Review Article :
Background: Fruits, vegetables
and legumes for their complex carbohydrates, dietary fiber and micronutrients,
should form an essential part of every diet. In order to give good dietary
advice to diabetic patients, it is necessary to know the glycemic index of
foods commonly consumed locally. The objective of this study was to determine
the Glycemic Index (GI) and Glycemic Load (GL) of commonly available and
consumed Guava (Psidium guajava),
Watermelon (Citrullus vulgaris),
Gungo (Cajanus cajan), Papaya (Carica papaya) and tomato (Solanum lycopersicum) in Jamaica. Methods: Ten (10)
healthy Jamaican subjects (5 males, 5 females) with mean age 30 ± 2 years and
mean BMI 25 ± 1 kg/m2 were recruited to the study. Using a
non-blind, crossover design trial, the subjects consumed 50 (or 25) grams of
available carbohydrate portions of glucose (standard food) and test foods after
an overnight fast and their serum glucose levels were determined at 0, 15, 30,
45, 60, 90 and 120 minutes after the consumption of each test food. Glucose was
tested on three separate occasions, and the test foods once. The GI value was
calculated geometrically by expressing the Incremental Area Under the Blood
Glucose Curve (IAUC) for the test foods as a percentage of each subjects
average IAUC for the standard food. Results: The results
indicated that the IAUC for Watermelon (95 ± 11) was significantly higher
(p<0.05) than that of Tomato (37 ± 12), and Gungo (58 ± 13). The differences
in IAUC of Watermelon (95 ± 11), Guava (83 ± 27) and Papaya (80 ± 7) were not
statistically significant. Similarly, there was no significant difference in GI
among the samples studied. Tomato, Gungo,
Papaya Watermelon and Guava were shown to have low glycemic index and glycemic
load values. The
prevalence of chronic
Non-Communicable Diseases (NCDs) are increasing globally and triggering
untimely deaths due to changes in diet and lifestyle. Therefore, effective
strategies for prevention and controlling the spread of these diseases are of
absolute importance [1-3]. It is often
reported that increased consumption of dietary fiber may lead to better control
and management of diabetes mellitus, cancer and cardiovascular diseases.
The physical and chemical profiles of dietary fiber, such as, fibrous structure
and viscosity have a major role in the digestion and absorption of nutrients,
despite the amount of available carbohydrates [4]. The
effect a carbohydrate has on post-prandial blood
glucose concentration is best described by its Glycemic Indices (GI). The
concept of dietary GI was first reported as a factor that should be managed to
prevent chronic diseases, more than three decades ago by researchers at the
University of Toronto, Canada [5]. GI can be defined as a relative ranking of carbohydrates in foods
on a scale of 0 to 100, based on the extent to which they increase blood
glucose levels after consumption [5-8]. Foods with carbohydrates that are
digested, absorbed and utilized quickly are referred to as high glycemic indexed foods
(GI ≥ 70). Those absorbed moderately
(56–69) are referred to as medium
GI foods, while those that take a relatively longer time to break down and
release glucose slowly into the bloodstream are referred to as low GI (GI ≤ 55) foods index [5,9-12]. Further, it
is important to know that both the quantity and the quality of carbohydrates
may affect blood glucose response; this is documented as the glycemic load
[5,8]. Glycemic Load (GL)
investigates the total impact of the dietary carbohydrates on blood glucose
level after a meal. The glycemic load is
computed by determining the product of the GI and the total available
carbohydrate content in a specified portion of food divided by 100. GL values are also categorized as low (≤ 10),
medium (>10 to <20) or high (≥ 20) [10,13,14]. It is recommended that the
GL should be considered when guiding individuals in making healthy food choices
[3,6,15-17]. The
higher the GL of the food, the greater the rise of blood glucose and insulin
levels. Long-term consumption of a high GL diet has been linked with increased
risk of type 2 diabetes and related complications [10,16,18,19]. Research has
shown that consuming low GI foods is a possible inexpensive dietary alternative
in the management of diabetes [6,8,20,21]. The nutritional benefits of fruits, vegetables
and legumes make them a good choice for weight and health management [3,22].
Therefore, it is important that the GI and GL of our fruits, vegetables and
legumes be determined to better guide the choices of the consumer. This study
investigated the glycemic
index and glycemic load of commonly available and consumed fruits,
vegetables and legumes in Jamaica. Food
samples Freshly
harvested, Guava (Psidium guajava),
Watermelon (Citrullus vulgaris), Tomato
(Solanum lycopersicum), Gungo (Cajanus cajan) and Papaya (Carica papaya) were sourced from a local
market in Kingston, Jamaica. Methods Adult
Jamaican subjects between the ages of 25 to 45 years were recruited among the
students and staffs of the University of the West Indies (Mona Campus). Ten
healthy subjects, with an active lifestyle, not using any prescribed medication
and without any diagnosed diseases were selected for study. Exclusion criteria
were as follows: Smokers, overweight, obese, diabetic individuals and pregnant
or lactating women. Proximate
analysis for carbohydrate, fat, crude protein, moisture, dietary fiber
content and ash were determined using the AOAC (2002) standard. Total
carbohydrate was done by difference according to FAO/WHO Expert Consultation
protocol (Food and Agriculture Organization & World Health Organization,
1998). Proportion of fruits equivalent
to 50 (or 25) grams of available carbohydrate was fed to subjects after an
overnight fast and their serum glucose levels were determined at 0, 15, 30, 45,
60, 90 and 120 minutes for each test food on different days during the study. The
Incremental Areas under the Curve (IAUC) were calculated according to the method
of Brouns et al. [13]. Pure glucose was used as the standard, which was assigned
a GI of 100. Glucose was tested on three separate occasions, and the test foods
once. The GI rating (%) for each food, was calculated for each subject by
expressing the IAUC of the test food as a percentage of the average IAUC of the
glucose standard consumed by that volunteer [23]. The protocol was approved by
the Ethics Committee of the University Hospital of the West Indies and the
Faculty of Medical Sciences at the University of the West Indies Mona Campus,
Kingston, Jamaica (Ethical approval number: AN 14, 12/13). Statistical
analysis Data
obtained from the experiments are expressed as mean ± SE. Differences between
the control and the treatments in the experiments were analyzed using ANOVA and
Duncans multiple range tests, while values of P ≤ 0.05 were considered
significant. The
10 Jamaican subjects, comprising five (5) males and five (5) females were
between ages 25 and 45 years with a mean age of 30 ± 2 years and BMI 25 ± 1
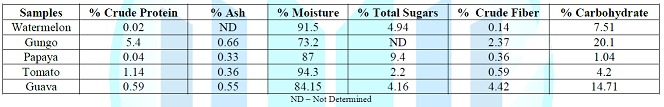
kg/m2. Table 1 represents
the proximate compositions of the foods studied. Gungo was found to have the
highest crude protein content (5.4 [g/100g]), while Watermelon had the lowest
(0.02 [g/100g]). The crude protein content of the other samples ranged from
0.04 to 1.14 [g/100g]. Table 1: Proximate composition of five food samples (100g) studied. The
moisture content of the foods was highest in Tomato (94.3%) and lowest in Gungo
(73.2%). Papaya was found to have the highest total sugars (9.4%) and Tomato
the lowest (2.2%). Crude fiber content
was highest in Guava (4.42%), while Watermelon had the lowest crude fiber
content of 0.14%. The carbohydrate
content of the food samples ranged from 1.04 [g/100g] to 20.10 [g/100g] with
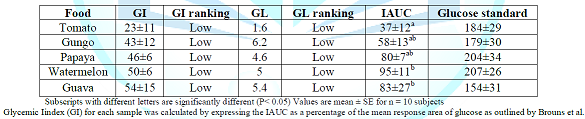
Papaya having the lowest and Gungo the highest content. Table 2 shows the GI values of the food samples determined relative
to the reference food (glucose GI=100) and categorized as high (70 to 100),
intermediate (56 to 69), or low (<55).
The GI of the test food samples ranged from 23 ± 11 to 54 ± 15. Tomato was observed to have the lowest of 23
± 11; this was followed by Gungo with a value of 43 ± 12. The highest GI was observed in Guava (54 ±
15). Similarly, the GL (high ≥ 20, medium 11-19 and low ≤ 10) ranged from 1.6
to 6.2. Tomato was observed to have the lowest GL of 1.6 and the highest GL was
observed in Gungo (6.2). In addition,
Watermelon showed the highest incremental area under (IAUC) the glucose
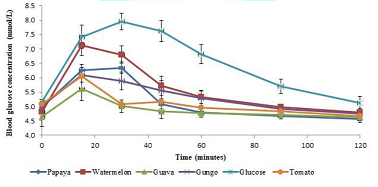
response curve of 95 ± 11 and Tomato the lowest with 37 ± 12. Figure 1 illustrates the mean glucose
responses of the five food samples studied.
The blood glucose response to the food samples increased with time,
reaching their peak at approximately 15 minutes except for Papaya that peaked
at 30 minutes, after which a decline in the response with increasing time was
observed. Values represented as mean ± SE for n = 10 subjects. It
has long been recognized that “not all carbohydrates are created equal” with
regard to their effects on glucose metabolism and insulin action [24,25]. Furthermore,
it is understood that different complex carbohydrates could have different physiological effects. Food with high GI is reported to have a
deleterious effect on health and therefore should be avoided [1,3]. This
study was done to determine the glycemic indices of three fruits, a vegetable
and a legume that are frequently consumed in the Caribbean, thus contributing
to the Caribbean
Glycemic Index Database. The
glycemic indices of the selected foods ranged from 23 to 54 (Table 2). The results showed that at fixed quantities
of available carbohydrates, there were distinct variations in the glucose
response. This supports the knowledge that equal carbohydrate portions of
different foods can display different glycemic response on human subjects [25].
To give good dietary
guidance, it is important to know the glycemic index of the food consumed
in different ethnic groups. In this study the GI (54) and GL (5.4) of Guava was
determined to be low. Similar findings
were documented in Brazil, where researchers reported low GI (12) and GL (1.1)
of Guava samples studied [3]. Further, the legume Black-eyed beans (Cowpeas)
were documented to have low GI (34) and GL (10) in a study conducted in Canada [5],
while researchers Sabeetha, Nisak and Barakatun [26] determined the GI of
Watermelon to be low (51) which is consistent with the findings of this and
other studies conducted [27]. However, in Australia the GI (56) of Papaya was reported to be medium and GL (5)
low in work done by researchers Miller et al., while in this study the GI (46)
and GL (4.6) of Papaya were both determined as low. It
is important to mention that the GI values of the same type of fruits
cultivated in different geographical location may vary and could be due to the
environmental conditions or difference in sugar composition, fiber content,
stage of ripening, acid content, method of storage and harvesting or it could
be the methods used to determine the GI of the test foods [3,27-29]. Tomato had the lowest GI and AUC (23 ± 11 and
37 ± 12 respectively), this could be due to the fiber content. The dietary fiber could influence
the digestion and adsorption of the carbohydrate present and thereby influence
blood glucose response [30,31]. In Figure
1, the test foods displayed a high initial peak at approximately 15
minutes, followed by a gradual decrease in blood glucose. The relatively low
glucose peak displayed by Tomato, Gungo and Papaya may be promising results in
terms of their recommendation to patients with diabetes and its associated
metabolic dysfunctions or other NCDs [3,6,8,21]. From
the present study, the glycemic index of Tomato (23), Gungo (43) and Papaya
(46), Watermelon (50) and Guava (54) were shown to have low glycemic index and
glycemic load values.
It is reported that reduce consumption of high GI foods and increase the intake
of low and intermediate GI may lead to better management of diabetes, coronary
heart disease and obesity [10,11]. Therefore, it is important that low and
intermediate GI foods be identified, and their consumption recommended. It
is our pleasure to express profound gratitude to the Faculty of Medical
Sciences (UWI), Biotechnology Centre (UWI), Scientific Research Council (SRC),
National Health Fund (NHF) and UWI Yam Group for their contributions to this
study. 1.
Adedayo
BC, Oboh G and Akindahunsi AA. Estimated glycemic indices and inhibitory action
of some yam (Dioscorea spp.) products
on key enzymes linked with type 2 diabetes (2015) Futa J Res in Sci 11:25-35. 2.
Ojo
O. Nutrition and chronic conditions (2019) Nutr 459:1-6. http://doi:10.3390/nu11020459 3.
Passos
TU, Sampaio HADC, Sabry, MOD, Melo MLPD, Coelho MAM, et al. Glycemic index and
glycemic load of tropical fruits and the potential risk for chronic diseases
(2015) Food Sci Techn (Campinas) 35:66-73. http://dx.doi.org/10.1590/1678-457X.6449 4.
Fatema
K, Rahman F, Sumi N, Kobura K, Afroz A and et al. Glycemic and insulinemic
responses to pumpkin and unripe papaya in type 2 diabetic subjects (2011) Int J
Nutr Metab 3:1-6. 5.
Jenkins
DJ, Wolever TM, Taylor RH, Barker H, Fielden H, et al. Glycemic index of foods:
A physiological basis for carbohydrate exchange (1981) Am J Clin Nutr 34:362-366. https://doi.org/10.1093/ajcn/34.3.362 6.
Bhupathiraju
SN, Tobias DK, Malik VS, Pan A, Hruby A, et al. Glycemic index, glycemic load,
and risk of type 2 diabetes: results from 3 large US cohorts and an updated
meta-analysis (2014) Am J Clin Nutr 100:218-232. http://dx.doi.org/10.3945/ajcn.113.079533 7.
Foster-Powell
K, Holt SH and Brand-Miller JC. International table of glycemic index and
glycemic load values: 2002 (2002) Am J Clin Nutr 76:5-56. https://doi.org/10.1093/ajcn/76.1.5 8.
Ludwig
DS. The glycemic index: physiological mechanisms relating to obesity, diabetes,
and cardiovascular disease (2002) Jama 287:2414-2423. 9.
Atayoglu
AT, Soylu M, Silici S and Inanc N. Glycemic index values of monofloral Turkish
honeys and the effect oftheir consumption on glucose metabolism (2016) Turkish
J Med Sci 46:483-488. https://doi.org/10.3906/sag-1502-102 10.
Augustin
LS, Kendall CW, Jenkins DJ, Willett WC, Astrup AA, et al. Glycemic index,
glycemic load and glycemic response: an International Scientific Consensus
Summit from the International Carbohydrate Quality Consortium (ICQC) (2015) Nutr
Metab Cardiovasc Diseases 25:795-815. https://doi.org/10.1016/j.numecd.2015.05.005 11.
Brand-Miller
J, Holt S, Pawlak D and McMillan J. Glycemic Index and Obesity (2002) Am J Clin
Nutr 76:281-285. 12.
Rahelić
D, Jenkins A, Božikov V, Pavić, E, Jurić K, et al. Glycemic index in diabetes
(2011) Collegium Antropologicum 35:1363-1368. 13.
Brouns
F, Bjorck I, Frayn KN, Gibbs AL, Lang V, et al. Glycemic Index Methodology
(2005) Nutr Res Rev 18:145-171. https://doi.org/10.1079/NRR2005100 14.
Kouamé
CA, Kouassi NK, Coulibaly A, Ndri DY, Tiahou GG, et al. Glycemic index and
glycemic load of selected staples based on rice, yam and cassava commonly
consumed in Côte dIvoire (2014) Food Nutr Sci 5:308-315. http://dx.doi.org/10.4236/fns.2014.54037 15. Barclay
AW, Petocz P, McMillan-Price J, Flood VM, Prvan T, et al. Glycemic index,
glycemic load, and chronic disease risk—a meta-analysis of observational
studies (2008) Am J Clin Nutr 87:627-637. https://doi.org/10.1093/ajcn/87.3.627 16.
Esfahani
A, Wong JM, Mirrahimi A, Srichaikul K, Jenkins DJ, et al. The glycemic index:
physiological significance (2009) J Am Coll Nutr 28:439-445. 17.
Sluijs
I, Beulens JW, van der Schouw YT, Buckland G, Kuijsten A, et al. Dietary
glycemic index, glycemic load, and digestible carbohydrate intake are not
associated with risk of type 2 diabetes in eight European countries (2013) J Nutr
143:93-99. https://doi.org/10.3945/jn.112.165605 18.
Miller
JC. Importance of glycemic index in diabetes (1994) Am J Clin Nutr 59:747-752. https://doi.org/10.1093/ajcn/59.3.747S 19.
Willett
W, Manson J and Liu S. Glycemic index, glycemic load, and risk of type 2
diabetes (2002) Am J Clin Nutr 76:274-280. 20.
Bahado-Singh
PS, Wheatley AO, Ahmad MH, Morrison EY and Asemota HA. Food processing methods
influence the glycemic indices of some commonly eaten West Indies
carbohydrate-rich foods (2006) British J of Nutrition 96:476-481. https://doi.org/10.1079/BJN20061792 21.
Sun L, Lee DEM, Tan WJK, Ranawana DV, Quek YCR, et
al. Glycaemic index and glycaemic load of selected popular foods consumed in
Southeast Asia (2015) Br J Nutr 113:843-848. https://doi.org/10.1017/S0007114514004425 22.
Fatema
K, Sumi N, Rahman F, Kobura K and Ali L. Glycemic index determination of
vegetables and fruits in health Bangladeshi subjects (2011) Malays J Nutr 17:393-399. 23.
Karthik
D and Ravikumar S. A Study on the Protective Effect of Cynodon dactylon Leaves
Extract in Diabetic Rats (2011) Biomed Environ Sci 24:190-199. https://doi.org/10.3967/0895-3988.2011.02.014 24.
Bahado-Singh
PS, Riley CK, Wheatley AO and Lowe HI. Relationship between processing method
and the glycemic indices of ten sweet potato (Ipomoea batatas) cultivars
commonly consumed in Jamaica (2011) J Nutr Metabo 1-6. http://dx.doi.org/10.1115/2011/584832 25.
Francis
R, Bahado-Singh PS, Smith A, Wheatley AO and Asemota HA. Glycemic index of
traditional foods in Jamaica (2018) Eur J Exp Bio 8:1-5. https://doi.org/10.21767/2248-9215.100056 26.
Sabeetha
S, Nisak MY and Amin I. Glycemic index of selected watermelon (Citrullus lanatus) (2018) Int Food Res J
25:2547-2552. 27.
Robert
SD, Ismail AAS, Winn T and Wolever TM. Glycemic index of common Malaysian
fruits (2008) Asia Pac J Clin Nutr 17:35-39.
28.
Premanath
M, Gowdappa HM, Mahesh M and Babu MS. A study of glycemic index of ten Indian fruits
by an alternate approach (2011) E-Int Sci Res J 3:11-18. 29.
Ha
MA, Mann JI, Melton LD and Lewis-Barned NJ. Relationship between the glycaemic
index and sugar content of fruits (1992) Diabetes Nutr Metab 5:199-203. 30.
Truswell
AS. Glycemic index of foods (1992) Eur J Clin Nutr 46:91-101. 31.
Chang
KT, Lampe JW, Schwarz Y, Breymeyer KL, Noar KA, and et al. Low glycemic load
experimental diet more satiating than high glycemic load diet (2012) Nutr Cancer
64:666-673. https://doi.org/10.1080/01635581.2012.676143 Asemota HN, Department of Basic
Medical Sciences, The University of West Indies, Mona Campus, Jamaica, West
Indies, Tel: 876-927-1828, E-mail: helen.asemota@uwimona.edu.jm Glycemic index,
Glycemic load, Diabetes, GlucoseGlycemic Index of Selected Foods in Jamaica
Ryan D Francis, Perceval S Bahado-Singh, Andrew
O Wheatley, Ann Marie Smith and Helen N Asemota
Abstract
Full-Text
Introduction
Materials
and Methods
Results
Discussion
Conclusion
Acknowledgment
References
*Corresponding
author
Citation
Keywords