Isolated Coronary Artery Stenoses (CAS) involving
the ostium of the Left Anterior Descending (LAD) artery is very challenging,
especially in a setting of primary Angioplasty in Myocardial Infarction (PAMI).
Intimal atherosclerosis in the Left Main Coronary Artery (LMCA) bifurcation is
primarily in area of low shear stress which is the lateral wall, close to the
LAD and Left Circumflex (LCx). Thus, carina is usually free of disease, which
can explain why single-stent strategy can be successful. However, precise stent
placement is often difficult due to unwanted stent movement within vessel or
its proximity to side branches. A decision must be made at the outset, to
decide on the approach to be employed, to treat osteal LAD lesions. Limited
data is available on patients undergoing primary PCI of osteal LAD lesions.
Here, we present our experience and problems encountered during the management
of osteal LAD lesions in the setting of PAMI.
Introduction
Ostial Left Anterior Descending Coronary Artery
(LAD) lesions were for long regarded as those clinical subset that are unsuitable
for coronary stenting. An ostial stenosis is defined as angiographic narrowing
of ≥ 70% located within 3 mm of a vessel origin. Ostial LAD artery lesion
presenting with acute anterior wall myocardial infarction is not uncommon [1].
Ostial lesions tend to be more fibrotic, calcified and rigid. This causes
increased vessel recoil post angioplasty, resulting in higher rates of repeat
revascularization. It has been shown that >70% of LMCA lesions involve the
distal bifurcation [2].
On conventional angiography, the lesion may appear
localised to ostium of LAD, but in most cases, the plaque also extends into the
LMCA or Left circumflex (LCx) artery. No specific guidelines have been
formulated for Primary Angioplasty in Myocardial Infarction (PAMI) in ostial
LAD lesions. Yakushiji, et al [3] have reported that osteal LAD plaque was
continuous from LMCA in 96% cases and from the LMCA into circumflex ostium in
78% cases and from the LMCA into both LAD and circumflex in 74% cases. This
evidence may be considered during ostial stenting of LAD or LCx [3,4]. Here we
report two cases of osteal LAD stenting in PAMI, detailing the procedure and
complications, both immediate and long term, for better understanding of the
disease progression.
Case Reports
Case study-1
A 53-year old male, with history of hypertension and
dyslipidemia presented with chest pain of 2hrs duration. Vitals were stable.
ECG is suggestive of Anterior Wall Myocardial Infarction (AWMI) or anterior
STEMI (ST Segment Elevation Myocardial Infarction). Patient was immediately
transferred to cardiac catheterization lab after giving loading doses of
aspirin (325 mg), ticagrelor (180 mg) and atorvastatin (80 mg). 2D Echo showed
hypokinesia of anterior wall of left ventricle with preserved wall thickness.
LV ejection fraction was 40%. There was no mitral valve regurgitation. Coronary
Angiography (CAG) showed total thrombotic occlusion of LAD from ostium with
mild non-occlusive plaques in mid LMCA and proximal segment of a non-dominant
left circumflex artery (Figure 1a, 1b).
Dominant Right Coronary Artery (RCA) was normal.
No collaterals to LAD could be demonstrated on RCA angiogram. It was decided to
perform Percutaneous Transluminal Coronary Angioplasty (PTCA) of LAD from its
ostium, which was the culprit artery. Initial bolus of integrilin
(eptifibatide) and weight adjusted dose of Low-Molecular Weight Heparin (LMWH)
was given intravenously. Thrombosuction of LAD with Thrombuster 6F catheter was
done. Successful PTCA of LAD was performed after balloon dilatation with 2.5 mm
x 10 mm NC balloon and stenting of LAD from ostium with a 3 mm x 18 mm Drug
Eluting Stents (DES). Final angiogram showed successful PTCA to LAD, with
TIMI-3 flow, no residual stenosis, no dissection or flow limitation observed.
Mild plaques in LMCA and LCx were similar to pre-PTCA angiogram (Figure 1c,d).
Patient was hemodynamically stable and pain free. He was transferred to ICCU in
stable condition.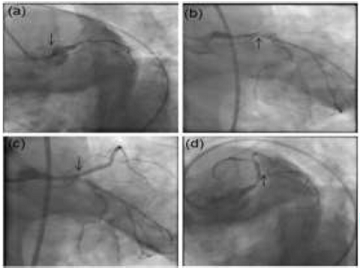
Figure 1: CAG showing total occlusion of LAD with haziness
However, immediately after transfer to Intensive
Cardiac Care Unit (ICCU) he had a episode of projectile vomiting with hypotension
and ST segment depression in V1 to V4 chest leads on ECG. Patient was rushed
back to catheterization laboratory. Coronary angiography performed showed
patent LAD stent, but LCx showed a sluggish flow with Thrombolysis in
Myocardial Infarction (TIMI-2) probably due to plaque shift (Figure 2a-c).
Patient developed cardiac asystole on table with a flat ECG trace. Repeat
angiography now showed near total thrombotic occlusion in the LMCA and
circumflex with sluggish flow in LAD (TIMI-1). Cardiopulmonary Resuscitation
(CPR) was initiated and inotropic supports, along with temporary transvenous
pacing and endotracheal intubation/ventilation. Intra-Aortic Balloon Pump
(IABP) was inserted from the left femoral artery.
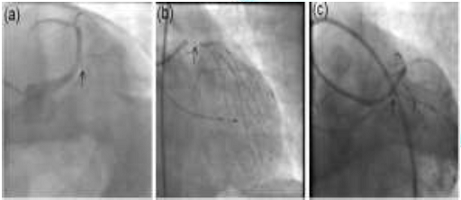
Figure 2: Repeat CAG shows patent stent in LAD (a-c) near total occlusion of circumflex artery (arrows).
Unfortunately, there was no response to any of the
above measures. Decision was made to proceed immediately with PTCA to LCx and
LMCA. Using Double Kiss (DK) crush technique, stenting of LCx was performed with
a 3.0x15 mm DES and LM to LAD stenting done, overlapping the older LAD stent with
a 3.5 x 18 mm DES. Proximal Optimisation Technique (POT) with a 4 x 10 mm NC
balloon was done in the LMCA (Figure 3a-d). Patient’s vital parameters improved
soon after. Eventually patient was weaned off IABP, inotropic and ventilator
supports in next 72 hrs. Subsequent recovery was uneventful. CAG repeated after
12 months showed patent stents in LMCA, LAD and LCx.
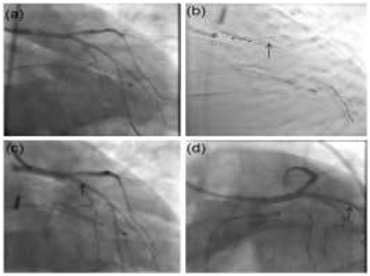
Figure 3: PTCA to Left circumflex and LMCA using DK crush method (a,b). Final CAG shows patent stents in LM and LCx and LAD (c,d) (arrows).
Case Study-2
A 62-year-old female with history of diabetes and
hypertension presented with chest pain of 3 hrs duration. ECG is suggestive of
acute anterior wall myocardial infarction. 2D-Echo showed anterior wall
hypokinesia with Left Ventricular Ejection Factor (LVEF) of 35%. Patient was
given loading dose of aspirin (325 mg), ticagrelor (180 mg) and atorvastatin
(80 mg). Coronary angiography showed total thrombotic occlusion of ostial LAD,
minor plaques in ostium of a large dominant LCx (Figure 4a, 4b). Thrombosuction
of LAD with EXPORT 6F catheter was done. Intra-Vascular Ultra Sonography (IVUS)
showed stenosis mainly involving LAD ostium with calcification and minor plaque
extension into the LMCA. A 3 x 12 mm Flextome cutting balloon was used to
prepare ostial LAD lesion and then stented with 3.5 mm x 18 mm DES. Final
result showed good stent apposition in LAD with mild haziness noticed in the
ostium of the LCx, likely due to carinal shift (Figure 4c, 4d).

Figure 4: CAG shows total ostial occlusion of LAD (a) with minor plaques
Post-PTCA IVUS assessment showed well apposed stent
in LAD with few stent struts overhanging or protruding into lumen of LMCA (Figure
5a-d). She made a good recovery and was later discharged. However, she
developed exertional angina 8 months later. ECG and troponin-I levels were
normal. Treadmill ECG stress Test (TMT) was strongly positive at 5 minutes of
exercise. 2D-Echo was normal. Repeat coronary angiography showed 90% ostial
circumflex stenosis, with mild increase in LMCA plaque lesion when compared to
the previous CAG. The stent in the LAD was patent with TIMI-3 flow (Figure 6a, 6b).
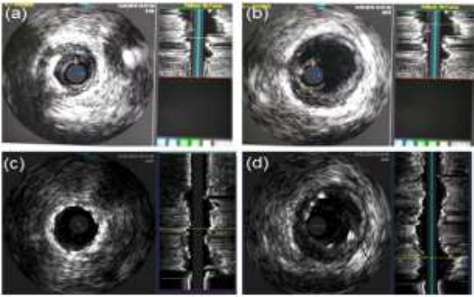
Figure 5: IVUS assessment of LMCA and ostial LAD
Figure 6: Repeat CAG was done 8 months later for exertional angina
Optical Coherence Tomography (OCT) was performed
which showed well endothelialized LAD stent with excellent ostial coverage and
stent apposition, but with few struts overhanging LCx ostium and presence of mixed
plaque partially occluding the ostium of the circumflex (Figure 7a-c). Using DK
crush technique, LCx stenting was done with 3.0 x 12 mm DES and LM to LAD
stenting with 3.5 x 12 mm DES with final kissing balloon angioplasty and POT in
LM. Patient was hemodynamically stable and was discharged after three days. She
is on a regular follow-up and is asymptomatic.
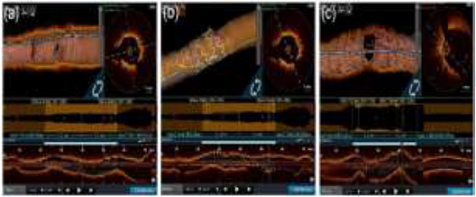
Figure 7: OCT analysis showing good coverage of LAD ostium
Discussion
Ostial LAD lesions have been an enigma for a long
time. Various techniques have been described to treat osteal lesions, with good
short and long-term results. A large percentage of ostial LAD plaques are not
confined, but also extend into distal LM and/or LCx. Various imaging techniques
such as IVUS and OCT give an excellent perspective with regards to plaque
morphology, location and its extension. Objective of revascularisation in PAMI
is complete and rapid establishment of TIMI-3 flow in culprit vessel to reduce
myocardial injury. Primary PCI is the preferred reperfusion strategy in
patients with STEMI within 12 hrs of symptom onset, provided it can be
performed expeditiously. Coronary stenting is the technique of choice during
PAMI. Focal stenting of culprit lesion at ostium seems to be obvious choice,
but this may result in disastrous short or long term consequences as
demonstrated by above two cases. Aggressive lesion preparation in fibrotic,
calcific ostial lesions using cutting balloons and non-compliant or high
pressure balloons is essential for proper stent deployment.
Extension of dissection into LMCA or circumflex,
plaque shift into circumflex ostium, carinal shift, thrombus embolization and
inability to recognize primary plaque rupture extending into adjacent vessels
are some of the problems associated with osteal PTCA. Relying only on visual
assessment during angiography before performing osteal PTCA has its drawbacks.
Hence, intravascular imaging may be the better option in such cases. However,
intravascular imaging may not be available or not cost-effective and is often time
consuming especially in emergency situation like primary angioplasty. Other
factors such as coronary dominance also play a role in prognosis. Dominant left
coronary circulation interventions have poor post-revascularisation outcomes
due to dependence of one single coronary artery supplying a large myocardial
territory [5].
Various techniques of both single vessel and
bifurcation stenting have been described [6-8]. Floating-stent technique [9].
Szabo method [6] “inverted" provisional T stenting, T-stent and small
protrusion (TAP stenting), ‘V’ stenting, the cross-over stenting with or
without Final Kissing Balloon (FKB), Culotte, DK crush and new dedicated ostial
stents (e.g. CAPELLA SIDEGUARDTM) are some of the techniques in use. POT is
recommended in all cases after Stent side-cell re-cross and kissing balloon
inflation.
Plaque shift has been considered as the main
underlying mechanism for side-branch compromise following main vessel stenting,
but current IVUS studies suggest that side branch ostial stenosis after stent
implantation may also be as the result of carina shift, a phenomenon that may
not result in functional flow limitation. In such cases, side branch balloon
dilatation should be performed when there is a suboptimal result at the side
branch ostium on angiogram. Fractional Flow Reserve (FFR) assessment in
patients with evidence of severe side-branch ostial compromise on angiography
after cross-over stenting is often needed to assess the degree of vessel
compromise.
Option of provisional side-branch stenting should be
considered when patient is significantly symptomatic with fresh ECG changes
suggestive of coronary ischaemia or haemodynamic instability. FFR evidence of
physiologically significant lesion or life-threatening arrhythmias have good
corroborative value. Poor outcomes include >75% lesion in side-branch, TIMI
flow <3, dissection, residual ostial circumflex vessel with MLA <4 mm2
after stenting or FFR <0.8 after provisional stenting. In complex lesions
with pre-PTCA evidence of critical disease in both LAD and circumflex ostia,
planned bifurcation stenting is a better option. If circumflex lesion is >70%
and extends for more than 10 mm beyond ostium, bifurcation stenting is a clear
choice.
Ostial stent deployment is often complicated by the
influence of cardiac and respiratory motion, which causes the stent to
oscillate back and forth during the cardiac or respiratory cycle. Breath-holding,
shallow-breathing, pharmacological agents (such as esmolol, adenosine and
atropine) or rapid ventricular pacing are some of the techniques in use to aid
in accurate stent deployment at the ostium. Other methods include low-pressure
inflation of the balloon on which the stent is mounted. This stabilizes the
stent within the stenosis, while permitting adjustment of the stent to osteal
location prior to deployment. LAD stent can also be stabilized by simultaneous
balloon placement in the side branch to prevent unwanted stent movement.
Excessive protrusion of LAD stent into LMCA makes re-wiring, balloon-stent
crossing into side-branch difficult if bifurcation stenting is planned. This is
further compounded in trifurcations with moderate sized ramus or early obtuse
marginal branch (>2 mm) which may get compromised due to jailing of vessels.
DK crush technique is superior to Provisional
Stenting (PS), minicrush and culotte [10] in certain side branch anatomy.
Results of DK CRUSH-V study, 2019 have shown that Target Lesion Failure (TLF)
at 3 years after DK crush stenting was 8.3% compared to 16.9% for PS in
unprotected left main distal bifurcation lesions. Furthermore, definite or
probable Stent Thrombosis (ST) rates at 3 years were 0.4% in the DK crush group
compared to 4.1% in the PS group. DK crush technique is associated with a
significant reduction in both primary and secondary endpoints for patients with
complex bifurcation lesions [10]. However, DK crush technique has many steps
and is challenging to perform. It involves a steeper learning curve before
optimal results are achieved. Teaching and adequate practice along with
intravascular image interpretation is mandatory for optimal results.
Deferred stenting in primary PCI has also been
investigated as an option to reduce Microvascular Obstruction (MVO) and
preserve microcirculatory function. Ostial LAD occlusion in setting of STEMI is
treated immediately to establish TIMI-3 flow using thrombosuction, balloon
dilatation, and/or GPIIb /IIIa inhibitors, and subsequently after few hours,
with IVUS or OCT imaging and stenting if required.
Learning
Points from the Cases
- One should not ignore stenosis
or thrombus in the side-branch when treating the occluded main (culprit)
artery as restenosis rates at the ostium of the side branch is high
.Furthermore, disease of LAD ostium is usually continuous with ostial disease
of LAD and LCx and vice versa. Performing a bifurcation stenting could
have averted potential complications in the first case. Once TIMI-3 flow is
established in Infarct Related Artery (IRA), re-assessment of anatomy and
need for intra-vascular imaging should be considered.
- In STEMI, PTCA to IRA is the
preferred reperfusion strategy. Multi-Vessel Disease (MVD) is present in
about 50% of patients with STEMI. Intervention should be performed on
culprit vessel or all major occlusive lesions (>50% stenosis).
Non-culprit lesions may also be biologically active, inflamed and
potential targets for thrombosis. It is imperative to emphasize the
importance of clinical judgment when deciding on performing multi-vessel
PCI or staged multivessel PCI, or just Plain Old Balloon Angioplasty
(POBA) or referral for Coronary Artery Bypass Surgery (CABGS).
- In the presence of persistent
ischaemia after culprit vessel PTCA or when patient continues to have chest
pain or is haemodynamically unstable or if the operator is uncertain
regarding the culprit vessel involved or the non-culprit lesion appears
unstable or thrombotic, it is imperative to revascularize the most critical
lesions, even if it implies performing a more complex bifurcation stenting
[11].
- Mild to moderate side-branch
stenosis post stenting due to plaque or carinal shift or spasm should not
be neglected, even if there are no symptoms or ECG changes. Imaging of
side-branch or FFR can help in assessment of severity of lesion in order
to decide on various treatment options.
Conclusion
Each case is different and strategy should be
customised based on anatomy and pathophysiology of the lesion with
institutional or operator expertise. Simple ostial LAD
lesions can be managed with focal stenting only. It is apparent that one may
adopt a more aggressive approach and perform stenting from LMCA to LAD based on the main vessel and side branch
anatomy. Crossover stenting is commonly used technique to treat significant
distal LM or ostial LAD disease,
in the absence of angiographically significant ostial LCx disease. Adequate
lesion preparation prior to stenting is mandatory in all cases. However, low
threshold to bifurcation stenting in the event of complications will have
better short as well as long term results.
References
- Karabulutan A and Cakmak M.
Treatment strategies in the left main coronary artery disease associated
with acute coronary syndromes (2015) J Saudi Heart Assoc 27: 272-276. https://doi.org/10.1016/j.jsha.2015.03.002
- Ramadan R, Boden WE and
Kinlay S. Management of left main coronary artery disease (2018) J Am
Heart Assoc 7: e008151. https://doi.org/10.1161/JAHA.117.008151
- Yakushiji T, Maehara A,
Mintz GS, Saito S, Araki H, et al. An intravascular ultrasound comparison
of left anterior descending artery/first diagonal branch versus distal
left main coronary artery bifurcation lesions (2013) Euro Intervention 8:
1040-1046. https://doi.org/10.4244/eijv8i9a160
- Rigatelli G, Zuin M, Baracca
E, Galasso P, Carraro M, et al. Long-term clinical outcomes of isolated
ostial left anterior descending disease treatment: Ostial stenting versus
left main cross-over stenting (2019) Cardiovasc Revasc Med 20: 1058-1062. https://doi.org/10.1016/j.carrev.2019.01.030
- Kuno T, Numasawa Y, Miyata
H, Takahashi T, Sueyoshi K, et al. Impact of coronary dominance on
in-hospital outcomes after percutaneous coronary intervention in patients
with acute coronary syndrome (2013) PLoS ONE 8: e72672. https://doi.org/10.1371/journal.pone.0072672
- Shengli Y. Safety and
feasibility of Szabo technique in percutaneous coronary intervention of
ostiallesions (2012) Heart 98: E210-E211. https://doi.org/10.1136/heartjnl-2012-302920l.30
- Sulaiman MJ and Chen SL.
Intravascular ultrasound-guided percutaneous coronary intervention in left
main coronary bifurcation lesions: a review (2017) Res Reports Clinical
Cardiol 8: 49-59. https://doi.org/10.2147/rrcc.s140850
- Fajadet J, Capodanno D and
Stone GW. Management of left main disease: an update (2019) Eur Heart J
40: 1454-1466. https://doi.org/10.1093/eurheartj/ehy238
- Medina A, Martín P, Suarez
de Lezo J, Amador C, Suarez de Lezo J, et al. Vulnerable carina anatomy
and ostial lesions in the left anterior descending coronary artery after
floating-stent treatment (2009) Rev ESP Cardiol 62: 1240-1249. https://doi.org/10.1016/s1885-5857(09)73351-1
- Chen SL, Xu B, Han YL,
Sheiban I, Zhang JJ, et al. clinical outcome after dk crush versus culotte
stenting of distal left main bifurcation lesions, the 3-year follow-up
results of the DKCRUSH-III study (2015) JACC: Cardiovascular Interventions
8: 1335-1342. https://doi.org/10.1016/j.jcin.2015.05.017
- Tamis-Holland JE and
Suleiman A. The Management of MVD in STEMI: The science and art of
decision-making in STEMI (2018) Expert Analysis: Latest in Cardiology ACC.
*Corresponding author
Charan
Reddy KV, Department of Clinical and Interventional Cardiology, Lilavati
Hospital and Research Centre, Mumbai, India, E-mail: chrnr@rediffmail.com
Citation
Sanzgiri P, Reddy KVC, Thanedar R and Srinivas
K. Clinical profile and approach to osteal lad lesion during primary angioplasty
in myocardial infarction (PAMI) (2020) Clinical Cardiol Cardiovascular Med 4: 16-19.