Introduction
Stress, a main contributor to various psychiatric and neurodegenerative disorders, leads to cellular death through destruction of biomolecules such as DNA. Stress-induced detrimental effects are mediated by oxidative and nitrosative stress i.e., increased lipid peroxidation and production of free radicals such as Reactive Oxygen Species (ROS) and Nitric Oxide Species (NOS) [1]. Free radicals stimulate neuroinflammation via activation of inflammatory pathways, which involve stimulation of inflammasome - a key component that activates IL-1β and IL-18 [2]. On the other side, activation of inflammatory pathways results in further internal NO cellular production [3].
In addition, stress suppresses hippocampal neurogenesis; significantly smaller hippocampi of depressed patients compared with healthy individuals indicate sub-optimal neurogenesis [4]. Research documents that stress-related mood disorders (e.g., depression and anxiety) are associated with impaired function of hippocampal Brain-Derived Neurotrophic Factor (BDNF), a member of the neurotrophin family of neurotrophic factors that plays roles in neurogenesis as well as preservation of neuronal function and plasticity during development and adulthood [5].
Unfortunately, the mechanism through which psychiatric treatments such as antidepressants work results in byproducts that stimulate oxidative stress, activate inflammatory pathways such as nuclear factor-κB (NF-κB), alter neuroplasticity and neurotrophins, impair mitochondrial function and neuronal bioenergetics; and cause DNA damage, apoptosis, and cell death. Hence, the available psychiatric drugs fail to produce the intended therapeutic goals and further result in a trail of noxious adverse effects and serious complications [6,7]. Therefore, failure of existing treatments necessitates the search for safer natural alternatives.
Royal Jelly (RJ), a creamy secretion of the hypopharyngeal and mandibular glands of bee workers, is the main food of queen bees. Because it is a rich mixture of proteins, lipids, sugars, vitamins, and minerals, RJ is considered a target nutraceutical, and it has been used to treat various health problems [8]. Lipids constitute between 7 and 18% of the content of RJ [9], and they are composed mainly of short hydroxy fatty acids with 8-12 carbon atoms in the chain and dicarboxylic acids. 10-hydroxy-trans-2-decenoic acid (10H2DA), known as royal jelly acid or queen bee acid, is a unique medium-chain unsaturated fatty acid that exists only in RJ [8,10,11].
10H2DA constitutes the vast majority of the RJ lipid content (0.75 to 3.39%), and it represents one of the main bioactive components of RJ [9]. A limited number of studies demonstrate the potential of 10H2DA for enhancing neuronal functioning. This mini review summarizes the available experimental and cell culture studies that address the effects of 10H2DA on psychiatric and neurological disorders as well as the underlying molecular mechanisms.
Evidence from Experimental Studies
Few experimental studies were conducted to test the effect of 10H2DA on neuropsychiatric disorders, and it seems that 10H2DA may counteract stress-induced destructive effects (Table 1).
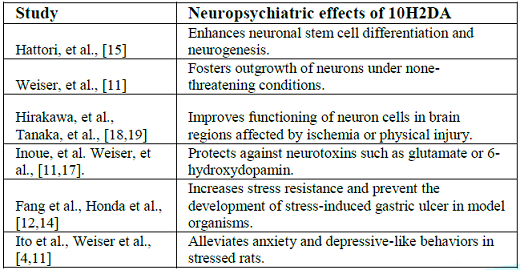
For example, 10H2DA prevented the development of stress-induced gastric ulcer in rats [12]. Needless to say, the gastrointestinal protective events of 10H2DA against stress may contribute to protection of the brain given that enteric neurons of the gut represent a main site for the production of neurotransmitters such as serotonin and that the gut-brain axis plays a major role in the development of neuropsychiatric disorders [7,13]. More, 10H2DA was found to enhance longevity and increase stress resistance of Caenorhabditis elegans against thermal, irradiation, and oxidative stress [14].
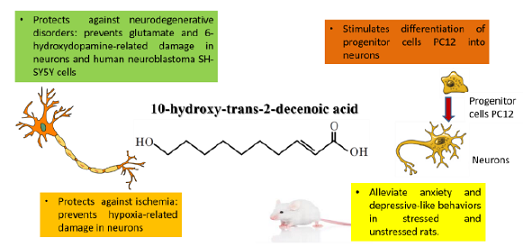
Evidence documents that 10H2DA mimics the effect of brain-derived nerotrophic factor in progenitor cells (PC12) and stimulates neurogenesis by enhancing neuronal differentiation from progenitor nerve cells (PC12) [15]. Simultaneous intraperitoneal administration of 10H2DA (once a day for 3 weeks) in rats exposed to stress resulted in improvement of depressive-like behaviors [4].
Similarly, long term oral supplementation of 10H2DA (12–24 mg/kg/day for 5 months) to aging rats significantly relieved anxiety-related behaviors as it increased the amount of time spent in the open arm of the elevated plus maze. In the same study, intraperitoneal injection of 10H2DA (100–500 g/kg/day for 3 weeks) significantly reduced anxiety-like and depressive-like behavior in stressed young mice (49–70 days old). Authors attributed this effect to the estrogenic activity of 10H2DA as well as to its neurotrophic activity—inducing the expression of BDNF through activation of Extracellular Signal-Regulated Kinase (ERK) signaling [11] (Figure 1).
Trans-2-decenoic acid ethyl ester (DAEE) and 4-Hydroperoxy-2-decenoic acid ethyl ester (HPO-DAEE) are 10H2DA-related esters that represent artificially synthesized derivatives of RJ lipids [16]. These derivatives demonstrate neuroprotective effects. Both DAEE and HPO-DAEE significantly prevented cell death in human neuroblastoma SH-SY5Y cells that were challenged with 6-hydroxydopamine (6-OHDA) as a model of Parkinsons disease [17]. Intraperitoneal administration of DAEE (100 and 150 μg/kg body weight) after hemisection of the spinal cord or unilateral permanent middle cerebral artery occlusion in rats significantly decreased the lesion size, prevented neurological deficits such as motor paralysis, and improved functional recovery of the remaining intact neurons in damaged regions. These effects were attributed to DAEE activation of ERK1/2, and increased expression of bcl-2 and BDNF in the injury site [18,19].
Possible Mechanisms Underlying the Action of 10H2DA
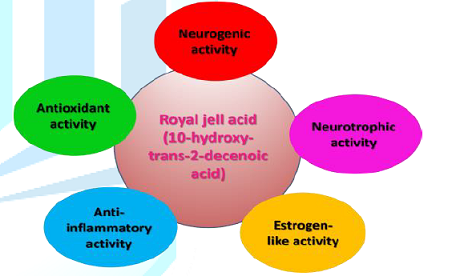
Figure 2 summarizes the most probable mechanisms through 10H2DA might affect the Central Nervous System (CNS) in order to produce therapeutic effects, namely: neurogenic and neurotrophic activity, estrogen-like activity, antioxidant activity, and anti-inflammatory activity. The following section explores these activities in detail.
Neurogenic and Neurotrophic Activity
A main mechanism through which 10H2DA exerts its therapeutic activity involves enhancement of neurogenesis and neuronal functions. It is reported that 10H2DA increases neurogenesis, but decreases glial generation, of cultured neural stem/progenitor cells. The exact mechanism involved is not clear; however, the authors suggested that 10H2DA mimics the effect of BDNF [10]. Neurons treated with 10H2DA significantly grew larger and made more interconnections with one another. It also prevented cellular death and increased properly polarized mitochondria in neurons challenged with glutamate and hypoxia as models of age-related neurodegeneration and stroke compared with untreated controls [11].
In addition, treatment of neuron cell cultures of embryonic rats with DAEE resulted in increase of synapse-specific proteins [20].
The neurogenesis, neurite outgrowth-promoting activity, synapse formation- promoting activity, and neuroprotective properties of 10H2DA and its derivatives are attributed to their neurotrophin-like effect fostering the expression of bcl-2, BDNF, neurotrophin-3, and synapse-specific proteins (e.g., synaptophysin, synapsin-1, and syntaxin) which is mediated by the phosphorylation of extracellular signal-regulated kinase 1 or 2 (ERK1/2), Mitogen Activated Protein Kinase (MAPK), and cAMP Response Element-Binding Protein (CREB) in neurons [18-20].
Estrogen-Like Activity
Among the various fatty acids of RJ, 10H2DA, 10-hydroxydecanoic acid, trans-2-decenoic acid, and 24-methylenecholesterol were found to affect estrogen receptors and mediate estrogen signaling by modulating the activity of Estrogen Receptors (ERs) ERα, ERβ [21,22]. Indeed, RJ fatty acids favorably bind to ERα at the co-activator-binding site [21]. ERs regulate gene expression and transcriptional processes through several mechanisms: 1) binding of estrogen to receptors in the nucleus, then ERs dimerize and bind to Estrogen Response Elements (EREs) located in the promoters of target genes; 2) protein-protein interactions with other DNA-binding transcription factors in the nucleus; and 3) altering functions of cytoplasmic proteins through mediation of nongenomic actions of estrogen leading to regulation of gene expression [23]. This effect is likely to contribute to the neurogenic effects of these fatty acids since estrogen can modulate cell proliferation and the expression of genes associated with brain function and body composition [22].
Antioxidant Activity
Counteracting oxidative stress and enhancing antioxidant capacity represent another mechanism through which RJ lipids and their derivative might work. HPO-DAEE, 10H2DA, and two other main fatty acids that exist in RJ (10-hydroxydecanoic acid and sebacic acid) stimulated the expression of Extracellular Superoxide Dismutase (ECSOD) in THP-1 cells through enrichment of acetylated histone H3 and H4 in the in the proximal promoter region of ECSOD whereas only HPO-DAEE activated the phosphorylation of ERK [16]. Nonetheless, reports on the antioxidant effects of RJ fatty acids in the CNS are controversial. 10H2DA, 10-hydroxydecanoic acid, and sebacic acid failed to counteract 6-OHDA-induced cellular death in in human neuroblastoma SH-SY5Y cell cultures. However, both DAEE and HPO-DAEE significantly prevented cell death and stimulated the production of antioxidant enzymes such as heme oxygenase-1 (HO-1).
HPO-DAEE demonstrated stronger antioxidant effect compared with DAEE [17]. On the contrary, 10H2DA (300 mM) caused acute massive drop of the mitochondrial electrical potential along with a simultaneous reduction of the NAD(P)H signal in astrocytes, but not in neurons. Despite blocking of the respiratory chain, intracellular ATP levels were unchanged secondary to a compensatory mechanism that involved stimulation of glycolysis and augmentation of lactate formation by 49.6% [24].
Anti-Inflammatory Activity
One possible mechanism through which 10H2DA functions is by counteracting inflammation indicated in a number of studies by NO production, which occurs as a result of activation of NF-κB that follows TNF-α production; the latter was stimulated by cellular challenge (e.g., with Lipopolysaccharide (LPS) and interferon (IFN)-γ) [3,25].
Nevertheless, reports on the molecular events involved in the anti-inflammatory effects of 10H2DA are inconsistent. 10H2DA, 10-hydroxydecanoic acid, and sebacic acid inhibited the release of the major inflammatory-mediators, NO, and interleukin-10 in a dose-dependent fashion, whereas only sebacic acid inhibited TNF-𝛼 production in LPS-challenged RAW264.7 macrophages. These effects were mediated by regulation of proteins involved in MAPK and NF-𝜅B signaling pathways [26].
Similarly, 10H2DA inhibited the production of NO and TNF-α induced by LPS, IFN-β, and IFN-γ challenge through modulation of cellular responses to these components via suppression of IFN-β-induced NF-κB signaling and IFN-γ-mediated induction of interferon regulatory factor (IRF)-8. On the contrary, 10H2DA had no effect on LPS-stimulated IFN-β production, IFN regulatory factor-1 induction and IFN-stimulated response element activation, which are required for NOS induction [3,25].
Moreover, treating rheumatoid arthritis synovial fibroblasts with 10H2DA (0.5 mM, 1 mM, and 2 mM) for 2 h followed by stimulation with TNF-α (10 ng/ml) for 2 h had no effect on ERK activity, NF-κB DNA-binding activity and IκBα degradation. Meanwhile, 10H2DA caused blocking of p38 kinase and c-Jun N-terminal kinase–activator protein-1 (AP-1) signaling pathways [27].
Conclusion
This review indicates that 10H2DA possesses neurotrophic, estrogen-like, antioxidant, and anti-inflammatory properties, which suggest that 10H2DA might be a promising avenue for ameliorating symptoms of depression and anxiety as well as for improving neuronal survival and functional recovery after CNS injury. Synthesized derivatives of 10H2DA (DAEE and HPO-DAEE) demonstrate superior antioxidant and neurotrophic properties compared with 10H2DA and other RJ fatty acids [16,17]. They even uphold an extra merit of functioning at better conditions (e.g., neutrality) whereas 10H2DA produces its effects under certain conditions (e.g., it prevents the development of transplantable AKR leukemia when the pH is below 5.6) [28].
Thus, it might be crucial to compare the biological activities of 10H2DA, its derivatives, and other RJ fatty acids, which seemed to produce effects similar to 10H2DA. Remarkably, most of the reported estrogenic, antioxidant, and anti-inflammatory properties of 10H2DA were obtained from cell lines other than neurons and glial cells. Even more, the antioxidant effect of 10H2DA on neurons was suboptimal while application of 10H2DA to astrocytes inhibited the metabolism of mitochondria which could be a toxic effect given that 10H2DA suppressed gliogenesis from neural stem cells. Therefore, sound evaluation of the properties of 10H2DA and its derivatives in neurons and microglia is necessary. Experimental studies should address issues related to dosing as well as proper route and duration of administration before it could be used in clinical trials. Special attention should be given to confounders of treatment as well.
References
1. Ali AM and Kunugi H. Bee honey protects astrocytes against oxidative stress: A preliminary in vitro investigation (2019) Neuropsychopharmacol Rep 1-3.
https://doi.org/10.1002/npr2.12079
2. Czarny P, Wigner P, Galecki P and Sliwinski T. The interplay between inflammation, oxidative stress, DNA damage, DNA repair and mitochondrial dysfunction in depression (2018) Prog Neuropsychopharmacol Biol Psychiatry 80: 309-321.
https://doi.org/10.1016/j.pnpbp.2017.06.036
3. Takahashi K, Sugiyama T, Tokoro S, Neri P, and Mori H. Inhibition of interferon-gamma-induced nitric oxide production by 10-hydroxy-trans-2-decenoic acid through inhibition of interferon regulatory factor-8 induction (2012) Cell Immunol 273: 73-78. https://doi.org/10.1016/j.cellimm.2011.11.004
4. Ito S, Nitta Y, Fukumitsu H, Soumiya H, Ikeno K, et al. Antidepressant-like activity of 10-hydroxy-trans-2-decenoic acid, a unique unsaturated fatty acid of royal jelly, in stress-inducible depression-like mouse model (2012) Evidence-based Complementary and Alternative Medicine: eCAM 139140. http://dx.doi.org/10.1155/2012/139140
5. Toratani A, Soga H, Fukumitsu H, Soumiya H, Furukawa Y, et al. Caffeic acid phenethyl ester ameliorates depression- and anxiety-like behaviors of mice exposed to chronic mild stress (2014) J Neurophysiol Neurol Disord 1: 1-8.
6. Ali AM, Hassan AA and Hendawy AO. The adverse effects of antidepressant medication treatments on the offspring of women with perinatal depression (2019) Scientific Journal of Research and Review 1.
7. Ali AM and Hendawy AO. So, antidepressant drugs have serious adverse effects, but what are the alternatives? (2018b) Nov Appro Drug Des Dev 4: 555636.
http://dx.doi.org/10.19080/NAPDD.2018.04.555636
8. Kunugi H and Ali AM. Royal jelly and its components promote healthy aging and longevity: from animal models to humans (2019) Int J Mol Sci 20: 4662.
https://doi.org/10.3390/ijms20194662
9. Kocot J, Kielczykowska M, Luchowska-Kocot D, Kurzepa J, and Musik I. Antioxidant potential of propolis, bee pollen, and royal jelly: possible medical application (2018) Oxid Med Cell Longev 7074209. http://dx.doi.org/10.1155/2018/7074209
10. Hattori N, Nomoto H, Fukumitsu H, Mishima S andFurukawa S. Royal jelly and its unique fatty acid, 10-hydroxy-trans-2-decenoic acid, promote neurogenesis by neural stem/progenitor cells in vitro (2007b) Biomed Res 28: 261-266. http://dx.doi.org/10.2220/biomedres.28.261
11. Weiser MJ, Grimshaw V, Wynalda KM, Mohajeri MH, and Butt CM. Long-term administration of Queen Bee Acid (QBA) to rodents reduces anxiety-like behavior, promotes neuronal health and improves body composition (2017) Nutrients 10. http://dx.doi.org/10.3390/nu10010013
12. Fang E, Zhou H, Xu H and Xing M. Antiulcer effects of 10-hydroxy-2-decenoic in rats (1994) Chinese Pharmacological Bulletin 10.
13. Ali AM and Hendawy AO. Bee honey as a potentially effective treatment for depression: a review of clinical and preclinical findings (2018a) JOJ Nurse Health Care 9: 555764.
14. Honda Y, Araki Y, Hata T, Ichihara K, Ito M, et al. 10-Hydroxy-2-decenoic acid, the major lipid component of royal jelly, extends the lifespan of caenorhabditis elegans through dietary restriction and target of rapamycin signaling (2015) J Aging Research 425261-425261.
http://dx.doi.org/10.1155/2015/425261
15. Hattori N, Nomoto H, Fukumitsu H, Mishima S and Furukawa S. Royal jelly-induced neurite outgrowth from rat pheochromocytoma PC12 cells requires integrin signal independent of activation of extracellular signalregulated kinases (2007a) Biomed Res 28: 139-146.
http://dx.doi.org/10.2220/biomedres.28.139
16. Makino J, Ogasawara R, Kamiya T, Hara H, Mitsugi Y, et al. Royal jelly constituents increase the expression of extracellular superoxide dismutase through histone acetylation in monocytic THP-1 cells (2016) J Nat Prod 79: 1137-1143.
http://dx.doi.org/10.1021/acs.jnatprod.6b00037
17. Inoue Y, Hara H, Mitsugi Y, Yamaguchi E, Kamiya T, et al.
4-Hydroperoxy-2-decenoic acid ethyl ester protects against 6-hydroxydopamine-induced cell death via activation of Nrf2-ARE and eIF2α-ATF4 pathways (2018) Neurochemistry International 112: 288-296.
https://doi.org/10.1016/j.neuint.2017.08.011
18. Hirakawa A, Shimizu K, Fukumitsu H, Soumiya H, Iinuma M, et al. 2-Decenoic acid ethyl ester, a derivative of unsaturated medium-chain fatty acids, facilitates functional recovery of locomotor activity after spinal cord injury (2010) Neuroscience, 171: 1377-1385.
https://doi.org/10.1016/j.neuroscience.2010.10.004
19. Tanaka Y, Fukumitsu H, Soumiya H, Yoshimura S, Iwama T, et al. 2-decenoic acid ethyl ester, a compound that elicits neurotrophin-like intracellular signals, facilitating functional recovery from cerebral infarction in mice (2012) Int J Mol Sci 13: 4968-4981. https://doi.org/10.3390/ijms13044968
20. Makino A, Iinuma M, Fukumitsu H, Soumiya H, Furukawa Y, et al. 2-Decenoic acid ethyl ester possesses neurotrophin-like activities to facilitate intracellular signals and increase synapse-specific proteins in neurons cultured from embryonic rat brain (2010) Biomed Res 31: 379-386.
https://doi.org/10.2220/biomedres.31.379
21. Moutsatsou P, Papoutsi Z, Kassi E, Heldring N, Zhao C, et al. Fatty acids derived from royal jelly are modulators of estrogen receptor functions (2010) PLoS One 5: e15594-e15594. https://doi.org/10.1371/journal.pone.0015594
22. Suzuki KM, Isohama Y, Maruyama H, Yamada Y, Narita Y, et al. Estrogenic activities of fatty acids and a sterol isolated from royal jelly (2006) eCAM 5: 295-302.http://dx.doi.org/10.1093/ecam/nem036
23. Bjornstrom L and Sjoberg M. Mechanisms of estrogen receptor signaling: convergence of genomic and nongenomic actions on target genes (2005) Mol Endocrinol 19: 833-842. http://dx.doi.org/10.1210/me.2004-0486
24. Thevenet J, Marchi UD, Domingo JS, Christinat N, Bultot L, et al. Medium-chain fatty acids inhibit mitochondrial metabolism in astrocytes promoting astrocyte–neuron lactate and ketone body shuttle systems (2016) The FASEB Journal 30: 1913-1926. http://dx.doi.org/10.1096/fj.201500182
25. Sugiyama T, Takahashi K, Kuzumaki A, Tokoro S, Neri P, et al. Inhibitory mechanism of 10-hydroxy-trans-2-decenoic acid (royal jelly acid) against lipopolysaccharide- and interferon-beta-induced nitric oxide production (2013) Inflammation 36: 372-378. http://dx.doi.org/10.1007/s10753-012-9556-0
26. Chen YF, Wang K, Zhang YZ, Zheng YF and Hu FL. In Vitro anti-inflammatory effects of three fatty acids from royal jelly (2016) Mediators of inflammation 3583684-3583684. http://dx.doi.org/10.1155/2016/3583684
27. Yang XY, Yang DS, Wei Z, Wang JM, Li CY, et al. 10-Hydroxy-2-decenoic acid from Royal jelly: A potential medicine for RA (2010) J Ethnopharmacology 128: 314-321. https://doi.org/10.1016/j.jep.2010.01.055
28. Townsend GF, Brown WH, Felauer EE and Hazlett B. Studies on the in vitro antitumor activity of fatty acids: IV. The esters of acids closely related to 10-hydroxy- 2-decenoic acid from royal jelly against transplantable mouse leukemia (1961) Canadian J Biochem Physiol 39: 1765-1770. https://doi.org/10.1139/o61-195
*Corresponding author:
Citation:
Keywords
10-hydroxy-trans-2-decenoic acid, Royal jelly, Complementary therapy, Bioactive food, Neuropsychiatric disorders.


 PDF
PDF