Introduction
Across the world, diabetes has
been a major healthcare matter and given a burden for countries and districts
socially and medically. Among them, estimated adults with Type 2 Diabetes
Mellitus (T2DM) would be 415 million. Furthermore, there may be 200 million
people with undiagnosed diabetes yet. Impaired glucose variability for long
time brings T2DM patients macrovascular and microvascular complications. This
pathophysiology will increase the risk of Chronic Kidney Disease (CKD) and
Cardiovascular Disease (CVD). Consequently, improved achievement of glycemic
control less than 7% of glycated hemoglobin (HbA1c) would be necessary. For
fundamental therapy for diabetes, principle treatments in usual clinical
practice include diet therapy and pharmacotherapy [1-5].
As to nutritional therapy for
diabetes, former method of standard diet treatment was Calorie Restriction
(CR). CR has been rather difficult to improve diabetic condition and to
maintain CR for long. After that, Low Carbohydrate Diet (LCD) was introduced in
practice medical region and health care field by Dr Atkins and Bernstein. LCD
has become rather well-known for its clinical effects of glucose-lowering and
weight reduction in Western countries. In contrast, authors and co-researchers
had started LCD in Japan. We have developed three types of LCD-meal for
everyone to understand and continue LCDs. These are petite-LCD, standard-LCD
and super-LCL, including carbohydrate ratio of 40%, 26%, and 12%, respectively.
Furthermore, we have established Japan LCD Promotion Association (JLCDPA) and
developed social movement of developing LCD leading to healthy life for
everyone. We have also continued diabetic research on glucose variability,
Morbus (M) value, CR/LCD, Meal Tolerance Test (MMT) of CR/LCD, elevated ketone
bodies in the axis of pregnant mother, newborn, umbilical cord and placenta.
Thus, standard diet therapy for diabetes has been gradually changing from CR to
LCD [6-11].
Regarding pharmacotherapy for
diabetes, there have been long history and also standard therapeutic method
recommended by American Diabetes Association (ADA) and International Diabetes
Federation (IDF). Currently, several types of Oral Hypoglycemic Agents (OHAs)
and injections have been available. Among them, it is metformin that has been
evaluated to be the first provided OHA for T2DM. Primary care physicians and
also diabetologists have provided metformin to T2DM patients for a fundamental
OHA. It shows various beneficial effects, including low cost, safety, no
hypoglycemia, weight neutrality and positive results for cardiovascular
outcomes [12-14].
Consequently, there have been
various discussions about add-on to metformin as first line of OHA. In the case
of elderly diabetic, decreased Estimated Glomerular Filtration Rate (eGFR) has
to be considered for first line metformin administration. Then, dulaglutide has
been beneficial for an option of add-on therapy.
Another benefit would be clinical
convenience for elderly, which can be administered once a week with
satisfactory clinical efficacy in the diabetic practice. Its beneficial
efficacy on glycemic control and weight control were maintained at 52 weeks and
also 104 weeks. In the case of add-on treatment to sulfonylurea, administration
of dulaglutide showed significant decreased HbA1c with satisfactory achieving
target HbA1c levels. Furthermore, the beneficial point of dulaglutide is rather
safer administration for diabetic patients with higher age and also CKD [15-17].
Authors have continued clinical
practice for patients with diabetes, CKD, Non-Communicable Diseases (NCDs) and
elderly having multiple medical and health problems for years. We have large
complex medical association of hospitals, nursing homes, nursing college,
international hospitals and others. It is called the Heisei Medical Welfare
(HMW) group with more than 10000 beds, 15000 working staffs and 80 facilities [18-20].
HMW has specialized in
convalescent and chronic medical care, and has developed many facilities for
many years in Tokushima, Tokyo, Osaka, Indonesia, etc. It has important
missions and functions such as saving all people, adequate Post-Acute Care (PAC)
and Sub-Acute Care (SAC). Among our daily practice, we have experienced
impressive elderly cases with T2DM. They have received standard therapy, but
not been in satisfactory situation. We have started the administration of
dulaglutide as add-on therapy, then their clinical progress showed improvement.
From their responses in detail, we will discuss the dulaglutide administration
in the elderly with DM and the adequate parameters or outcomes that can be
assessed in their clinical courses in this article.
Case 1
Present
History
The case was 81-year-old female
patient with the diabetic duration of 1 year. She had fatty liver and right
total hip arthroplasty in 2014, followed by sometimes complaining of right
ankle pain and right calf swelling. She received the operations of intraocular
lens and has continuing eye drops, because of bilateral cataract, glaucoma,
conjunctivitis and dry eye after 2015 until now. She was diagnosed with left
peripheral facial nerve palsy and started rehabilitation and oral medication in
2018. After that, she was pointed out to have T2DM for annual health check-up
in autumn, 2019. Then, she was provided metformin 500 mg, and glimepiride 1mg
per day as OHA before dulaglutide administration.
Physicals and
Labs
She has unremarkable findings for
consciousness, vitals, and physical exams. She did not show progressed
retinopathy or nephropathy, but slight neuropathy in the hands and feet. Her
physique showed 156.9cm in height, 59.0kg in weight, Body Mass Index (
Clinical Progress
The case with T2DM was provided
Metformin 1000mg/day, Glimepiride 1mg/day as OHAs. However, her glycemic
control was not satisfactory. Then she was started to be given Dulaglutide
0.75mg once a week by injection from December, 2019 as an add-on therapy for
diabetes. After that, her glycemic variability showed remarkable improvement.
Three months later, her HbA1c and fasting blood glucose was 6.7% and 95 mg/dL (Figure 1). As for clinical symptoms,
she had no complaints concerning diabetes before and after Dulaglutide
treatment. As regards to lipids, values of HDL-C and Triglyceride did not
change significantly. In contrast, LDL-C showed a little elevated tendency by
the administration of Dulaglutide. The treatment other than Dulaglutide was not
changed. Currently, she has been on Metformin, Glimepiride, Fexofenadine
120mg/day and Dulaglutide 0.75mg/week. The treatment has been the same as
before, with the addition of Dulaglutide.
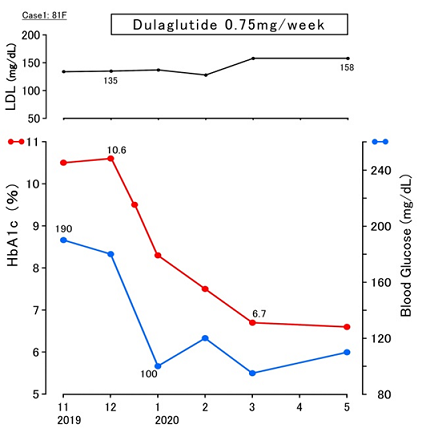
Figure 1:Clinical progress of case 1 (81F)
Case 2
Present
History
The case was 83-year-old male
patient. He was diagnosed as T2DM at the age of 56 years old, and treated for
27 years. Successively, he has suffered from arteriosclerotic diseases,
including hypertension, hyperlipidemia and angina pectoris for years from 56 to
74 years old (2010). From 2011, he had various medical health problems, such as
Gastroesophageal Reflux Disease (GERD) with lansoprazole, diabetic simple
retinopathy, iron-deficiency anemia with dried ferrous sulfate, constipation
with magnesium oxide and pneumonia during winter. Successively, diabetic
control has not been satisfactory with HbA1c around 9% for last 1-2 years. He
has been transferred to our hospital in May, 2019.
Physicals and
Labs
He showed unremarkable findings
for consciousness, vitals, lung, heart, abdomen and extremities. He showed a
little numbness in the hands and feet. His physique showed 165.0 cm in height,
65.2 kg in weight, Body Mass Index (BMI) 23.9 kg/m2. Blood tests
including complete blood count and biochemistry were in the following: WBC 6500/μL,
RBC 5.03 x 106/μL, Hb 13.7 g/dL, Plt 27.1 x 104/μL, TP
7.1 g/dL, Alb 4.5 g/dL, AST 16 IU/mL, ALT 11 IU/mL, r-GTP 22 U/L, BUN 18 mg/dL,
Cre 0.8 mg/dL, Uric Acid 4.7 mg/dL, eGFR 69.9 ml/min/1.73m2, HDL 81
mg/dL, LDL 66 mg/dL, TG 57 mg/dL, CPK 86 U/L (62-287). Data related diabetes
were HbA1c 9.4%, pre-prandial glucose 156 mg/dL. Urinalysis revealed 30 mg/dL
of protein, 1.0g of glucose g/dL, +/- of ketone bodies, negative occult blood,
0.1 mg/dl of urobilinogen and pH of 5.0. He had unremarkable findings for ECG
and chest X-P, prostate hypertrophy and colon diverticulum in abdominal CT
scan.
Clinical Progress
He was provided pharmacotherapy
for diabetes using OHAs. The HbA1c value persisted around 9.0%-9.4%, then he
was started to be given Dulaglutide 0.75 mg once a week from December, 2019 as
an add-on therapy for diabetes. After that, his HbA1c level decreased to 8.2%
in March 2020 (Figure 2). As regards
to lipid profile, LDL-C value showed increased tendency in 2-3 months, whereas HDL-C
and TG showed no changes. As for clinical symptoms, he had not shown diabetic
complaints before and after the administration of dulaglutide. His current
medication has included Metformin 1000mg, Glimepiride 1mg, Ipragliflozin 50mg,
Valsartan 80mg, rosuvastatin 2.5mg, rabeprazole 10mg and magnesium oxide 990mg
per day. These medications were not changed for several months.
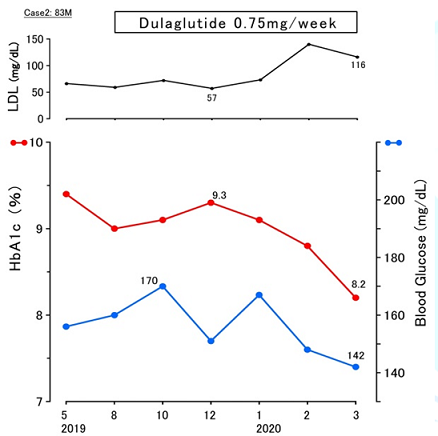
Figure 2:Clinical progress of case 2 (83M)
Discussion
Recent developments of diabetic
research and practice include the presence of Intestine Secretion Insulin (INCRETIN).
Among them, Gastric Inhibitory Polypeptide (GIP) was identified by the
purification of intestinal hormone and, Glucagon-Like Peptide-1 (GLP-1) with
the incretin action was introduced. Successively, lots of investigations
revealed the mechanism of GLP-1, and its physiological role includes various
clinical functions. They are stimulating insulin secretion, inhibiting glucagon
secretion, increasing glucose production and glucose uptake in the liver,
reducing food intake through delay of gastric emptying and central nerve system.
These developments have brought Glucagon-Like Peptide-1 Receptor Analogue (GLP-1
RA) and also Dipeptidyl Peptidase-4 Inhibitor (DPP-4i) into clinical practice
for diabetic treatment. In particular, GLP-1 RA has been used for effective Anti-Hyperglycemic
Agent (AHA), associated with lower risk of reverse effects and some weight
reduction expected. There are a few kinds of GLP-1 RA introduced so far, with
satisfactory efficacy [21-24].
Among several GLP-1RA,
dulaglutide has been effective and useful for its one a week administration. As
to add-on therapy for diabetic patients of poorly controlled OHA, comparison of
Dulaglutide once-week and glargine once-day were investigated. T2DM (n=25) were
randomly assigned into Dulaglutide and glargine groups and treated 52 weeks.
From the studies of Continuous Glucose Monitoring (CGM) in both groups, they
showed similar results of Mean Blood Glucose (MBG), Mean Amplitude of Glycemic
Excursion (MAGE) and Standard Deviation of Blood Glucose (SDBG). These results
suggested equivalent clinical efficacy for glucose variability [25].
Subcutaneous dulaglutide
(Trulicity®) has been recognized as a GLP-1 RA, which has shown clinical
efficacy for patients with T2DM for monotherapy or add-on therapy to other
OHAs. Especially, it shows effects for high-risk patients, such as elderly,
obese, those with Chronic Kidney Disease (CKD) as stage 3 or 4 and/or Atherosclerotic
Cardiovascular Disease (ASCVD). It shows the effect of significant risk
reduction for Major Adverse Cardiac Event (MACE), which includes nonfatal
stroke, nonfatal myocardial infarction and primary composite outcome comprising
CV death. As add-on therapy to Oral Hypoglycemic Agents (OHAs), there have been
a series of AWARD studies, which stands for Assessment of Weekly Administration
of LY2189265 in Diabetes. They include the add-on therapy to SGLT2 inhibitors,
insulin glargine, glimepiride and others [26-29].
The add-on therapy of Dulaglutide
has been used and investigated. There were several studies on phase 2 and phase
3 including cardiovascular meta-analysis, such as AWARD 1,2,3,4,5. Among them,
AWARD 2 was based on the add-on therapy for Metformin and Glimepiride. Its
protocol included 810 patients randomized to 3 groups, which were dulaglutide
1.5 mg, dulaglutide 0.75 mg, or glargine for 78 weeks. Baseline HbA1c (mean ±
SE) was 8.1%, and HbA1c change at 52 weeks was-1.08 ± 0.06%, -0.76 ±
0.06%-1.08% and-0.63 ± 0.06% in 3 groups, respectively [30,31].
AWARD had 5 post-hoc, pooled
analysis studies for dulaglutide administration weekly, in AWARD 1,2,3,5,6.
Among them, dulaglutide 1.5mg was added to OHA. T2DM patients (n=1424) were
investigated lipids changes for 6 months after dulaglutide therapy. There were
no significant changes in HDL-C and triglycerides, but significant decrease of
LDL-C as (-0.11 mmol/L [-4.2%], p=0.002). Furthermore, cases with higher
baseline of lipids tended to show larger reduction in respective indices [32].
In this study, two elderly T2DM
patients showed improved HbA1c values by the administration of dulaglutide. It
is also noteworthy that changes in LDL were found to be higher than the
previous values in the lipid profile. Therefore, the influence of dulaglutide
for lipids will be described.
In previous report, dulaglutide
therapy for 26 weeks brought significant decrease of LDL-C with no significant
change in HDL-C. Such effects seem to be mediated by the alterations in the
expression of genes and proteins, which are involved in lipid metabolism. In
particular, the expression change of Adipocyte complement-related protein of 30
kDa (Acrp30) may be influenced. Acrp30 has a role of regulation for glucose and
lipid homeostasis from several studies. The expression and serum levels of
Acrp30 are reduced in patients with obesity and insulin resistance [33-35].
There was another recent
report as to the lipid changes for administration of dulaglutide. The protocol
was to switching therapy from sitagliptin 50 mg daily to dulaglutide 0.75mg
weekly in T2DM patients. Clinical efficacy was observed in decreased HbA1c from
7.80% to 7.25% for 24 weeks. Further, the degree of decreased HbA1c (delta
HbA1c) showed significant correlation with baseline HbA1c, fasting plasma
glucose and Body Mass Index (BMI). Simultaneously, lipids profile was
investigated. As a result, LDL-C was significantly decreased from 90.0 mg/dL to
78.6 mg/dL (p=0.0010), and HDL-C and TG showed no significant changes [36].
Among these, the correlation
between the previous LDL value and decreased value of LDL (delta-LDL) shows a
significant correlation. In other words, as the pre-LDL is higher, delta LDL
becomes higher. Similarly, as the pre-LDL is lower, delta LDL becomes lower
and/or becomes minus value. There are actual data of delta-LDL after
Dulaglutide therapy. The results were that i) 6 cases (pre-LDL<70 mg/dL)
showed increased LDL-C in all cases, ii) 21 cases (71 mg/dL<pre-LDL<100
mg/dL) showed increased LDL for 6 cases, and decreased LDL for 15 cases, iii) 6
cases (pre-LDL>101 mg/dL) showed decreased LDL-C in all cases. For the
correlations of delta-LDL, significant correlation was found between delta-LDL
and previous LDL (p=0.0012), but no significant correlation was found between
delta-LDL and TG, AST, or ALT. These results suggest that lipid metabolism
would be modified by the administration of Dulaglutide [36].
In this study, two subjects were
elderly patients who were 81 and 83 years old. Their eGFR showed 63.2 and 69.9
ml/min/1.73m2, which were more than 60 ml/min/1.73m2 and
were probably from age-related decline. One of the beneficial points of
Dulaglutide would be possible administration to patients with CKD. There was a
multicenter, open-label trial done at 99 sites in nine countries, which was
AWARD-7 study [37].
Subjects were T2DM with CKD (stage 3-4), which
were randomly assigned to three group of dulaglutide 1.5 mg, dulaglutide 0.75
mg, and daily insulin glargine. As a result, once-weekly dulaglutide showed
glycemic control with similar degree of insulin glargine, associated with
reduced decline in eGFR. Consequently, dulaglutide seems to be safe and
effective for glycemic control for 3-4 stage CKD. In this perspective, stage 3
or 4 means that eGFR shows 30-59, 15-29 ml/min/1.73m2, respectively.
Consequently, dulaglutide can be used for patients with impaired renal
function.
There are some limitations in
this study. Subjects were 2 elderly diabetic patients, who showed improved
glucose variability to Dulaglutide with some elevation of LDL values. We cannot
suggest the mechanism of Dulaglutide for lipids profile from small numbers of
clinical cases, but would investigate other elder cases successively.
Furthermore, authors have continued comprehensive medical practice with chronic
medical care for years. Each patient has various needs of adequate Post-Acute
Care (PAC) and Sub-Acute Care (SAC), then we should deal with each problem from
holistic point of view.
In summary, we described two
diabetic patients treated by dulaglutide and some discussions concerning to GLP-1RA,
lipids profiles and others. Some recommendations for dulaglutide administration
may include elderly, subjects with CKD or decreased eGFR for expecting better
clinical results. This report would be expected to become a reference for
diabetic practice and research in the future.
Ethical Considerations
Current research has been
basically conducted in compliance with the ethical principles presented on the
Declaration of Helsinki. In addition, there was commentary for the Ethical
Guidelines against the Research in the medical field for Human beings and also
against the conduction of the Good Clinical Practice (GCP). Regarding the
protection of human rights, some ongoing considerations were present.
Furthermore, we applied adequately “Ethical Guidelines for Epidemiology
Research” for the related guideline. These principles were originated from
Japan by the Ministry of Health, Labor and Welfare and also by the Ministry of
Education, Culture, Sports, Science and Technology.
As regards to the current subjects,
the written informed consents from two patients were obtained. Moreover, we
established the ethical committee for the clinical research in the Hakuai
Memorial Hospital. The committee had several professional persons including the
president, directors of the administration and pharmaceutical departments, the
head nurse of the nursing department, and also related experts in the medical
and legal specialties. There were fully and satisfactory discussion for the
research content and conclusion confirmation that this study would be adequate
without any problems and agreed with all participants.
Conflict of
Interest
The author declares no conflict
of interest. Relationship of related organizations would be explained. Dr.
Takehisa has been the top director of HMW group with many hospitals, and a
physician of Hakuai Memorial Hospital (HMH) which is one of HMW. Current cases
are from HMH where Dr. Bando has been an advisor from diabetic and primary care
points of views. Japan LCD Promotion Association (JLCDPA) was established for
the development of LCD education by Dr. Ebe, Kyoto, Japan. Dr. Bando is one of
the main members of JLCDPA and often gives lectures and advices for various
opportunities.
References
1. Buse
JB, Wexler DJ, Tsapas A, Rossing P, Mingrone G, et al. 2019 update to:
management of hyperglycaemia in type 2 diabetes, 2018. a consensus report by
the american diabetes association (ADA) and european association for the study
of diabetes (EASD) (2019) Diabetologia 63: 221-228. https://doi.org/10.1007/s00125-019-05039-w
2. Shin
JI. Second-line glucose-lowering therapy for type 2 diabetes mellitus (2019)
Curr Diab Rep 19: 54. https://doi.org/10.1007/s11892-019-1171-0
4. American
Diabetes Association. 10. Cardiovascular disease and risk management: standards
of diabetes care (2019) Diabetes Care 4: 103-123.
5. American
Diabetes Association. 6. Glycemic Targets: Standards of Medical Care in
Diabetes-2020 (2020) Diabetes Care 43: 66-76. https://doi.org/10.2337/dc20-s006
6. Atkins
RC. Dr. Atkins' new diet revolution (2002) Harper, United states.
7. Bernstein
R. Dr. Bernstein's diabetes solution newly revised and updated (2007) Little
Brown and Company, United States.
8. Feinman
RD, Pogozelski WK, Astrup A, Bernstein RK, Fine EJ, et al. Dietary carbohydrate
restriction as the first approach in diabetes management: Critical review and
evidence base (2015) Nutrition 31: 1-13. https://doi.org/10.1016/j.nut.2014.06.011
9. Ebe
K, Ebe Y, Yokota S, Matsumoto T, Hashimoto M, et al. Low carbohydrate diet (LCD)
treated for three cases as diabetic diet therapy (2004) Kyoto Med Associat J
51: 125-129.
10. Bando
H, Ebe K, Muneta T, Bando M and Yonei Y. Effect of low carbohydrate diet on
type 2 diabetic patients and usefulness of M-value (2017) Diabetes Res Open J 3:
9-16. https://doi.org/10.17140/DROJ-3-130
11. Muneta
T, Kagaguchi E, Nagai Y, Matsumoto M, Ebe K, et al. Ketone body elevation in
placenta, umbilical cord, newborn and mother in normal delivery (2016) Glycat
Stress Res 3: 133-140. https://doi.org/10.24659/gsr.3.3_133
12. International
Diabetes Federation.
13. Mauricio
D, Alonso N and Gratacòs M. Chronic diabetes complications: the need to move
beyond classical concepts (2020) Trends Endocrinol Metab 31: 287-295. https://doi.org/10.1016/j.tem.2020.01.007
14. Palmer
SC, Mavridis D, Nicolucci A, Johnson DW, Tonelli M, et al. Comparison of
clinical outcomes and adverse events associated with glucose-lowering drugs in
patients with type 2 diabetes: a meta-analysis (2016) JAMA 316: 313-324. http://doi.org/10.1001/jama.2016.9400
15. Nauck
M, Weinstock RS, Umpierrez GE, Guerci B, Skrivanek Z, et al. Efficacy
and safety of dulaglutide versus sitagliptin after 52 weeks in type 2 diabetes
in a randomized controlled trial (AWARD-5) (2014) Diabetes Care 37: 2149-2158. https://doi.org/10.2337/dc13-2761
16. Weinstock
RS, Guerci B, Umpierrez G, Nauck MA, Skrivanek Z, et al. Safety and efficacy of
once-weekly dulaglutide versus sitagliptin after 2 years in metformin-treated
patients with type 2 diabetes (AWARD5): a randomized, phase III study (2015)
Diabetes Obes Metab 17: 849-858. https://doi.org/10.1111/dom.12479
17. Dungan KM, Weitgasser R, Perez Manghi F, Pintilei E, Fahrbach JL, et al. A 24-week study to evaluate the efficacy and safety of once-weekly dulaglutide added on to glimepiride in type 2 diabetes (AWARD-8) (2016) Diabetes Obes Metab 18: 475-482. https://doi.org/10.1111/dom.12634
18. Shimizu
E, Takehisa Y, Bando H, Fujita M, Kusaka Y, et al. Effective SGLT2 inhibitor
for patient with type 2 diabetes mellitus (T2DM) and depression (2020) Diab Res
2: 26-32. https://doi.org/10.36502/2020/droa.6160
19. Nishikori
Y, Bando H, Yoshioka A, Fujita M, Kusaka Y, et al. Trials of Additional
Effective Movements for Music Therapy Session for the Elderly (2020) Curr Res
Complement Altern Med 4: 138.
20. Heisei Medical Welfare (HMW) group.
21. Kreymann
B, Williams G, Ghatei MA and Bloom SR. Glucagon-like peptide-1 7-36: a
physiological incretin in man (1987) Lancet 330:1300-1304. https://doi.org/10.1016/s0140-6736(87)91194-9
22. Smith
EP, An Z, Wagner C, Lewis AG, Cohen EB, et al. The role of β cell glucagon-like
peptide-1 signaling in glucose regulation and response to diabetes drugs (2014)
Cell Metab 19: 1050-1057. https://doi.org/10.1016/j.cmet.2014.04.005
23. Gurung
T, Shyangdan DS, O'Hare JP, Waugh N. A novel, long-acting glucagon-like peptide
receptor-agonist: dulaglutide (2015) Diabetes Metab Syndr Obes 10: 363-386. https://doi.org/10.2147/dmso.s34418
24. Hedrington MS and Davis SN. Oral semaglutide for the treatment of type 2 diabetes (2019) Expert Opinion Pharmaco 20: 133-141. https://doi.org/10.1080/14656566.2018.1552258
25. Wang
J, Li HQ, Xu XH, Kong XC, Sun R, et al. The effects of once-weekly dulaglutide
and insulin glargine on glucose fluctuation in poorly oral-antidiabetic
controlled patients with type 2 diabetes mellitus (2019) Biomed Res Int 2019: 2682657.
https://doi.org/10.1155/2019/2682657
26. Scott
LJ. Dulaglutide: A Review in Type 2 Diabetes (2020) Drugs 80: 197-208. https://doi.org/10.1007/s40265-020-01260-9
27. Ludvik
B, Frias JP, Tinahones FJ, Wainstein J, Jiang H, et al. Dulaglutide as add-on
therapy to SGLT2 inhibitors in patients with inadequately controlled type 2
diabetes (AWARD-10): a 24-week, randomised, double-blind, placebo-controlled
trial (2018) Lancet Diabetes Endocrin 6: 370-381. https://doi.org/10.1016/s2213-8587(18)30023-8
28. Pozzilli
P, Norwood P, Jodar E, Davies MJ, Ivanyi T, et al. Placebo-controlled,
randomized trial of the addition of once-weekly glucagon-like peptide-1
receptor agonist dulaglutide to titrated daily insulin glargine in patients
with type 2 diabetes (AWARD-9) (2017) Diabetes Obes Metab 19: 1024-1031. https://doi.org/10.1111/dom.12937
29. Dungan KM, Weitgasser R, Perez Manghi F, Pintilei E, Fahrbach JL, et al. A 24-week study to evaluate the efficacy and safety of once-weekly dulaglutide added on to glimepiride in type 2 diabetes (AWARD-8) (2016) Diabetes Obes Metab 18: 475-482. https://doi.org/10.1111/dom.12634
30. Weinstock
RS, Guerci B, Umpierrez G, Nauck MA, Skrivanek Z, et al. Safety and efficacy of
once-weekly dulaglutide versus sitagliptin after 2 years in metformin-treated
patients with type 2 diabetes (AWARD-5): a randomized, phase III study (2015)
Diabetes Obes Metab 17: 849-858. https://doi.org/10.1111/dom.12479
31. Giorgino
F, Benroubi M, Sun JH, Zimmermann AG and Pechtner V. Efficacy and safety of
once-weekly dulaglutide vs. insulin glargine in patients with type 2 diabetes
on metformin and glimepiride (AWARD-2) (2015) Diabetes Care 38: 2241-2249. https://doi.org/10.2337/dc14-1625
32. Pavo
I, García-Pérez LE, Tao Y, Thieu VT, Sattar NA, et al. Changes in lipid
concentrations in patients with type 2 diabetes on once-weekly dulaglutide 1.5
mg: Post hoc pooled analysis of the AWARD trials (2018) Diabetologie und
Stoffwechsel 13: 54-55. https://doi.org/10.1055/s-0038-1641919
33. Wysham
C, Blevins T, Arakaki R, Colon G, Garcia P, et al. Efficacy and safety of
dulaglutide added onto pioglitazone and metformin versus exenatide in type 2
diabetes in a randomized controlled trial (AWARD-1) (2014) Diabetes Care 37: 2159-2167.
https://doi.org/10.2337/dc13-2760
34. Li
L, Miao Z, Liu R, Yang M, Liu H, et al. Liraglutide prevents
hypoadiponectinemia-induced insulin resistance and alterations of gene
expression involved in glucose and lipid metabolism (2011) Mol Med 17: 1168-1178.
https://doi.org/10.2119/molmed.2011.00051
35. Tsao
TS, Lodish HF and Fruebis J. ACRP30, a new hormone controlling fat and glucose
metabolism (2002) European J Pharmacol 440: 213-221. https://doi.org/10.1016/s0014-2999(02)01430-9
36. Iwasaki
T, Kessoku T, Higurashi T, Taguri M and Yoneda M. Low body mass index and old
age are useful in predicting the hemoglobin A1c-lowering effect of switching
from sitagliptin to dulaglutide in Japanese patients with type 2 diabetes
mellitus: a single-center, open-label, single-arm, pilot study (2018) Diabetol
Intern 9: 189-195. https://doi.org/10.1007/s13340-018-0348-0
37. Tuttle
KR, Lakshmanan MC, Rayner B, Busch RS, Zimmermann AG, et al. Dulaglutide versus
insulin glargine in patients with type 2 diabetes and moderate-to-severe
chronic kidney disease (AWARD-7): a multicentre, open-label, randomised trial
(2018) The Lancet Diabetes Endocrinol 6: 605-617. https://doi.org/10.1016/s2213-8587(18)30104-9
*Corresponding author
Hiroshi Bando,
Tokushima University/Medical Research, Tokushima, Japan, Tel: +81-90-3187-2485,
E-mail: pianomed@bronze.ocn.ne.jp
Citation
Takehisa Y and
Bando H.
Elderly diabetic patients with effective add-on therapy of dulaglutide as a
GLP-1 receptor analogue (GLP-1 RA) (2020) Edel J Biomed Res
Rev 2: 31-35.
Keywords
Dulaglutide, Glucagon-like Peptide-1 Receptor
Analogue, Type 2 Diabetes Mellitus, Oral Hypoglycemic Agents, Heisei Medical
Welfare.


 PDF
PDF