Introduction
Addisons
disease, also known as primary adrenal insufficiency, is an endocrinological
condition with a deficiency in the production of glucocorticoids and
mineralocorticoids from the adrenal cortex [1]. Its a very life-threatening
disease that is often overlooked due to the unspecific range of symptoms
patients usually present with, thus a prompt diagnosis is essential.
There
have been multiple etiologies attributed to the development of Addisons
disease, including infections, drugs and even genetic factors. However, the
most common cause remains to be an autoimmune destruction of the adrenal cortex
[2].
Despite
the well-established relationship between cardiovascular complications and
Addisonian crisis, its thought to be limited mainly to hypovolemic hypotension,
syncope and arrhythmias, resolving with volume repletion and glucocorticoids
administration [3].
The
presentation of cardiomyopathy and heart failure in patients who have no
previous history of adrenal or cardiac abnormalities is not very common and
only 6 cases are reported as such in the literature [4-10]. However, in all
cases cardiomyopathy seemed to have been improved post glucocorticoids.
We
report a case of a 57 years old lady who presented with an acute onset of
adrenal insufficiency then developed severe HF that resolved on serial
echocardiography post hydrocortisone administration. The clinical presentation,
course of management and outcome will be discussed.
Case
A
57 years old female not known to have any medical complains, was brought in to
the emergency department with a few hours history of agitation, restlessness and
disturbed level of consciousness. She was completely alert the night before
until the morning of her admission when she was confused, unable to recognize
her family and very agitated. There was no documented loss of consciousness or
any seizure attacks. There were also no complaints of any chest symptoms or
abdominal pain.
The
patients only past history was chronic back pain on analgesia. On initial
assessment the patient had a GCS of 8, then was very agitated and sedated by
the emergency department with midazolam. She was hypotensive with a blood
pressure of 90/60, afebrile, maintaining saturation on a face mask attached to
8L oxygen. No abnormal skin pigmentations were noted, and no obvious dysmorphic
features. Her initial biochemical panel showed a hyponatremia of 121mmol/l and
hyperkalemia of 8.15 mmol/l, as well as a sight elevated Creatinine of 117
umol/l (Table 1). Her VBG showed deteriorating mixed acidosis and the patient
was awaiting intubation after the CT brain.
The
patient also underwent a cardiology review while in the emergency department,
her initial ECG showed normal sinus rhythm with down-sloping ST elevation in
V1-V2 attributed to hyperkalemia with no reciprocal changes. A repeat ECG after
hyperkalemia correction showed a descend in the down-sloping ST elevation to
isoelectric line, T wave inversion in anterior leads (Figure 1). Her bedside troponin was negative and her bedside ECHO
showed an ejection fraction of 60% with no regional wall motion abnormalities.
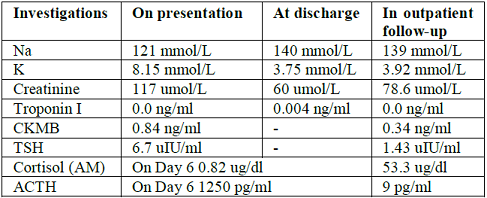
Table 1: Investigation readings.
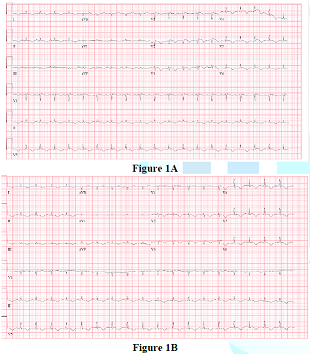
Figure 1: ECG after hyperkalemia correction showed a descend in the down-sloping ST elevation to isoelectric line, T wave inversion in anterior leads
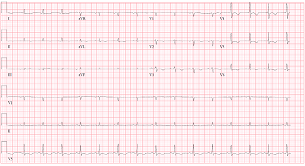
Figure 2: ECG showed poor R wave progression with T wave inversion in anterior leads.
The
patient was later on intubated, admitted to the intensive care unit and started
on hydrocortisone (200mg IV stat then infusion) during her hospital stay in the
intensive care unit, the patient was started on continuous renal replacement
therapy (CRRT), and her hydrocortisone infusion was converted to boluses of
100mg IV every 8 hours after an endocrine review. Her blood pressure was
maintained on the hydrocortisone and intravenous fluids. CT brain ruled out any
brain lesions, and a CT abdomen was also done which ruled out any adrenal
masses. She remained hyponatremic, on mechanical ventilation and sedated on
Propofol. TSH was 6.7 uIU/ml while LH and FSH were both normal (Table 1). On her second admission day,
a departmental echocardiography showed severe hypokinesia of mid to apical LV
segments and an overall systolic function of 20%. A further cardiology review
was requested as the Troponin was slightly elevated (highest of 5 ng/L), ECG
showed poor R wave progression with T wave inversion in anterior leads (Figure 2). An impression of stress
induced cardiomyopathy secondary to Addisonian crisis was made and the patient
was kept on full anti-ischemic measures. On
Day 5, the patient was extubated, and started self-ventilation on minimal O2
support, her clinical condition started to improve and her BP remained
adequate, thus she was transferred to the medical ward.
On
Day 6, her hydrocortisone was stopped for 24 hours before cortisol levels were
sent. The patients BP was maintained above 90 mmhg systolic off the
hydrocortisone, cortisol levels came back as 0.82 ug/dl, ACTH was 1250 pg/ml
and the patient was started on hydrocortisone tablets (10mg in the morning and
5mg in the evening). A repeat echocardiography was done which showed an
improvement in the ejection fraction, with hypokinetic mid anteroseptum, mid
inferoseptum and entire apex with an overall systolic function of 40%-45%.
The
patient was discharged on day 12 with further outpatient review in both
endocrinology and cardiology clinics. Follow up echocardiography studies were
done which demonstrated improvement in the ejection fraction with the latest
one showing an ejection fraction of 60% with well-preserved contractility. The
patient also underwent further cardiology assessments as an outpatient,
including a CT coronary angiography and a dobutamine stress test which were all
normal.
DiscussionPrimary
adrenal insufficiency is an endocrinological condition with a decrease in the
production of glucocorticoids and mineralocorticoids from the adrenal cortex,
most commonly secondary to an autoimmune destruction of the adrenal cortex.
Cardiovascular manifestations of Addisons mainly involve hypotension due to
volume depletion, syncope and arrhythmias secondary to electrolytes imbalance.
There
have been few cases in the literature demonstrating some sort of a relationship
between Addisonian crisis and reversible cardiomyopathy that usually resolved
after a stress-dose of glucocorticoids, and this case is an evident
demonstration of that The cause of left ventricular dysfunction in Takotsubo
cardiomyopathy is still unknown, It may be caused by a catecholamine-mediated
myocardial stunning secondary to either a myocardial ischemic injury or
epicardial spasm with metabolic injury. Catecholamines can directly damage the
myocardium. In Takotsubo cardiomyopathy, physical stress is considered as a
major predisposing factor [11,12].
Conclusion
This
case demonstrated a fairly uncommon presentation of stress induced
cardiomyopathy secondary to adrenal insufficiency, which resolved after high
doses of glucocorticoids. This is still a field of mystery and interest for
many in the medical field, more research is needed to demonstrate the mechanism
of such phenomenon and methods to tackle it.
References
1.
Nieman
LK and Chanco Turner ML. Addisons disease (2006) Clinics in Dermatology 24: 276-280.
https://doi.org/10.1016/j.clindermatol.2006.04.006
2.
Bensing
S, Hulting A, Husebye ES, Kämpe O and Løvås K. Management of endocrine disease:
epidemiology, quality of life and complications of primary adrenal
insufficiency: a review (2016) European J Endocrinology 175: 107-116. https://doi.org/10.1530/EJE-15-1242
3.
Punnam
SR, Gourineni N and Gupta V. Takotsubo cardiomyopathy in a patient with Addison
disease (2010) Int J Cardiol 144: 34-36. https://doi.org/10.1016/j.ijcard.2008.12.191
4.
Cushner
GB, Zahler SF and Hills AG. Untreated Addisons disease complicated by pulmonary
congestion due to left ventricular failure (1963) Annals Internal Med 58: 341-346.
DOI:
10.7326/0003-4819-58-2-341
5.
Afzal
A and Khaja F. Reversible cardiomyopathy associated with Addisons disease
(2000) Canadian J Cardiol 16: 377-379.
6.
Bhattacharyya
A and Tymms DJ. Heart failure with fludrocortisone in Addisons disease (1998) J
Royal Society Med 91: 433-434.
7.
Krishnamoorthy
A, Mentz RJ, Hyland KA, McMillan
EB, Patel CB, et al. A crisis of the heart: an acute reversible cardiomyopathy
bridged to recovery in a patient with addisons disease (2013) ASAIO Journal 59:
668-670. https://doi.org/10.1097/MAT.0000000000000001
8.Wolff
B, Machill K, Schulzki I, Schumacher D and Werner D. Acute reversible
cardiomyopathy with cardiogenic shock in a patient with Addisonian crisis: a
case report (2007) Int J Cardiology. 116: e71-e73. https://doi.org/10.1016/j.ijcard.2006.07.207
9.
Mozovleska
V, et al. Addisons Disease and Dilated Cardiomyopathy: A Case Report and Review
of the Literature (2016) Case Rep Cardiol 2016: 4362514. http://dx.doi.org/10.1155/2016/4362514
10.
Wolff
B, Machill
K, Schulzki I, Schumacher D and Werner D. Acute reversible cardiomyopathy with
cardiogenic shock in a patient with Addisonian crisis: A case report (2007) Int
J Cardiol 116: 71-73. https://doi.org/10.1016/j.ijcard.2006.07.207
11.
Mann
D, Kent RL, Parsons B and Cooper G 4th. Adrenergic effects on the biology of
the adult mammalian cardiocyte (1992) Circulation 85: 790-804.
12.
White
M, Wiechmann RJ, Roden RL, Hagan MB, Wollmering MM, et al. Cardiac
betaadrenergic neuroeffector systems in acute myocardial dysfunction related to
brain injury. Evidence for catecholamine-mediated myocardial damage (1995)
Circulation 92: 2183-2189.
*Corresponding author:
Dr Maysa Salman, 22 Malone Beeches, BT9 6UB, Belfast. United Kingdom, Tel: 0044-7498900777/ 00973 32002990, E-mail: maysah.almulla@gmail.com
Citation:
Salman M, AlKhoury J, AlSherooqi R and Assar W. Reversible Stress Induced Cardiomyopathy in Adrenal Insufficiency (2018) Clinical Cardiol Cardiovascular 1: 6-8.