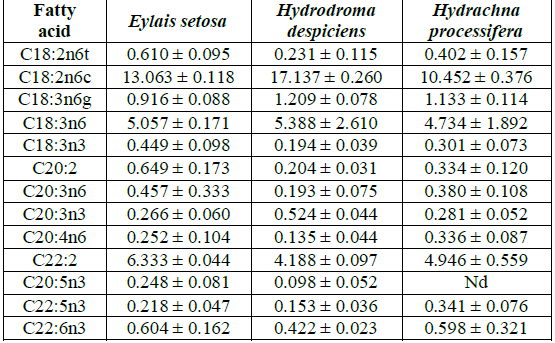
Table 3: Average polyunsaturated fatty acid compositions (%) of water mites species in Lake Karamik.
In ω3 fatty acids, eicosatrienoic acid was found to be the highest in Hydrodroma despiciens at 0.524% and the lowest in Eylais setosa at 0.266% and 0.281% in Hydrachna processifera. Hydrodroma despiciens in terms of eicosatrienoic acid ratio was significantly different when compared to the other two species, but no difference was observed between Eylais setosa and Hydrachna processifera.Eicosapentaenoic acid in ω3 fatty acids was found to be the highest in Eylais setosa at 0.248%, 0.098% in Hydrodroma despiciens and below the detection limit in Hydrachna processifera. When compared to Eylais setosa and Hydrodroma despiciens in terms of eicosapentaenoic acid ratios, significant differences were observed.The highest concentration of docosahexaenoic acid was found in Eylais setosa (0.604%) and the lowest level was found in Hydrodroma despiciens (0.422%) and in Hydrachna processifera (0.598%). Hydrodroma despiciens in terms of docosahexaenoic acid ratio was significantly different when compared to the other two species, but no difference was observed between Eylais setosa and Hydrachna processifera.
Linolenic acid content was highest in Eylais setosa (0.449%) and lowest in Hydrodroma despiciens (0.194%) and in Hydrachna processifera (0.301%). Significant differences were observed between species in terms of linolenic acid ratios.The highest ratio of arachidonic acid in ω 6 fatty acids was 5.388% in Hydrodroma despiciens while the lowest was 4.734% in Hydrachna processifera and 5.057% in Eylais setosa. There were no significant differences found between the species in terms of arachidonic acid ratios.Among the ω6 fatty acids, C18:2ω6 was found to be the highest in Hydrodroma despiciens at 17.137% and the lowest in Hydrachna processifera at 10.452% and 13.063% in Eylais setosa. There was a significant difference between species in terms of C18:2ω6 ratios.
Among ω6 fatty acids, the highest ratio of C22:2ω6 was 6.333% in Eylais setosa and the lowest was 4.188% in Hydrodroma despiciens and 4.946% in Hydrachna processifera. There was no significant difference between Hydrodroma despiciens and Hydrachna processifera when compared to the other two species of Eylais setosa in terms of C22:2ω6 ratio.Fatty acid compositions of different species are given comparatively in Table 1. In terms of the fatty acid (SFA, MUFA and PUFA) ratios found, the values of water mites species (Hydrodroma despiciens,Hydrachna processifera, Rhipicephalus sanguineus) were found to be close to each other. This was due to systematic closeness. Table 4 gives the fatty acid ratios in different species.
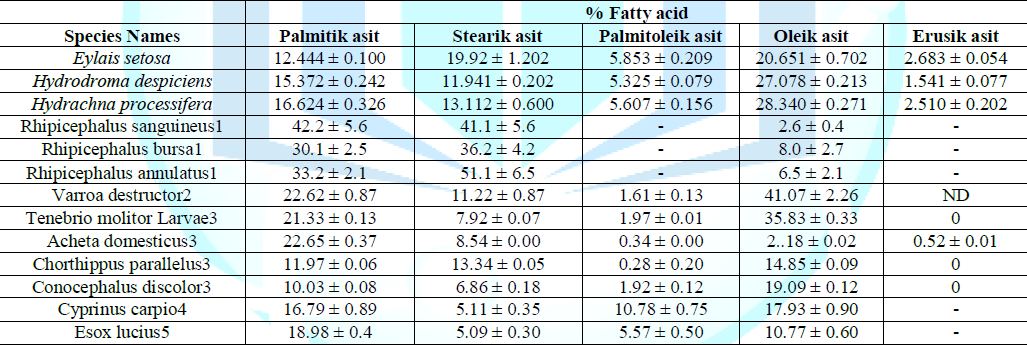
Table 4: Comparison of fatty acid ratios in different species (%).
It appears that the proportions of species close to each other. It is also seen that the proportions of fish and water mites species collected from the same locality are relatively similar. This can be explained by the same locality and seasonal cycle. On the other hand, when we look at the studies on other terrestrial parasites (Rhipicephalus sanguineus, Rhipicephalus bursa, Rhipicephalus annulatus), it is seen that each fatty acid composition compared to the water mites is quite variable for all three species given in the table. The main reason for this is that each of these parasite species is found as a host on a different animal.
When the fatty acid compositions of each species were examined in four different insect species belonging to insect class (Tenebrio molitor Larvae, Acheta domesticus, Chorthippus parallelus, Conocephalus discolor) where there is a great similarity in species belonging to the same team species there is a big difference found between the ratios of both teams. This is due to the fact that these three species belong to different teams of the insect class and therefore differ in their systematic distance and nutrition patterns. According to the table, another noteworthy situation is that the fatty acid composition ratios in two different species of fish caught in the same locality (Cyprinus carpio, Esox lucius) are surprisingly similar. In this case, these observed results for these two species can also be attributed to habitat similarity, seasonal characteristics and nutritional factors.
As a result, it can be said that each of the fatty acid compositions is unique, and that systemic affinity, habitat and seasonal cycles are also effective on these fatty acid compositions. All the results of this study showed that this method can help to solve future taxonomic problems in water mites.
References
1. Bulut S. Fatty acid composition and 6/3 ratio of the pike (Esox lucius) muscleliving in Eber Lake, Turkey (2010) Sci Res Essays 5: 3776-3780.
2. Bulut S, Uysal K, Cemek M, Gok V, Kuş SF, et al. Nutritional evaluation of seasonal changes in muscle fatty acid composition of common carp (Cyprinuscarpio) in Karamık Lake, Turkey (2012) Int J Food Prop 15: 717-724. https://doi.org/10.1080/10942911003664891
3. Smit IM, Cook DR. Ecology and classification of North American fresh water invertebrates (1991) Thorp JH and Covich AP (eds.,) Academic, San Diego, CA, USA, pp: 523-592.
4. Di Sabatino A, Smit H, Gerecke R, Goldschmidt T, Matsumoto N,et al. Global diversity of water mites (Acari, Hydrachnidia; Arachnida) in fresh water (2008) Hydrobiologia 595: 303-315. https://doi.org/10.1007/s10750-007-9025-1
5. Meyer E. Der Entwick lungs zyklusvon Hydrodroma despiciens (O.F. Müller, 1776) (Acari. Hydrodromidae) (1985) Arch Hydrobiol Suppl 66: 321-453
6. Smit H. The water mite family Hygrobatidae Koch in Australia. The genera Aspidiobatella Cook, Australorivacarus Viets, Gondwanabates Imamura and Rhynchaustrobates Cook (Acari: Hydrachnidia) (2015) Zootaxa 4033: 567-583. https://doi.org/10.1007/s10493-013-9713-7
7. Tuzovskij PV, Semenchenko KA. Morphology and taxonomy of deutonymphs of the genus Unionicola Haldeman, 1842 (Acari, Hydrachnidia, Unionicolidae) in Russia (2015) Zootaxa 3994: 69. https://doi.org/10.1007/s10886-005-7109-9
8. Boyacı YO and Gülle P. New records of the water mite family Hydryphantidae (Acari: Hydrachnidia) from Turkey, with the description of a new species (2014) Syst Appl Acarol 19: 160-165. https://doi.org/10.11158/saa.19.2.6
9. Aşçı F, Akkuş GU and Yaman İ. Determination of the ecological impact levels on Hydrodromadespiciens (Müller 1776) (Acari, Hydrachnidia) which is a common water mite in terms of heavy metal applications (2016) Pak J Zool 48: 345-348.
10. Aşçı F, Bahadır M and Akkuş GU. Study on the impact of elements in water on the diversity of water mites (Acari, Hydrachnidia) species (2015) Adv Bio Sci Biotechnol 6: 259-264. https://doi.org/10.4236/abb.2015.64025
11. Onrat ST, Asçi F and Ozkan M. A cytogenetics study of Hydrodroma despiciens (Müller, 1776(Acari: Hydrachnellae: Hydrodromidae) (2006) Genet Mol Res 30: 342-349.
12. Martin P, Koester M, Schynawa L and Gergs R. First detection of prey DNA in Hygrobates fluviatilis (Hydrachnidia, Acari): A new approach for determining predator-prey relationships in water mites (2015) Exp Appl Acarol 67: 373-380.https://doi.org/10.1016/j.cbpa.2007.02.010
13. Dorda BA, Valdecasas AG (2002) Traditional water mite fixatives and their compatibility with later DNA studies. Exp Appl Acarol 34: 59-65.https://doi.org/10.1023/b:appa.0000044439.21180.ec
14. Bohonak AJ, Smith BP and Thornton M. Distributional, morphological and genetic consequences of dispersal for temporary pond water mites (2004) Fresh water Biol 49: 170-180. https://doi.org/10.1111/j.1365-2426.2003.01177.x
15. Ernsting BR, Edwards DD, Vidrine MF, Myers KS and Harmon CM. Phylogenetic relationships among species of the subgenus Parasitatax (Acari: Unionicolidae: Unionicola) based on DNA sequence of the mitochondrial cytochrome oxidase I gene (2006) Int J Acarol 32: 195-202.https://doi.org/10.1080/01647950608684461
16. Więcek M, Martin P and Lipinski A. Water mites as potential long-term bioindicators in formerly drained and rewetted raised bogs (2013) Ecol Indic 34: 332-335.https://doi.org/10.1016/j.ecolind.2013.05.019
17. Yücecan S and Baykan S. Food chemistry, food control and analysis (1981) Vocational and Technical Education Books, Studies and Programming Department Publications, Turkey, pp: 105-124.
18. Farkas T. Adaptation of fatty acid composition to temperature a study on Carp (Cyprinuscarpio L.) liver slices (1984) Comp Biochem Physiol 79: 531-535.
19. Belitz HD and Grosch W. Food Chemistry (1999) Springer, Berlin, Heidelberg, Germany, pp: 152-236.https://doi.org/10.1007/978-3-662-07281-3
20. Karabulut HA and Yandı İ.Su ürünlerindeki omega-3 yağ asitlerinin önemi ve sağlık üzerine etkisi (2006) Ege Üniversitesi Su Ürünleri Dergisi 23: 339-342.
21. Aboshi T, Shimizu N, Nakajima Y, Honda Y, Kuwahara Y, et al. Biosynthesis of linoleic acid in Tyrophagus mites (Acarina: Acaridae) (2013) INS Bioch Mol Bio 43: 991-996. https://doi.org/10.1016/j.ibmb.2013.08.002
22. Peyou-Ndi MM, Watts JL and Browse J. Identification and characterization of an animal Δ 12 fatty acid desaturase gene by heterologous expression in Saccharomyces cerevisiae (2000) Arch Biochem Biophys 376: 399-408.
23. Takada W, Sakata T, Shimano S, Enami Y, Mori N, et al. Scheloribatid mites as the source of pumiliotoxins in dendrobatid frogs (2005) J Chem Ecol 31: 2403-2415. https://doi.org/10.1007/s10886-005-7109-9
24. Zhou X R, Horne I, Damcevski K, Haritos V, Green A and Singh S. Isolation and functional characterization of two independently‐evolved fatty acid Δ12‐desaturase genes from insects (2008) Ins Mol Bio 1: 667-676. https://doi.org/10.1111/j.1365-2583.2008.00841.x
25. Raspotnig G and Krisper G.Fatty acids as cuticular surface components in oribatid mites (Acari: Oribatida), Ebermann, E. [Ed] Arthropod Biology: Contributions to Morphology, Ecology and Systematics (1998) Biosyst Ecol Ser.
26. Shimshoni J A, Erster O, Rot A, Cuneah O, Soback S and Shkap V. Cuticularfatty acidpro file analysis of three Rhipi cephalu stick species (Acari: Ixodidae) (2013) Exp Appl Acarol 61: 481-489. https://doi.org/10.1007/s10493-013-9713-7
27. Folch J, Lees M and Sloane Stanley GH. Hayvan dokularından toplam lipitlerin izolasyonu ve saflaştırılması için basit bir yöntem (1957) J Biol Chem 226: 497-509.
28. Paquot C. Standard methods for the analysis of oils, fats and derivatives (1979) Pure Appl Chem 51: 2503-2526.
29. AOCS. Official methods and recommended practices of the american oil chemists society (2nd edtn) (1972) Is Oil Chem Soc, USA. https://doi.org/10.1007/bf02582467
30. Murungi LK, Kirwa H and Torto B. Differences in essential oil content of berries and leaves of Solanum sarrachoides (Solanaceae) and the effects on oviposition of the tomato spider mite (Tetranychusevansi) (2013) Ind Crop Prod 46: 73-79. https://doi.org/10.1016/j.indcrop.2013.01.022
31. Maazouzi C, Masson G, Soledad Izquierdo M and Pihan JC. Fatty acid composition of the amphipod Dikerogammarusvillosus: Feeding strategies and trophic links (2007) Comp Biochem Physiol 147: 868-875.https://doi.org/10.1016/j.cbpa.2007.02.010
32. Wen X, Chen L, Ku Y and Zhou K. Effect of feeding and lack of food on the growth, gross biochemical and fatty acid composition of juvenile crab, Eriocheirsinensis (2006) Aqua cult 252: 598-607. https://doi.org/10.1016/j.aquaculture.2005.07.027
33. Hayashi K and Takagi T. Seasonal variations in lipids and, fatty acids of Japanese anchovy, Engraulis japonica (1978) Full Faculty of Fisheries, Hokkaido University, Japan 29: 38-47.
Citation:
Aşçi F, İnak M and Bulut S. Systematic evaluation of fatty acid profiles in Hydrachna processifera, Eylais setosa and (Acari, Hydrachnidia) species by GC-MS method (2019) Biochem and Modern Appli 2: 46-50.
*Corresponding author:
Ferruh Aşçi, Department of Molecular Biology and Genetics, Afyon Kocatepe University, Afyonkarahisar, Turkey, Fax no: +902722281235, Email: f_asci@aku.edu.tr
Keywords
Water mite, Acari, Hydrachnidia, Fatty acid composition


 PDF
PDF